Nov 24 - Lecture 36 ~ Apoptosis
apoptosis is a gentle form of cell death (regulated dissolution)
compare to necrosis — a cellular explosion
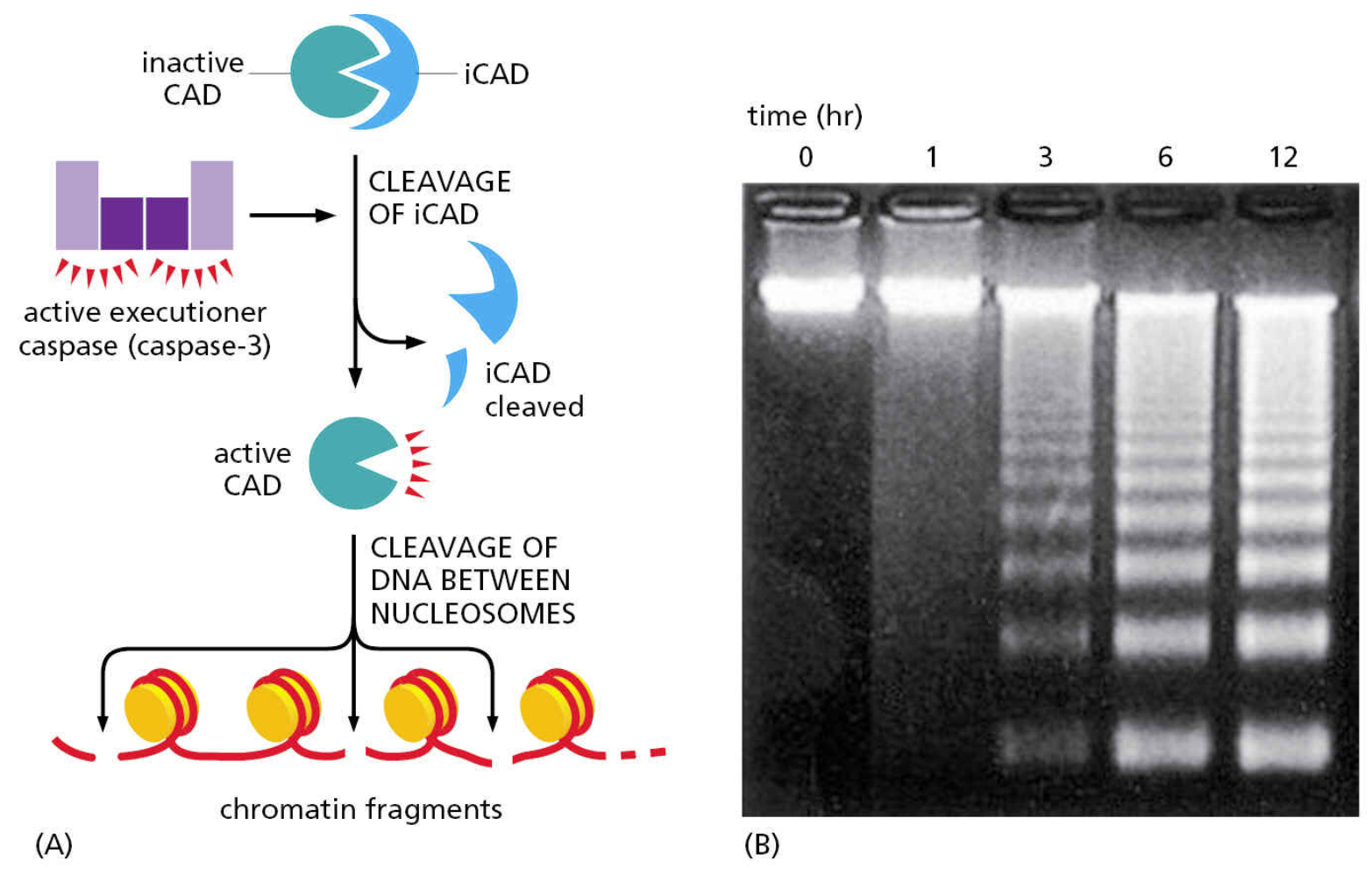
DNA gets fragmented; nuclear lamins break down & actin rearrangement to form blebbing
cell death is as important to development as cell proliferation
embryonic development is, of course, dependent upon proliferation of the initial zygote into the multitudes of cells that make up body tissues
Not all cells in a developing tissue, however, contribute to the final structure
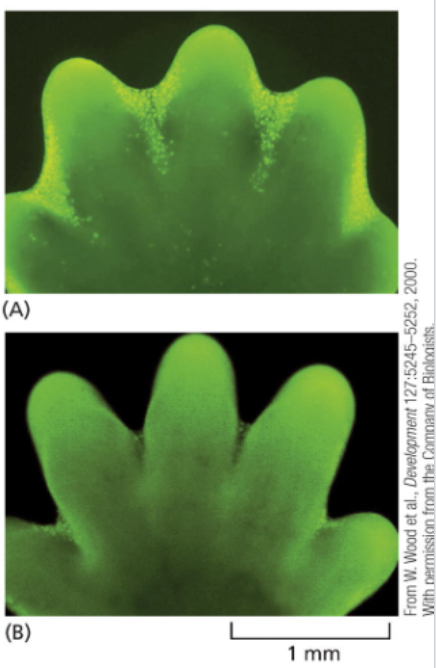
developing mouse paw cells stained in a yellow-green color are undergoing apoptosis to produce distinct digits
Failure of this process in mice (and humans) leaves a persistent webbing between the digits
