nitration of benzene
nitrobenzene is used to make:
explosives
pesticides
dyes
pharmaceuticals
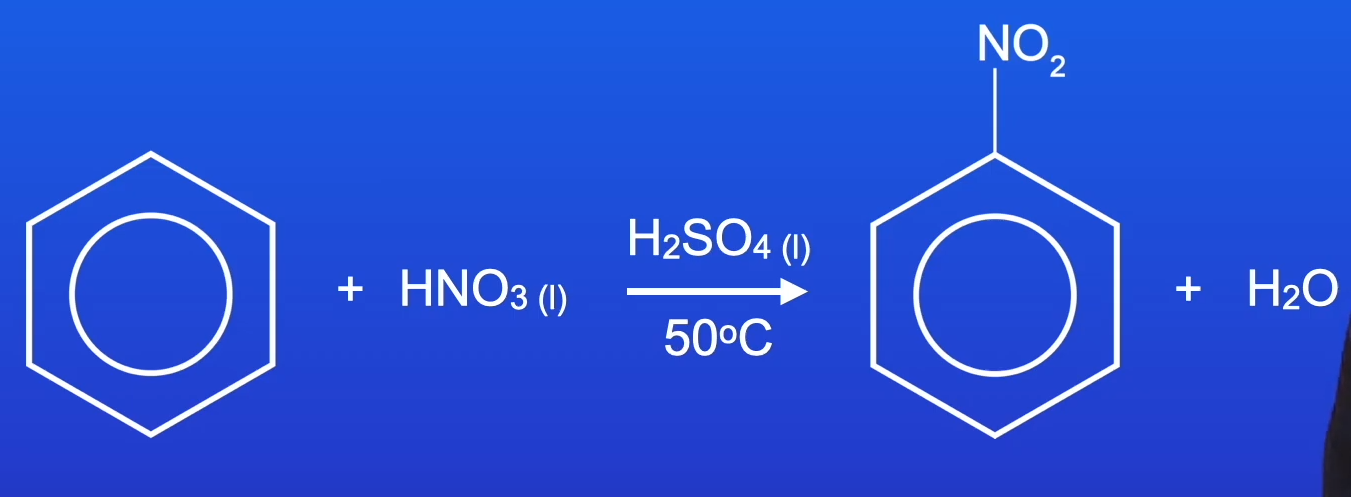
stage 1 : formation of the electrophile

the nitronium ion is the electrophile in this equation
stage 2 : electrophilic substitution of the nitronium ion into the benzene molecule
stage 3: regeneration of the catalyst

using higher temperatures increases the chance of a second substitution - forming the product 1,3-dinitrobenzene → so its important to keep temperature at 50^C to reduce this chance