1.2 Chapter 10: Properties of Gases
10.2: Measurement of Pressure
Pressure is force per unit area: Pressure = force / area
The molecules in the air collide with everything, which causes atmospheric pressure
For a barometer, if the atmospheric pressure drops, the mercury level in the tube will also drop, indicating a decrease in pressure.
If the atmospheric pressure increases, the mercury level in the barometer rises, reflecting a higher pressure exerted by the air molecules.
The standard atmosphere is defined as the pressure exerted by a column of mercury 760 mm high at a temperature of 0 degrees Celsius, which is equivalent to 101.325 kPa.
The torr is the best representation of it, due to 1 torr = 1 mm of mercury.
To measure pressure inside a container using gas as a product or reactant, a manometer can be used, which compares the pressure of the gas to the atmospheric pressure, allowing for accurate readings in various applications.
An open-end manometer is used to measure the pressure of a gas in relation to atmospheric pressure by having one end open to the atmosphere and the other connected to the gas container, enabling direct comparison.
A closed-end manometer is used when the gas pressure must be measured without direct exposure to the atmosphere, as it contains a sealed column of liquid that reacts to the pressure of the gas inside the container, providing a precise measurement.
In both cases, the height difference in the liquid column indicates the pressure of the gas, which can then be converted into appropriate units for analysis.
typically, mercury is used in the process as the liquid, but other liquids like water will provide a more precise measurement. So, to convert to mercury distance,
mm Hg = mm liquid x density of liquid / density of Hg
OHS: 1.2.3:
V = an is an equation representing how volume (V) changes with the number of moles of gas (n), where a is a constant.
Volume increases linearly with additional moles of gas
Avogadro’s law: At a given temperature and pressure, the volume of a gas is directly proportional to the amount of gas, or V = k × n.
I guess the equation your supposed to use is
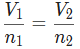
As heat is added to a gas, its volume will also increase if the pressure remains constant, following Charles's Law which states that the volume of a gas is directly proportional to its temperature in Kelvin.
This relationship can be expressed mathematically as V1/T1 = V2/T2, where V represents volume and T represents temperature, thus expressing a linear relationship.
1000 L = 1 m³
An decrease in volume will cause an increase in pressure, if temperature remains constant.
Boyles’s law states that at a constant temperature, volume of a fixed amount of gas is inversely proportional to the pressure of the gas, which can be formulated as P1V1 = P2V2, where P represents pressure and V represents volume
Between Avogadro’s law,. Charles’s law, and Boyle’s law, they can be combined into one equation called the ideal gas law: PV = nRT.
R is the combined proportionality constant called the universal gas constant (8.314 J/K x mol if using pascals for pressure and m³ for volume, or 0.0821 L x atm / K x mol if using atm for pressure and L for volume).


The volume of one mole of gas at 1 atm and 0 C is 22.4 liters, which is known as the molar volume of an ideal gas under standard conditions.
Dalton’s law of partial pressure states that the total pressure exerted by a mixture of non-reacting gases is equal to the sum of the partial pressures of each individual gas in the mixture.
This law can coincide with the ideal gas equation:
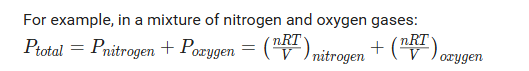
To find the partial pressure, you find the fraction of the gas compared to the whole
To explain why gases behave according to those laws, the Kinetic molecular theory is employed.
According to this theory, the relationship between the temp of a gas and the velocity of the particles in that sample can be represented as
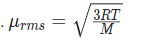
The first part is the root mean square velocity for a gas represents the square root of the average of the squares of the particle velocities.
The average velocity of a singe gas particle is equal to the square root of three times the gas constant times the absolute temperature, divided by the molar mass of the gas.
Smaller molar mass = quicker effusion
This only applied to ideal gases, real gases may deviate from this behavior due to intermolecular forces and the volume of real gas molecules
gases behave most similarly to ideal gases when high temp and low pressure, because it best allows them to overcome intermolecular forces by moving fast (temp) and maximizing the distance between particles (pressure).
1.2.5 Gas Behavior
Any time a carbon-based molecule is “burned“, it reacting with oxygen, it is a combustion reaction.
work done can be defined as
W = -P (delta) V
W = work; P = pressure; ( \Delta V ) = change in volume of the gas. In this equation, work is done on the system when the volume decreases, as indicated by a negative change in volume, while work is done by the system when the volume increases.
or work is done by the system if W is positive and work is done to the system if W is negative