As a tadpole metamorphoses into an adult frog, the cells making up its tail likewise die in a regulated fashion
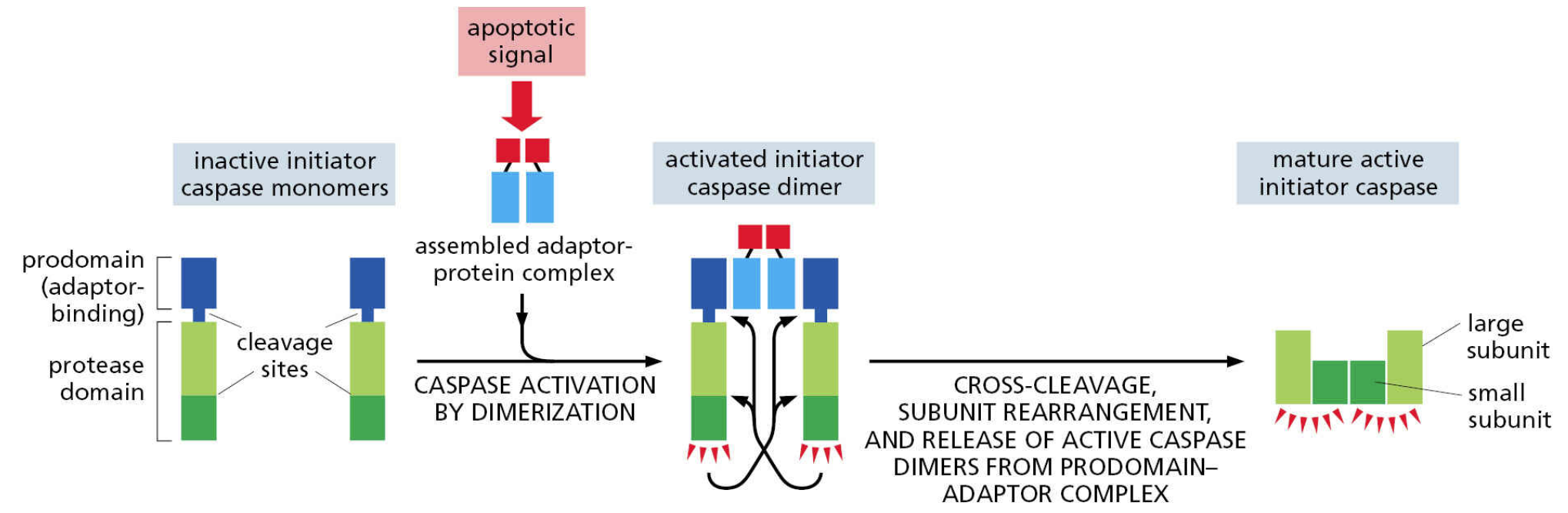
apoptosis is regulated by caspases (“cleave after aspartate”)
Apoptosis has a complex integrative regulatory regime (which you will see), but major events are controlled by caspase proteins
Caspases are cysteine proteases that are activated by proteolytic cleavage
Each caspase has multiple targets that are cleaved at conserved sequences that end with aspartate
Initiator caspases are activated early in the process
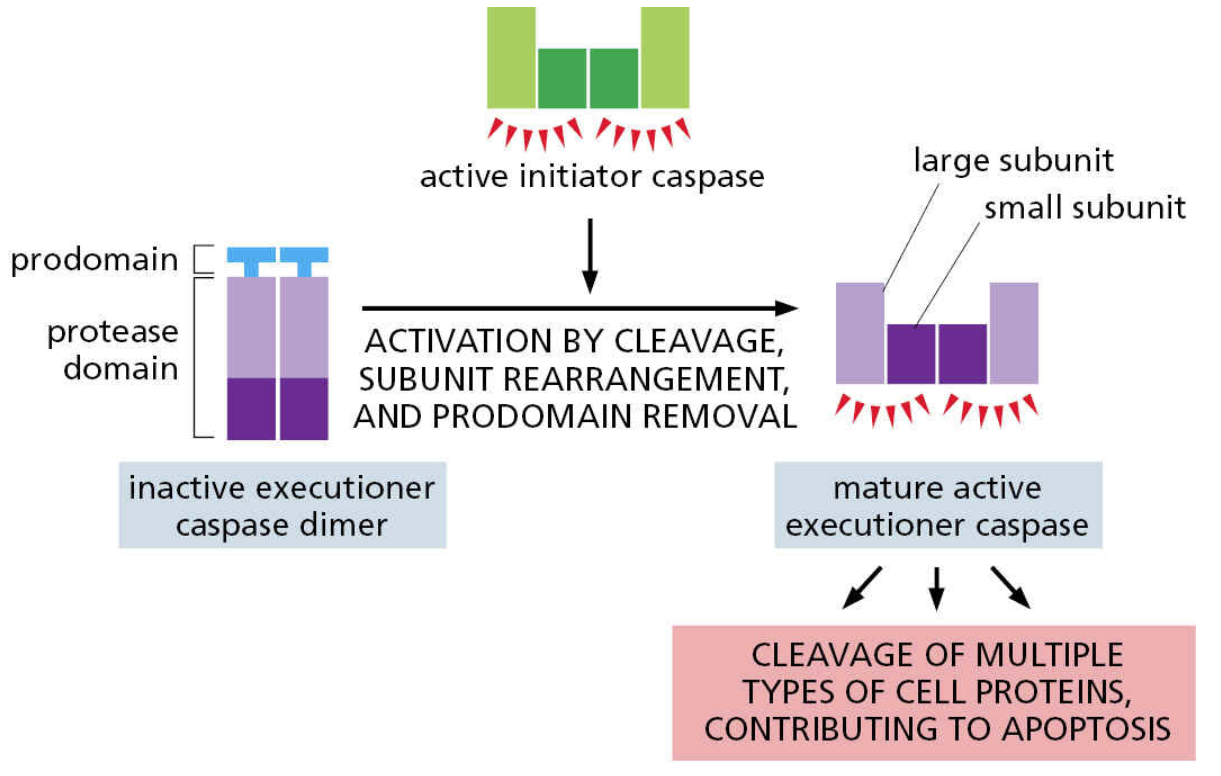
Executioner caspases are activated by initiator caspases and drive apoptosis by cleaving their own targets
