Chemistry 1 Review
Unit 1:
Unit 9: Chemical Bonding 1
Metallic & Ionic Chemical Bonds
The Lewis dot structures for ionic compounds have charges on each atom and brackets with a complete octet/duet around the anions. Positively charged atoms connect with negatively charged atoms; they NEVER connect to atoms with the same type of charge!
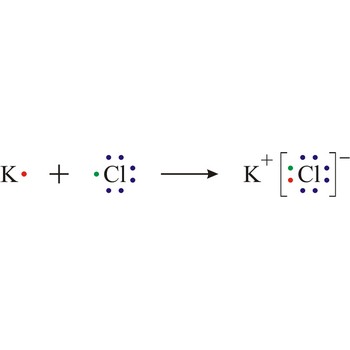
Covalent Bonds
Bond Energy: the amount of energy needed to break 1 mole of a bond. Breaking a bond is endothermic, while making a bond is exothermic.
Electronegativity: the ability of an atom to attract shared electrons; the measure (expressed by a number) of the atom’s attraction for electrons. Increases right-up.
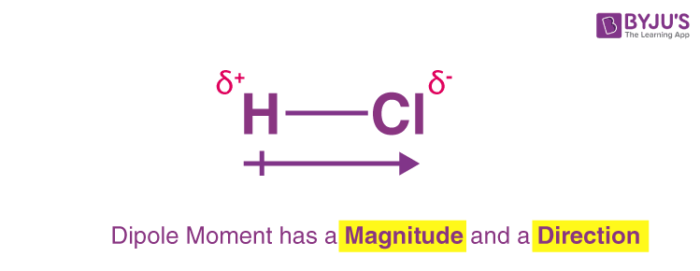
Bond Polarity: how equally the electrons in a bond are shared. The atom with a higher electronegativity will have a partial negative charge (δ-), while the atom with a lower electronegativity will have a partial positive charge (δ+). A dipole, a line with a dash, points to the atom with a higher electronegativity.

Ionic compounds are solid at room temperature, have strong attractions between ions, have high mp, are hard, brittle, and conduct electricity when melted or dissolved. Covalent compounds have lower mp than ionic compounds, are nonconductive, and dissolve as complete molecules in water (they don’t break apart).
Lewis Structures
***The trick for Lewis Structures is to connect the atoms with single bonds and add dots as needed. Then, add more bonds if the number of valence electrons is incorrect.***
General Rules: The central atom of a structure is the atom with the lowest electronegativity. Hydrogen can never be the central atom. Halogens should never have a double or triple bond unless there is no other choice. All polyatomic ions and anions are surrounded by brackets with a charge. In oxoacid structures, hydrogens bond to oxygens. Carbons don’t have dots unless there is only one other atom.
Acids are covalent compounds!
Exceptions to the octet rule: H wants 2e-, B wants 6e-, and N wants 7e- when there is an odd number of electrons. Only atoms at or below the third period in the PTE can have an expanded octet.
Resonance Structures: a set of two or more structures that describe the electron bonding of a single species. A double bond is shared throughout the whole molecule. Each bond has an extra dotted line to show the actual atom's resonance; this dotted line structure is called delocalized bonding.

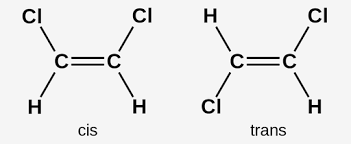
Isomers: molecules that have the same molecular formula but different Lewis Structures. If you have a single bond, you can rotate the outside atoms; if you have double or triple bonds, the bond group is “fixed,” and you cannot rotate the outside atoms, making more possible isomers. Cisisomers (cis- means on this side of) have the same element on the same side, while transisomers (trans- means the other side of) have the same element on the opposite side.
Formal Charge = # ve- - # shared e- /2 - # unshared e-. The shortcut is to count the electrons around the atom starting at # ve- - 1. The best Lewis Dot Structure has the lowest formal charges (as many as possible close to 0); if multiple structures have the same amount of formal charges, the best structure has any negative formal charge on the higher EN atom.
