Chapter 2: Chemistry and Measurements
Units of Measurement
Metric System
Scientists primarily use the metric system, with modifications for specific applications.
International System of Units (SI): A globally accepted system that standardizes measurements for length, volume, mass, temperature, and time.
Volume
Volume measures the space occupied by a substance.
SI Unit: cubic meters (m³)
Common units for chemists: liters (L) and milliliters (mL).
Tools for Measurement: Graduated cylinders are typically used for measuring small volumes.
Length
Length is measured in:
SI Unit: meters (m)
Common metric unit: centimeters (cm).
Mass
Mass reflects the quantity of material in an object.
Measured using an electronic balance.
SI Unit: kilograms (kg)
Commonly measured by chemists in grams (g).
Temperature
Indicates how hot or cold an object is.
Measured in degrees Celsius (°C) and Kelvin (K).
Key Temperatures:
Water freezes at 0 °C (32 °F)
Water boils at 100 °C (212 °F)
The Kelvin scale begins at absolute zero (0 K).
Time
Measured in seconds (s), consistent in both metric and SI systems.
Tools used: Stopwatches.
Measured Numbers
A measuring tool produces measured numbers, quantifying dimensions like length and mass.
Reporting Measurements
Example: Length: 4.5 cm
Estimation is crucial; report values reflecting the distance between measurement marks.
Significant Figures
Significant Figures (SFs): Include all known digits in a measurement plus one estimated digit.
General Rules:
Nonzero digits are always significant.
Zeros between nonzero digits are significant.
Leading zeros are not significant.
Trailing zeros in a decimal are significant (e.g., 40.00 has 4 significant figures).
In numbers without a decimal point, trailing zeros are not significant (e.g., 500 has 1 SF).
Exact Numbers
Exact numbers are defined quantities that have no uncertainty:
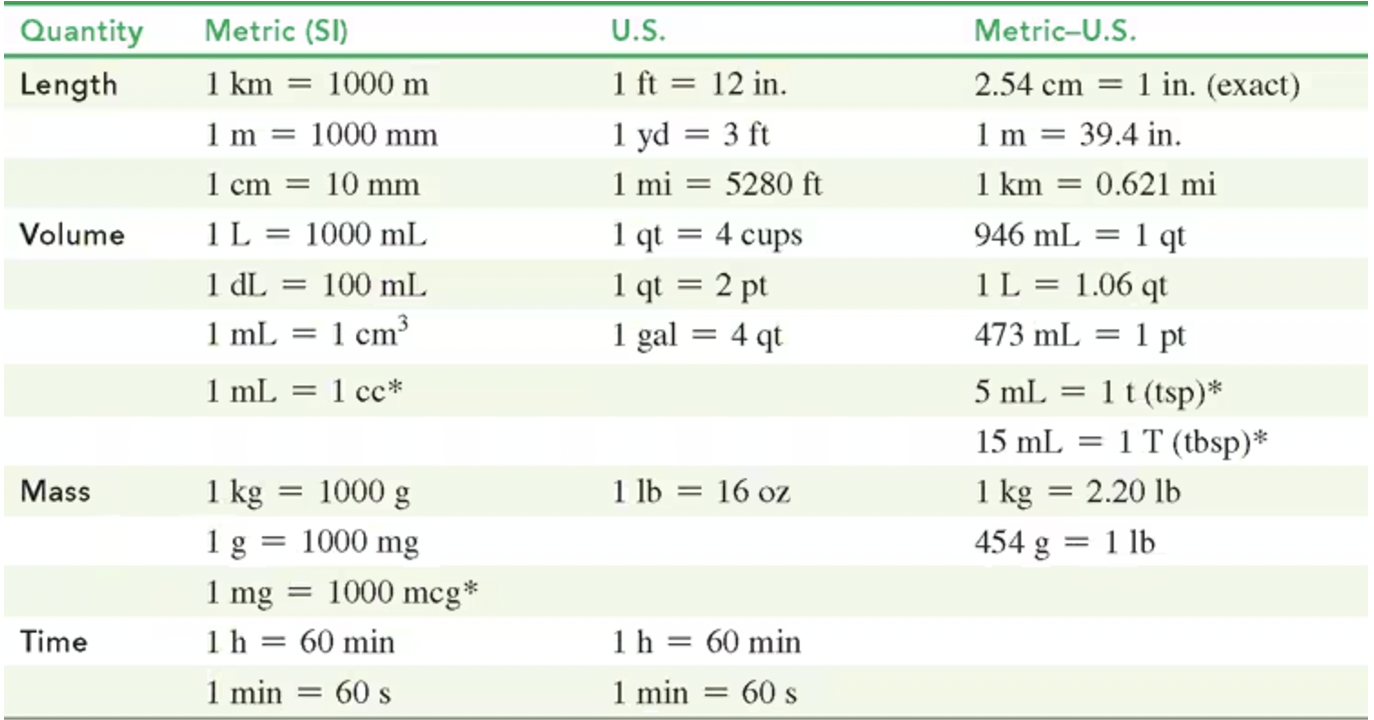
Example: Counting items (e.g., 8 cookies) or definitions (e.g., 1 ft = 12 in).
Rounding Off
In calculations, answers should typically be rounded off.
Rounding Rules:
Dropping digits less than 5 keeps the last retained digit the same.
Dropping digits 5 or more increases the last retained digit by 1.
Operations with Significant Figures
Multiplication/Division
Result should have the same number of significant figures as the measurement with the fewest significant figures.
Addition/Subtraction
Result should be reported to the same number of decimal places as the measurement with the fewest decimal places.
Metric Prefixes
Metric prefixes modify the size of units by factors of 10:
Example: 1 kilometer (1 km) = 1000 m; 1 millimeter (1 mm) = 0.001 m.
Measuring Length and Volume
Volume of a Cube:
Volume can be calculated as side x side x side.
1 cm³ is equivalent to 1 mL.
Equalities and Conversion Factors
Equalities compare two units to express the same quantity:
Example: 1 lb = 16 oz, 2.20 lb = 1 kg.
Conversion Factors:
Defined relationships between units, utilized in calculations.