E.g. inhibitor of Caspase-Activated DNase (iCAD)
capase-activated DNase (CAD)
different caspases deliver death signals from different parts of the cell
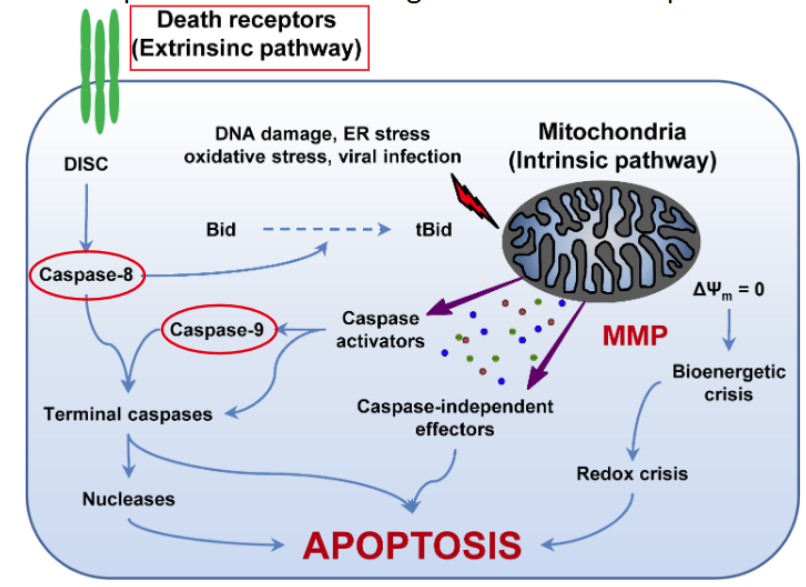
extrinsic and intrinsic pathway
pro-apoptotic signals can come from other cells
The extrinsic apoptotic pathway relies on activation of plasma membrane proteins collectively termed death receptors
The group of proteins recruited to death receptors is collectively referred to as the Death-Inducing Signaling Complex (DISC)
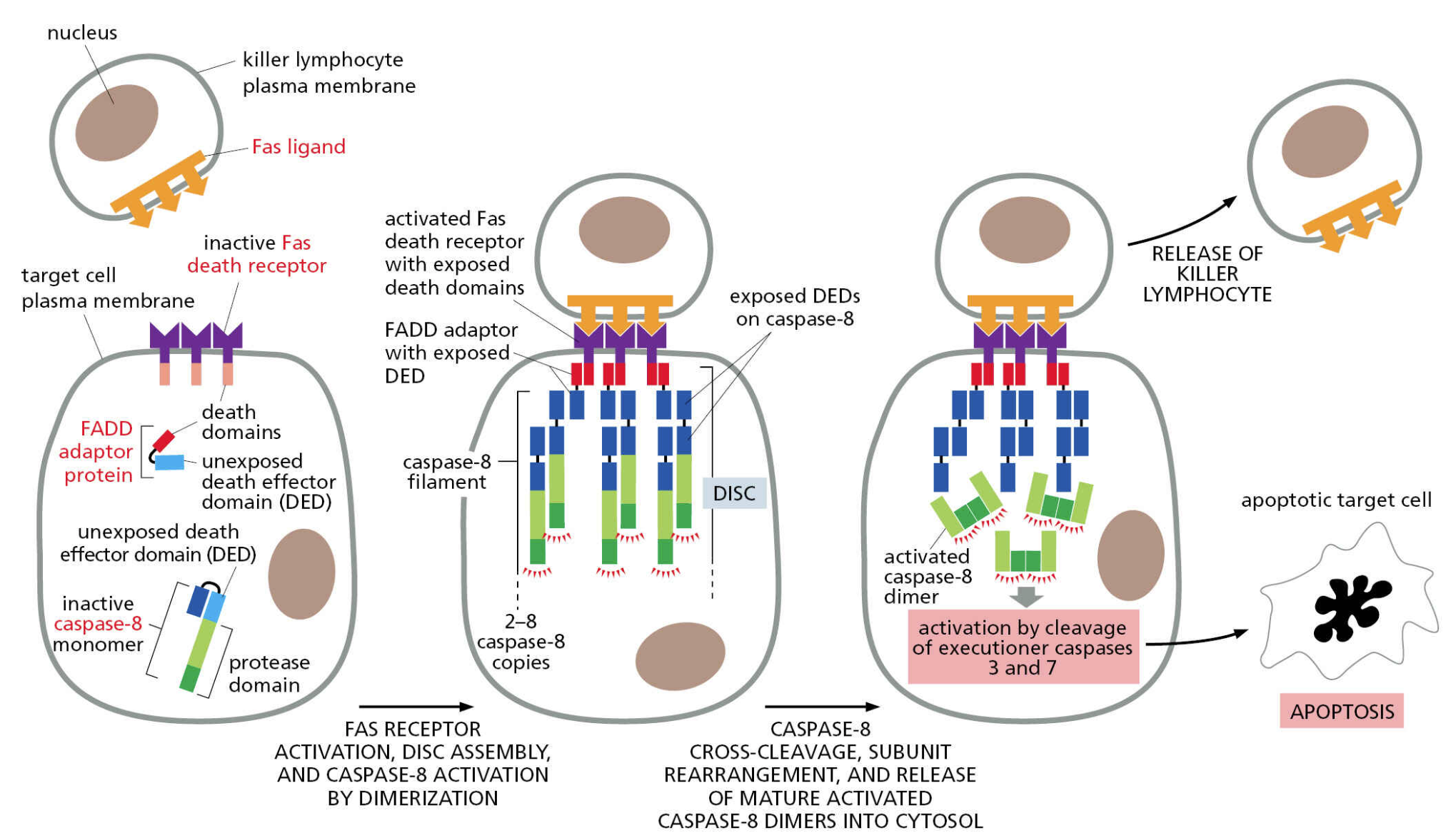
The best characterized death receptor is Fas. Cytotoxic T cells kill virally infected cells, in part, by signaling through FasL — contact-dependent pathway
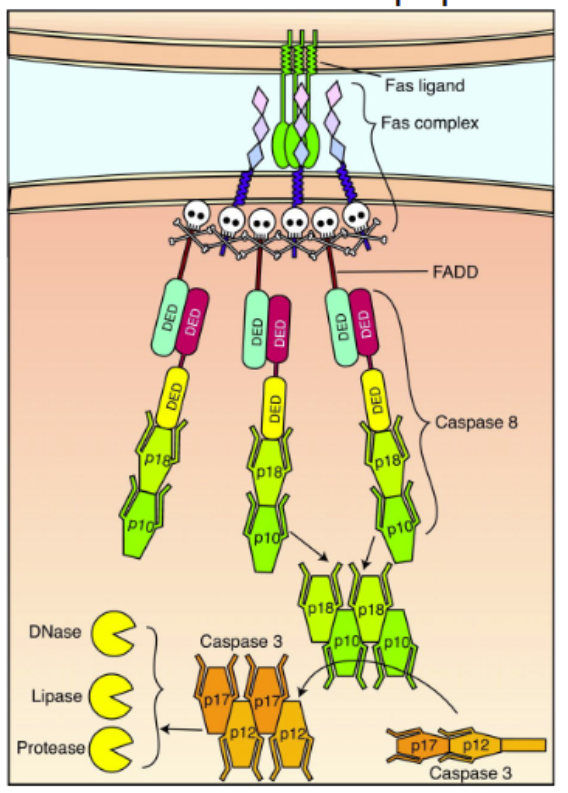
immune cell stimulation of apoptosis
