PARTICLE MODEL OF MATTER
Density
Density is how mass a substance has per unit of it’s volume
Density(kg/m3) = mass/volume — p=m/v
Calculating density of liquids - record the mass on a balance, measure the volume in a cylinder and calculate
Calculating density of solids - first measure the mass of a balance
For regular solids, measure the length, height and width and calculate
For irregular, fill a eureka can with water and place the solid in it - the volume of water that is displaced = volume
Generally, the larger the volume = a more accurate density, as it minimises the effects of uncertainty
Can also repeat and use a mean to reduce uncertainty
Specific heat capacity
Specific heat capacity is the amount of energy required to raise one kilogram of a material by one degrees Celsius - in J/kg degrees c
Specific heat capacity depends on the mass, substance of material and the amount of energy put into the system
Solids have a lower specific heat capacity than liquids
The particles are closer together and more likely to hit each other and pass the energy around
Change in thermal energy = mass x specific heat capacity x temperature
ΔEt (J)= m (kg)x c (J/kg°)x Δtemp (°c)
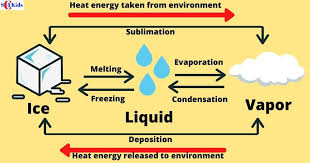
Specific latent heat
Specific latent heat is the amount of energy required to change the state of 1kg of a material without changing it’s temperature
Changing the internal energy of a material causes either a temperature of STATE change - specific latent heat
Every material has 2 specific latent heat values
Latent heat of fusion - melt/freezing
Latent heat of vaporisation - boil/condensing
For example, the latent heat of fusion for water is 334 kJ/kg, and is 2260 kJ/kg for latent heat of vaporisation
Change in potential energy = mass x specific latent heat
ΔE (J) = m (kg) x l (J/kg)
Horizontal lines on internal energy graph ↓
Particle motion in gases
Particles in a gas move in random directions in a straight line unless another particle hits it (or a wall)
They rebound when they hit a wall, which exerts a force and creates pressure
Pressure in gases
Total pressure depends on the number of collisions and how much energy each collision involves
Depends on temperature, concentration and volume
In a flexible container (like a balloon), the temperature and concentration will increase the volume
In a solid container:
Temperature - if you heat up the gas, it’s energy will increase, so there is more kinetic energy and the particles will move faster
This means there are more collisions, with more force, creating a higher pressure
Concentration - if you increase the number of particles in the same volume, the concentration increase and there are more particles to collide with, which causes more collisions and increases the pressure
Volume - if the volume is decreased, there will be more particles per unit of volume, which causes more collisions and a higher pressure
Increasing pressure of a gas
We can increase the pressure of a gas by:
Increasing the no. of particles
Decreasing the volume
Increasing the temperature
Pressure and volume are inversely related
Pressure increases = volume decreases
Pressure decreases = volume increases
Pressure x volume = constant value (if temperature is constant)
pV = constant
Internal energy
Internal energy is the total amount of kinetic and chemical potential energy of all particles in a system
When a material is heated or cooled, two changes can happen
Chemical bonds between the particles form or break (between states)
Thermal energy store changes as particles speed up or slow down (‘in’ states)
As the temperature increases, particles kinetic energy increases. When the substance melts, the energy is put into breaking bonds, which increases chemical potential energy
Conservation of energy shows that any energy transferred will be distributed between the chemical store and thermal store
When bonds break, the speed of the particles increase to overcome more bonds to further change state
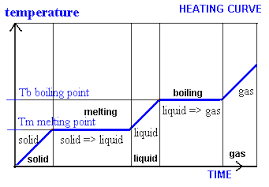
Horizontal represents increasing chemical energy, and increasing temp shows increase in kinetic/thermal energy
Changing states