Chem 331 Exam 2
Confirmations of Acyclic Compounds
different arrangements of atoms in 3D space resulting from rotation around a single bond
double bonds cannot rotate because a pi bond locks carbon in place
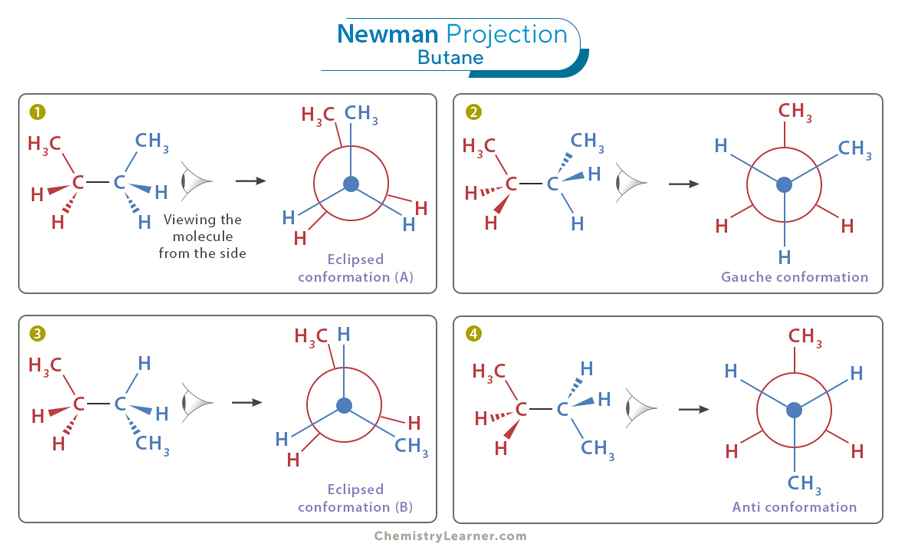
Newman projection- front carbon is represented by a dot while back carbon is represented by a circle
solid line is axial, wedge is equatorial up or equatorial down depending on what confirmation you’re looking at
staggered confirmation- 60 degree dihedral angle
eclipsed conformation- least stable, subs at the same angle to each other
torsional strain- caused by electron repulsion between eclipsing groups
gouche- 60 degree angle between CH3
Confirmation of Cyclohexane
atoms adapt a staggered shape that has no angular strain or torsional strain
every Carbon has a Hydrogen that either points straight up or down (axial) and closer to the flat up and down (equatorial)
cyclohexane changes via a ring flip, it does nor stay in a single chair confirmation. In a ring flip, up Carbons become down carbons and down carbons become up carbons. during a ring flip, any subs that are axial become equatorial and any subs that are equatorial become axial.
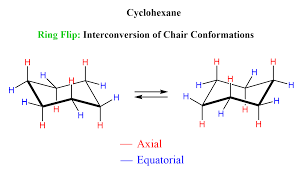
axial substituents (in methyl-cyclohexane) suffer from unfavorable 1,3 diaxial onteracrtions.
trans- opposite sides of the ring
trans can be trans axial (axial up and axial down), trans equatorial (equatorial up and equatorial down) or an equatorial and a axial.
cis- same side of the ring
most stable when the largest sub is at the equatorial position
Chapter 4 Isomers (Stereochemistry)
isomer- different compound with the same chemical formula
constitutional isomer- different atomic connectivity
stereoisomers- different 3D arrangement of atoms
Chirality and Asymmetric Centers
asymmetric center (aka chiral center)- atom bonded to four different groups, can be a group of atoms or a single atom, or a lone pair. each group has to be different. double bonds count as two of the same group.
a molecule with a chiral center is always said to be chiral.
RULE 1- a molecule is said to be chiral if the molecule and its mirror image are non-superimposable
RULE 2- if a molecule possesses a plane of symmetry, the molecule is superimposable on its mirror image and is achiral
enantiomers- non-superimposable mirror image. “same guy”
fisher projection- another way to represent molecules in their 3D arrangement where horizontal bonds represent bonds that point out and vertical bonds represent ones that point back
Ph=phenyl= benzene ring
okay to rotate a fischer projection 180 degrees. rotating 90 changes the stereochemistry
R/S configuration of a Chiral Center
Identify the atoms that are connected to the chiral center
prioritize atomic number (1 has highest priority-biggest atom; 2 has lowest priority-smallest atom)
draw curved arrow from 1-2 and 2-3
S Configuration- Counter clockwise
R Configuration- clockwise