natural killer cells and cytotoxic (CD8) T cells can kill abnormal or sick cells with Fas ligand
activation of Fas creates a Death-Inducing Signaling Complex (DISC) consisting of procaspase-8 and Fas-Associated Death Domain (FADD)
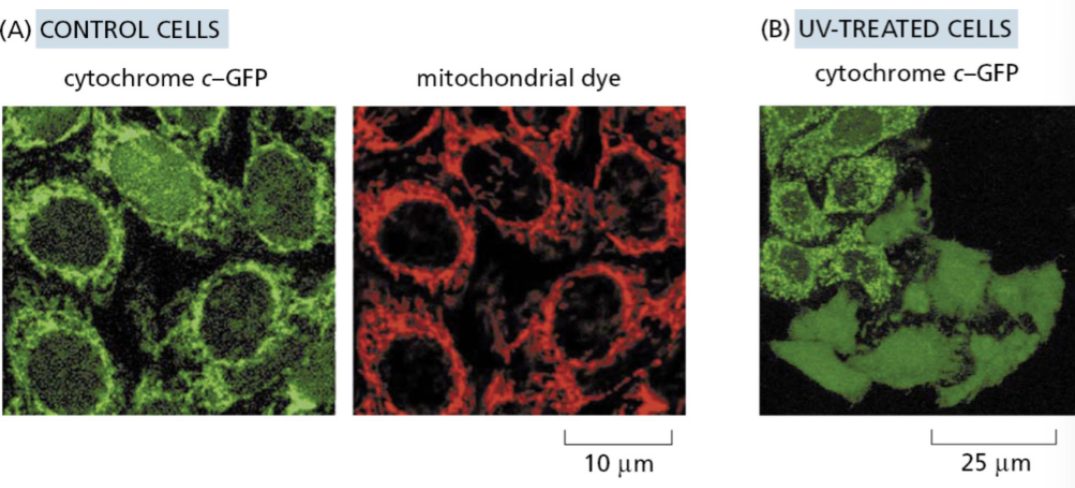
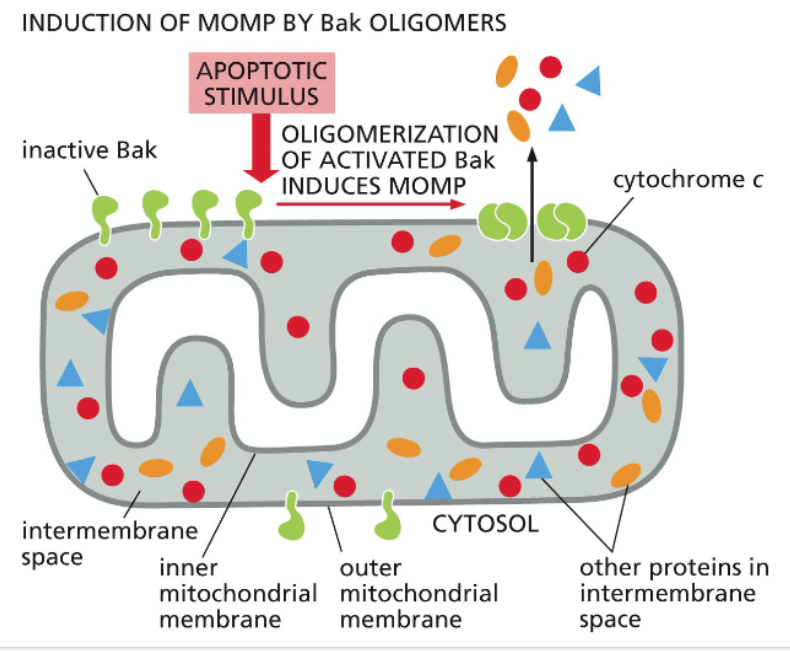
the intrinsic pathway requires release of cytochrome c from mitochondria
Cytochrome c (part of the electron transport chain) assembles with other proteins to form a complex called the apoptosome which activates initiator caspase-9
Cytochrome c and dATP (deoxy-ATP) bind and activate Apaf1, which assembles via its CARD domain into a wheel-like structure
Caspase-9 is recruited via its own CARD domain and activated
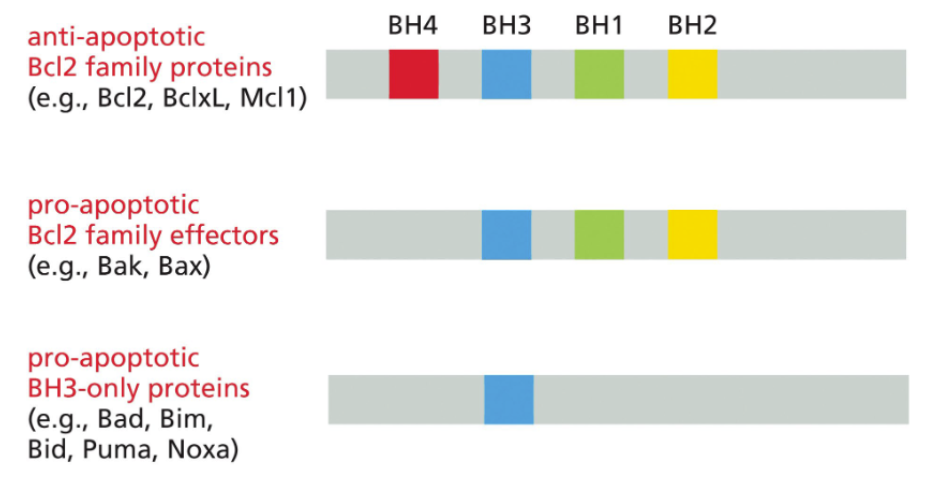