Energy Effects in Chemical Bonding
Lattice Energy (LE): the energy required to separate one mole of solid ionic compound into ions in a gaseous state. Higher charges mean higher Lattice E, which usually means higher mp. On the other hand, Lattice Energy becomes less exothermic with increasing atomic radius.
Bond Dissociation Energy (BE): the energy needed to break one mole of that bond in a molecule in a gaseous state.
ΔHrxn = ΣΔHbonds broken - ΣΔHbonds made. On each chemical equation, all the bonds of the reactant side break while the bonds on the product side are formed. Remember: breaking bonds is endothermic (+), while forming bonds is exothermic (-).
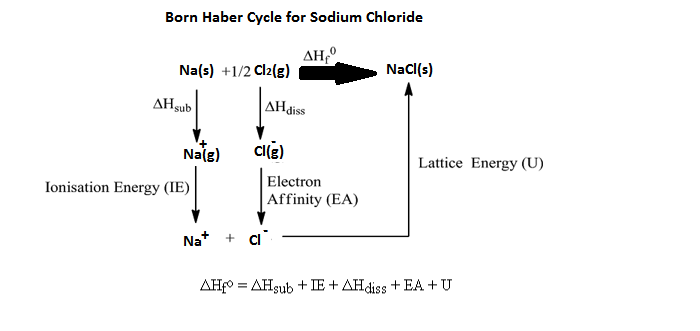
Born-Haber Cycle: the series of steps to find the ΔHrxn. Add each ΔH of each process, including phase changes, LE, BE, IE, and EA.
Unit 10: Chemical Bonding 2
VSEPR
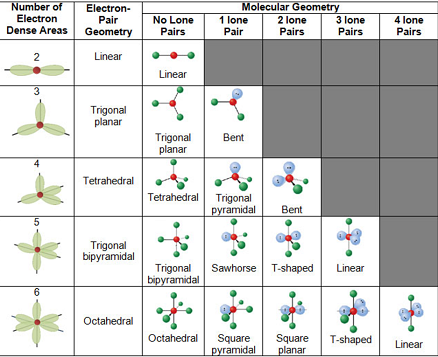
VSEPR stands for Valence Shell Electron Pair Repulsion. This theory determines the most likely 3D geometry of molecules, which are arranged around the central atom in a certain way to minimize repulsive forces.
Electron groups are lone pairs, single bonds, double bonds, and triple bonds (multiple bonds are NOT multiple electron groups).
Molecules with multiple central atoms have different geometries around each central atom.
A polar bond is where electrons are not equally shared (bonds have partial charges and dipoles); a nonpolar bond is where electrons are equally shared. Nonpolar molecules are “symmetrical” throughout or on each “plane;” attractive forces cancel out. Think of this concept as “a tug of war.”
Valence Bond Theory
Valence Bond Theory (VBT): when two atoms bond together, the valence orbitals of both atoms hybridize to form new atomic hybrid orbitals. The hybrid orbitals can be bonding or nonbonding. Only valence electrons interact in this theory; core electrons don’t participate.
Type | Number of Orbitals | Shape |
sp | 2 | Linear |
sp2 | 3 | Trigonal Planar |
sp3 | 4 | Tetrahedral |
sp3d | 5 | Trigonal Bipyramidal |
sp3d2 | 6 | Octahedral |
A sigma bond is an end-to-end orbital overlap (always single bonds), with free rotation around these bonds; a pi bond is a side-to-side overlap of parallel orbitals (always double or triple bonds), with no rotation around these bonds because they are rigid.
Molecular Orbital Theory
Molecular Bond Theory states that new molecular orbitals are created by mixing all the atomic orbitals of the atoms present in the molecule. All electrons of every atom participate in this theory. This theory still follows Hund’s rule and Pauli’s exclusion principle.
Bond Order (BO) = (1/2)(# e- in bonding MOs - # e- in antibonding MOs). This indicates the strength of the bond. If BO = 0, the species is unstable and doesn’t exist.
Diamagnetic has no unpaired electrons; paramagnetic has at least one upaired electron.