Cell Signaling III - signaling through receptors
Signaling Overview
Signaling through receptors: Involves extracellular signal molecules binding to receptor proteins, which then activate intracellular signaling pathways affecting target proteins that alter metabolism, gene expression, and cell shape/movement.
Types of Receptors
Cell-Surface Receptors: Located on the plasma membrane, and interact with hydrophilic signaling molecules.
Intracellular Receptors: Found within the cell and bind to small hydrophobic signal molecules which often require carrier proteins for cellular transport.
Molecular Switches in Signaling
Signaling pathways often involve molecular switches to activate proteins:
Phosphorylation: Reversible process involving the addition/removal of phosphate groups from ATP. Proteins in inactive state can be activated in this way.
GTP-binding Proteins: Similar to phosphorylation but involves GTP. Activation involves GDP-GTP exchange (binding of GTP and release of GDP), altering protein conformations to initiate signaling.
Second Messengers in Signaling
Important in signal amplification.
Types of secondary molecules such as:
3',5'-Cyclic AMP (cAMP)
First 2nd messenger discovered by Sutherland.
Generated from ATP by adenylate cyclase which is activated by Gs (stimulatory G-protein) protein.
Targeted by phosphodiesterase, which degrades cAMP to AMP turning off the signal.
cAMP activates Protein Kinase A (PKA), leading to phosphorylation of target proteins.
3',5'-Cyclic GMP (cGMP): Generated from GTP; also degraded by phosphodiesterase; activates PKG; involved in nitric oxide signally pathway.
Inositol 1,4,5-trisphosphate (IP3) and Diacylglycerol (DAG): Produced from phospholipase C-β (PLC) activity on phosphatidylinositol 4,5-bisphosphate (PIP2).
(aq subunit of G-protein activates PLCβ)
IP3 receptor is a ligand-gated channel in the membrane of ER. Binding of IP3 opens the channel and releases Ca2+ from ER. The released Ca2+ ions bind to calmodulin (CaM), activating CaM-dependent protein kinase II (CaMK-II).
Ca2+ induces a conformational change in CaMK-II and subsequent binding to target proteins.
Ca2+ signals are terminated (degraded) by Ca2+ pumps and exchangers.
DAG activates PKC in the presence of calcium released by IP3.
G-Protein-Coupled Receptors (GPCRs)
Large family of receptors (~800).
Activation of GPCRs involves the exchange of GDP for GTP on G-proteins.
Structure: Seven transmembrane domains; activated by various ligands including hormones (adrenaline) and neurotransmitters (glutamate).
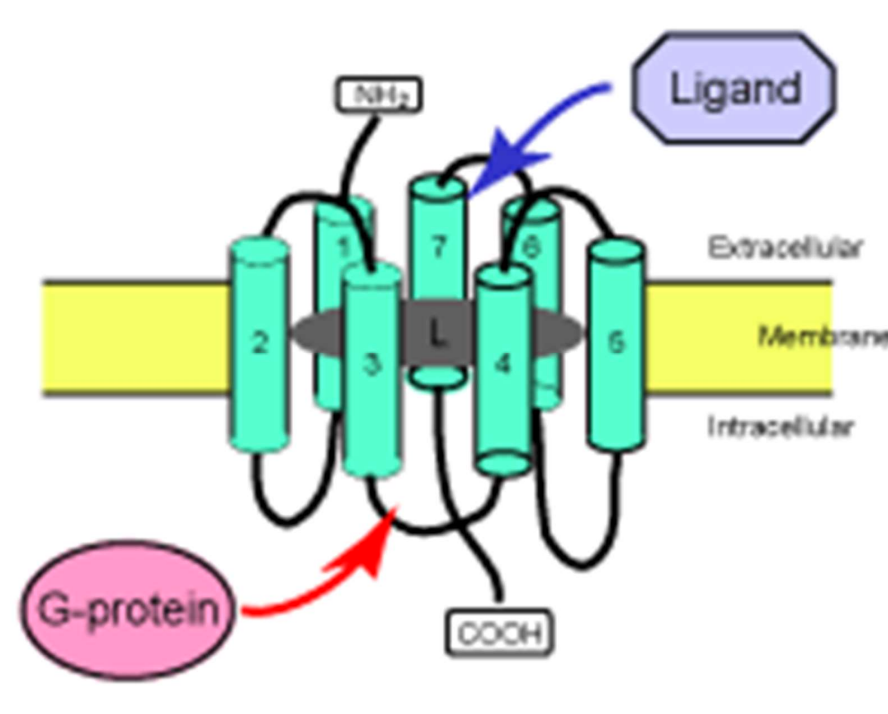
Function: GPCRs are coupled to heterotrimeric G-proteins, facilitating downstream signaling once activated.
GPCR Activation Cycle
Ligand binding causes a conformational change in the receptor.
This activates associated G-proteins by exchanging GDP for GTP.
Activated G-protein subunits relay the signal to effectors like adenylyl cyclase or phospholipase C.
Subunits alpha, beta/gamma each signals for a different sets of responses.
cAMP Signaling Pathway
Generated by adenylyl cyclase from ATP.
Regulated by:
G-protein α subunits:
αs increases cAMP levels.
αi decreases cAMP levels.
cAMP activates protein kinase A (PKA) by freeing (dissociating) the catalytic (C) subunit from regulatory (R) subunit. The catalytic subunit (C) is what phosphorylates target proteins.
Degradation of cAMP is performed by phosphodiesterases.
Example: Adrenaline activates β-adrenergic receptor (a GPCR) and the cAMP pathway in the heart.
Calcium Signaling
Calmodulin Activation: Ca2+ binds to calmodulin, leading to changes in protein conformation that activates downstream targets such as calcium-dependent protein kinases.
Termination: Ca2+ levels are regulated by pumps and exchangers; actions cease when levels normalize.
Receptor Tyrosine Kinases (RTKs) - a smart protein
Different structure from GPCRs, typically one or two transmembrane regions.
Often activated by growth factors like insulin, epidermal growth factor, fibroblast growth factor.
RTKs dimerize upon ligand binding, activating their intrinsic kinase activity (autophosphorylation).
Autophosphorylation leads to the recruitment of adaptor proteins (like Grb-2) and activation of downstream proteins e.g. Ras, a monomeric G-protein, instigating a MAPK signaling cascade. This is known as a transducer event.
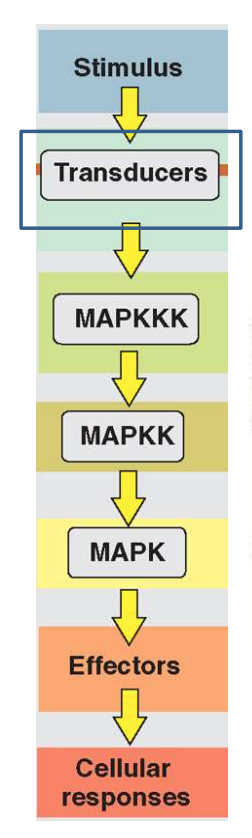
Mitogen Activated Protein Kinase (MAPK) Pathway
Initiates following activation of RTKs.
Involves a cascade of phosphorylation events starting with MAPK kinase kinase (MAP3K), then MAPK kinase (MAP2K), and finally MAPK itself.
MAPKKK e.g. Raf
MAPKK e.g. MEK
MAPK e.g. ERK p38 and JNK, which are involved in various cellular responses including proliferation, differentiation (particularly mesoderm induction during early development), and stress responses.