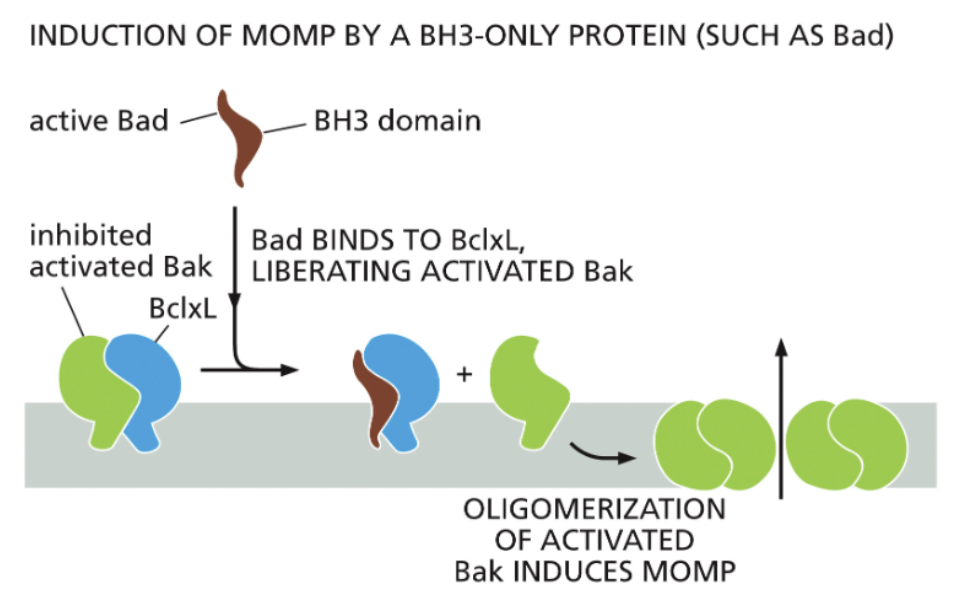
interaction domains of anti- and pro- apoptotic proteins
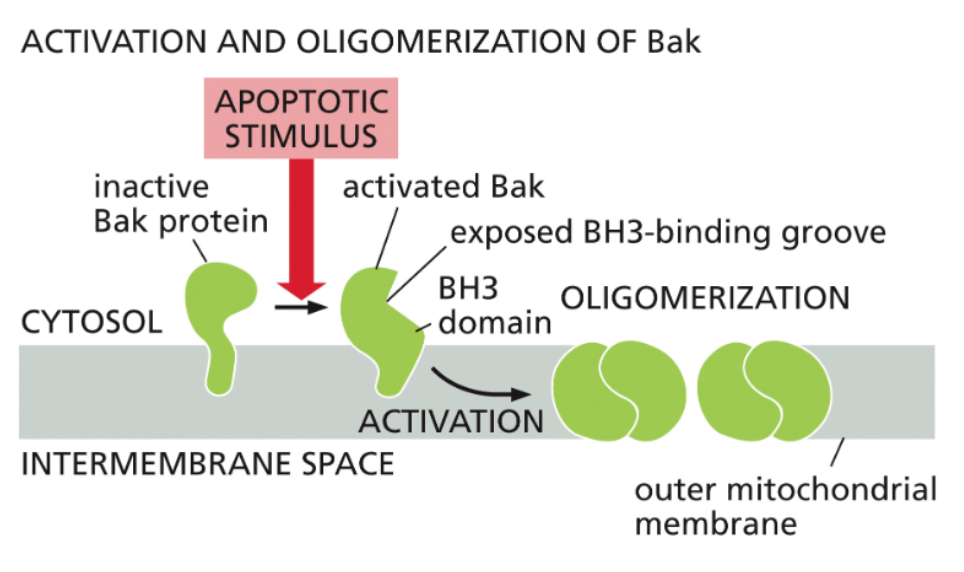
Bak oligomers promote Mitochondrial Outer Membrane Permeabilization (MOMP)
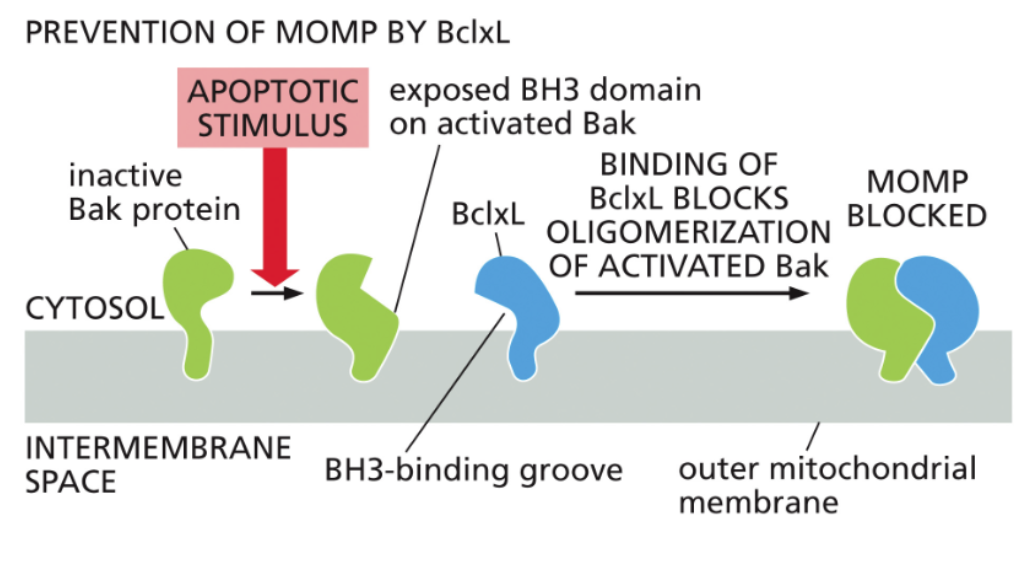
Bcl2 family proteins inhibit Bak oligomerization
BH3-only proteins competitively inhibit Bcl2 proteins
BH3 -| Bcl2
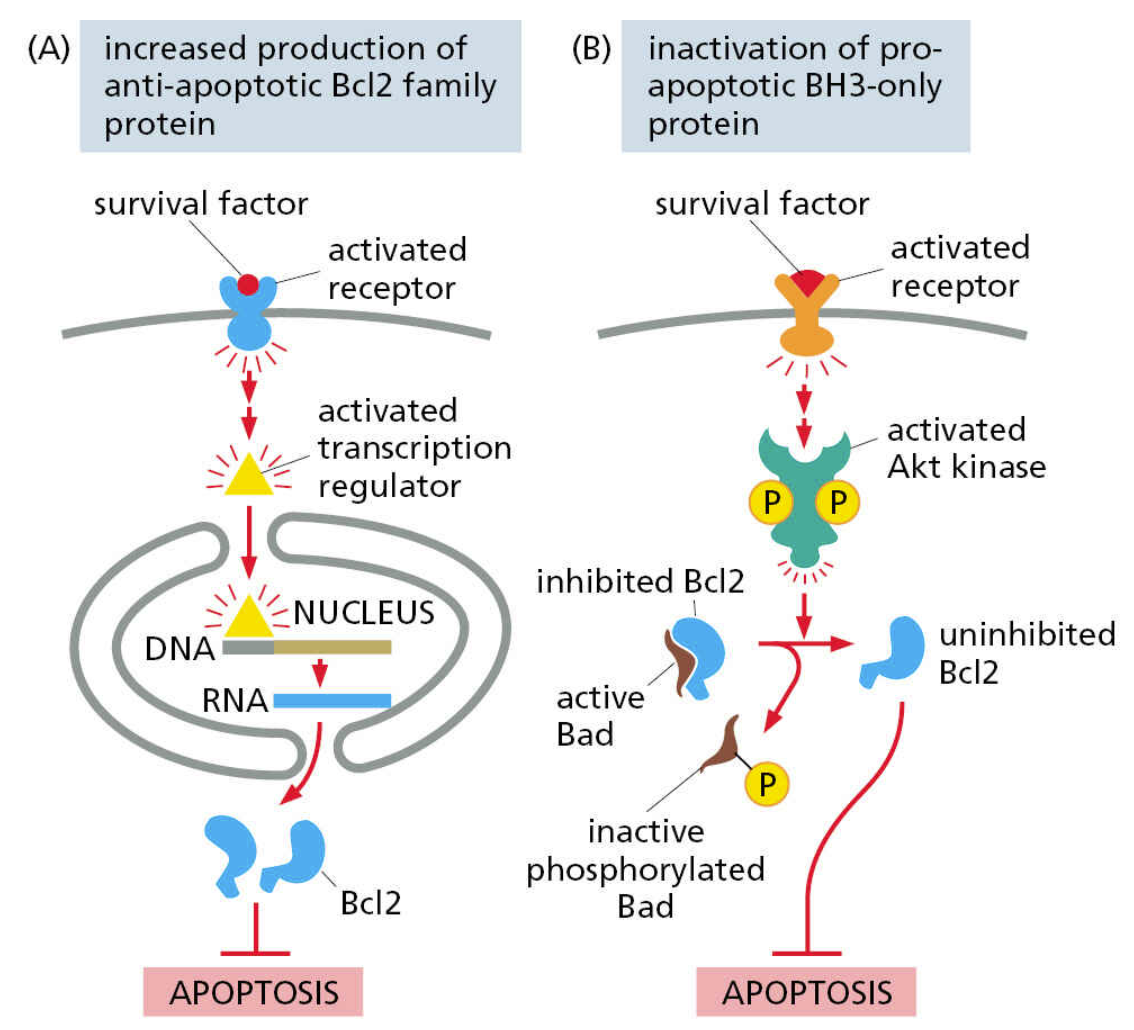
pro-survival signaling acts through inhibitors of apoptosis
Normal animal cells require regular pro-survival signaling
these pathways suppress apoptosis by stimulating the production and/or activation of apoptosis inhibitors
Most promoters of apoptosis are always present – only continuous suppression keeps the cell alive
this makes apoptosis a nimble process – a cell that is damaged or in danger of becoming a cancer can readily be induced to self-destruct
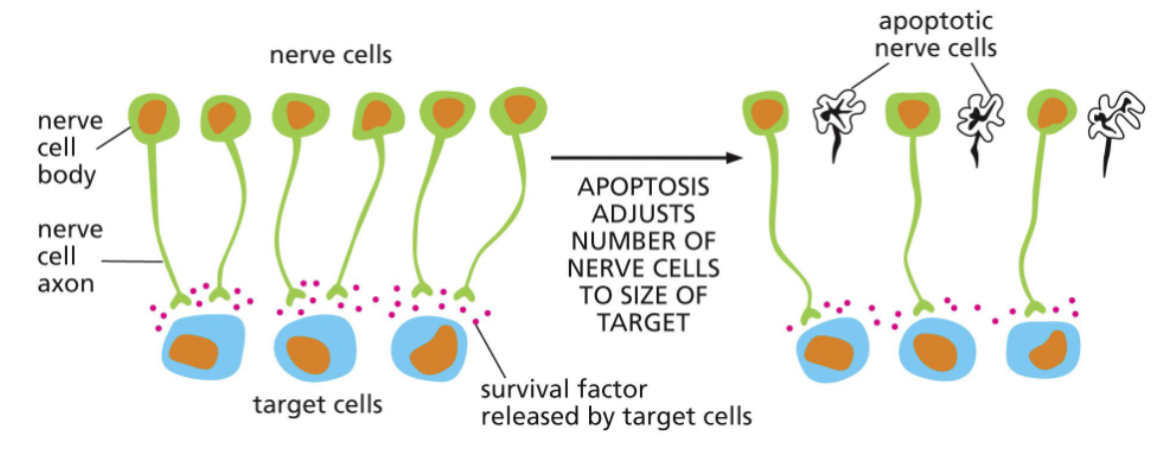
limited survival factors prune developing nervous systems
Many infant animals (including humans) are born with more neurons in their brains than adults
The target cells produce a finite amount of pro-survival factors
As the nervous system develops postnatally, neurons die and, ultimately, the number of neurons and target cells are balanced
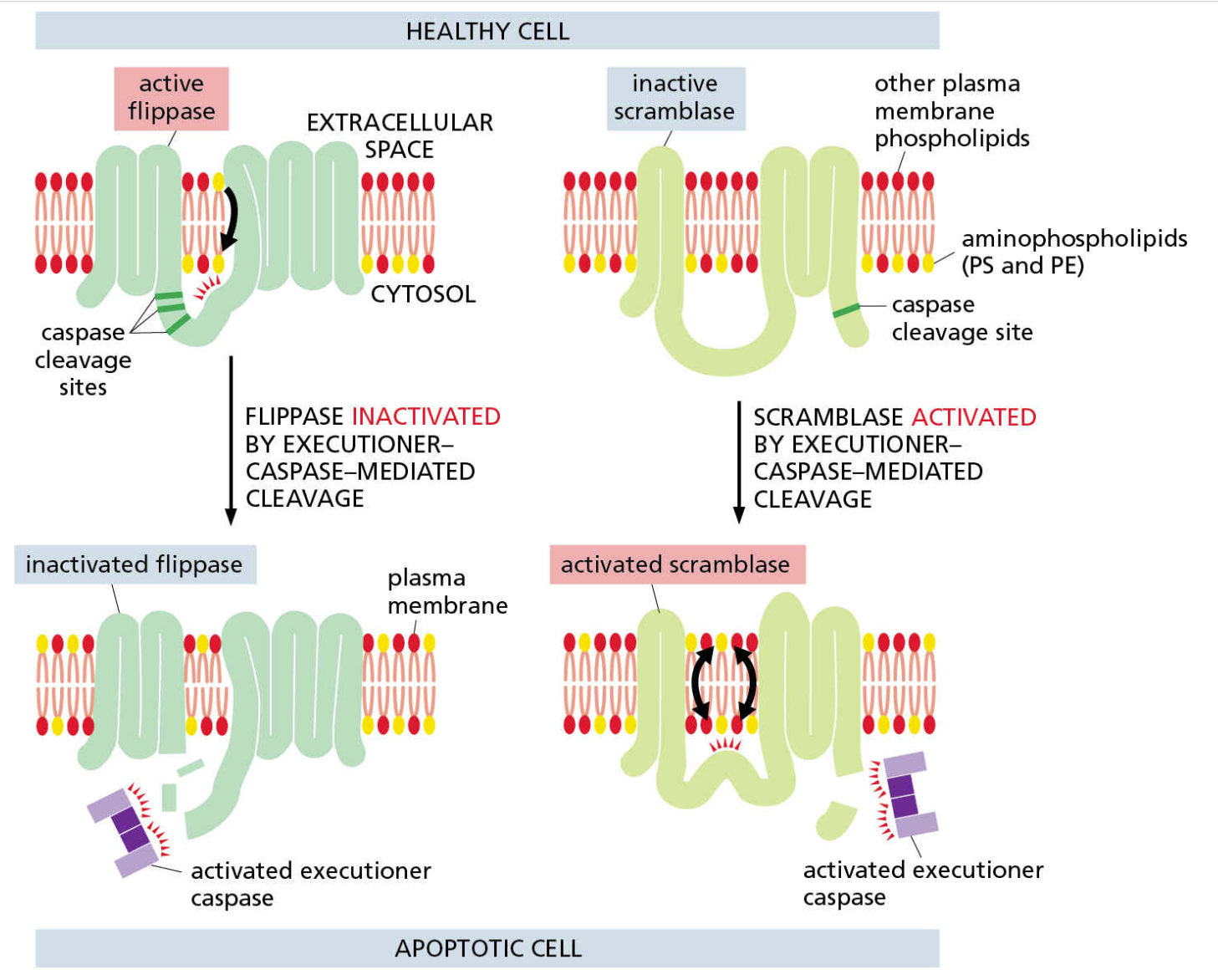
Inactivation of flippase and activation of scramblase relocalizes membrane lipids
In healthy cells, most of phosphatidylserine and phosphatidylethanolamine is on the cytosolic leaflet of the plasma membrane
Executioner caspases cleave both flippases and scramblases on the plasma membrane
This inactivates flippases, but activates scramblases
Altered flippase and scramblase activity allows cytosolic phospholipids to appear in greater numbers on the extracellular leaflet
Extracellular phosphatidylserine is an “eat me” signal that stimulates phagocytosis of apoptotic cells
Summary / Key Concepts
Apoptosis is a regulated form of cell death
in contrast to necrosis, which is simply disruption of the plasma membrane, apoptosis gradually breaks down cell contents and packages them into vesicles for consumption by phagocytes
Caspases are major drivers of apoptosis
initiator caspases are activated by different cellular events
executioner caspases activate mediators (like Caspase-Activated DNase) through proteolytic cleavage
The extrinsic pathway is activated by death-inducing ligands (like FasL)
death receptors signal through caspase-8
Pro-apoptotic factors include members of the Bak and BH3-only family
Bcl2 family members are anti-apoptotic
The intrinsic pathway of apoptosis relies on release of cytochrome c from the mitochondria via Bax and Bak;
these proteins are inhibited by Bcl2
Cytochrome c forms a complex with other proteins that activates initiator caspase-9
pro-survival signaling suppresses apoptosis
limitations in survival factors during development prune cells from forming tissues
Alterations in flippase and scramblase activity via Caspase cleavage allows formerly cytosolic phospholipids to appear on the outside of cells