WEEK 3.1 -Forensic Toxicology – Comprehensive Study Notes
Definition and Scope of Forensic Toxicology
Hybrid field merging analytical chemistry with fundamental toxicology to answer legal (criminal + civil) questions surrounding intoxication and poisoning.
Focus areas
Identification and quantification of xenobiotics (XBs) in humans, animals, environment.
Assessment of how XBs contributed to impairment, morbidity, or mortality.
Compliance with legislation that safeguards clean air, water, food, medicines, agriculture, industry.
Operates across criminal courts (e.g.
homicide, DUI) and civil courts (e.g.
workplace safety, environmental contamination).
Core Competencies Required
Strong analytical–instrumental skillset (GC-MS, LC-MS, ELISA, IR, TLC, etc.).
Working knowledge of pharmacokinetics (ADME) and pharmacodynamics (MOA, dose–response).
Case-specific insight: history, scene information, victim profile, timing of sampling.
Competence in specimen selection, extraction, data interpretation, and courtroom communication.
Fundamental Concepts
Toxicology
“Science concerned with the nature, effects, and detection of poisons.”
In practice: measurement/analysis of toxins, drugs, intoxicants, banned substances, and prescription meds in the body.
Xenobiotic (XB)
Adjective: foreign chemical not naturally produced by the organism/ecosystem.
Noun: the foreign substance itself.
Categories / examples
Plant constituents (alkaloids, glycosides)
Therapeutic drugs
Pesticides / industrial chemicals / environmental pollutants
Cosmetics, flavourings, fragrances, food additives
Can also denote endogenous compounds present at pathologically high concentrations.
Chemical Universe Magnitude
American Chemical Society: ≈ registered compounds.
EU estimate: chemicals in regular use (Greim & Snyder, 2008).
Implication: daunting analytical selectivity challenge for toxicologists (Dinis-Oliveira et al., 2010).
Paracelsus’ Dictum (1493-1541)
“All substances are poisons; none is not a poison. The right dose differentiates a poison from a remedy.”
Modern corollary: dose–response underpins risk assessment; therapeutic index critical in legal/medical interpretation.
Definitions of Poisoning / Intoxication (Forensic Context)
Condition (medical, social, legal) arising from exposure to an excessive dose of an XB relative to the individual.
Medical necessity caveat: clinicians may exceed ‘maximum safe dose’ if therapeutic benefit outweighs risk (Uges, 2001).
Cultural-legal overlay: social acceptability and legality vary across jurisdictions (e.g.
euthanasia, chemical abortion).
Typical Investigative Questions
Has poisoning occurred?
Identity and concentration of the poison?
Route and timing of administration?
Expected physiological / behavioural effects?
Was the amount dangerous or lethal?
Role of possible endogenous substances (e.g.
ethanol from decomposition)?
Poisoning Categories
Accidental: workplace exposure, dosing error, environmental spill.
Experimental: self-medication, recreational drug use.
Intentional:
Self-administration (suicide, para-suicide).
Factitious disorders (Münchausen’s syndrome/proxy).
Assisted/requested (euthanasia).
Criminal (homicide, assault, surreptitious drugging).
Testing Strategy: Screening → Confirmation → Interpretation
Screening (presumptive)
Rapid, broad, cost-effective; higher false-positive risk; generally qualitative.
Essential when structural analogues cross-react (immunoassays).
Confirmatory
Definitive identification/quantification; higher specificity & legal defensibility.
Utilises orthogonal physical principles (e.g.
chromatography + mass spectrometry).
Interpretation requires contextual data: patient history, sampling matrix, post-mortem redistribution, tolerance, metabolism.
Presumptive Tests (Least → Moderate Discriminatory Power)
Colour/spot reagents: Marquis, Duquenois-Levine, Cobalt thiocyanate, Ferric chloride, Koppanyi, p-DMAB, Froehde, Mecke, Zwikker, etc.
Microcrystalline tests.
UV / IR spectroscopy (quick scan mode).
Microscopy (e.g.
pollen, plant fragments, hair, crystals).Thin Layer Chromatography (TLC).
Immunoassays (ELISA, lateral-flow, fuel-cell breathalysers).
Confirmatory Tests (Most Discriminatory)
Gas Chromatography–Mass Spectrometry (GC-MS) & Headspace GC.
Liquid Chromatography–Mass Spectrometry (LC-MS or LC-MS/MS).
Fourier-Transform Infrared Spectrophotometry (FT-IR) with library matching.
Occasionally high-performance GC-FID or LC-DAD for targeted quantitation.
Subfields of Forensic Toxicology
Post-mortem Toxicology
Analyses fluids/tissues from autopsy to establish cause and mode of death.
Key matrices: peripheral/cardiac blood, vitreous humour, urine, liver, kidney, gastric contents, hair, nails.
Human-Performance Toxicology (Ante-mortem)
Determines whether XBs affected behaviour/competence (e.g.
DUI, aviation, workplace).Traffic safety: >50\% of fatalities involve alcohol or other drugs.
Sports doping control (WADA compliance).
Forensic Xenobiotic Testing (Workplace / Legal Compliance)
Demonstrates past use or abuse; usually urine but also hair, saliva, sweat.
Employed by law enforcement, military, private employers.
Analytical Techniques Overview
Immunoassays (ELISA, lateral flow) – rapid, matrix-adaptable.
GC-MS / Headspace GC-MS – gold standard for volatiles (ethanol), drugs.
LC-MS(/MS) – thermally labile, polar, high-mass analytes.
IR & FT-IR – functional group fingerprint for neat powders.
UV-Vis – quick concentration estimates for chromophores.
TLC – cheap separation, semi-quantitative.
Microscopic analysis – botanical, crystallography.
Critical Factors Affecting Result Validity
Pharmacokinetics
Biological half-life: persistence guides timing of sampling (hair vs blood).
Presence of active/inactive metabolites: sometimes measure metabolite instead (e.g.
morphine from heroin).
Matrix selection
Hair archives months of exposure; vitreous humour relatively post-mortem stable; adipose stores lipophilic toxins.
Test specificity
Presumptive vs confirmatory.
Potential for cross-reactivity/shared metabolites among multiple drugs.
Multi-analyte capability
Panels (e.g.
workplace 5-drug screen) vs targeted single-analyte methods.


Example Workflows
Blood Alcohol Concentration (BAC)
Roadside screening: breathalyser (fuel cell/semiconductor), must comply with .
Confirmatory: venous blood draw → headspace GC-MS quantification.
Samples preserved (e.g.
sodium fluoride) for retesting.
Alcohol pharmacology myths
Body size vs.
fat distribution: fat tissue absorbs less ethanol → higher % body fat yields higher BAC at same dose.Sex differences: women usually higher BAC owing to size & lower gastric ADH.
Empty stomach accelerates absorption but not clearance.
Coffee or stimulants do not affect metabolic clearance (≈ (0.015\,\text{mg·mL}^{-1}\,\text{h}^{-1}) irrespective of conditions).
Random Drug Testing
Screening: saliva swab → immunoassay panel (amphetamines, THC, opiates, cocaine, etc.).
Confirmation: GC-MS of blood, saliva, or hair; sample stored for chain-of-custody integrity.
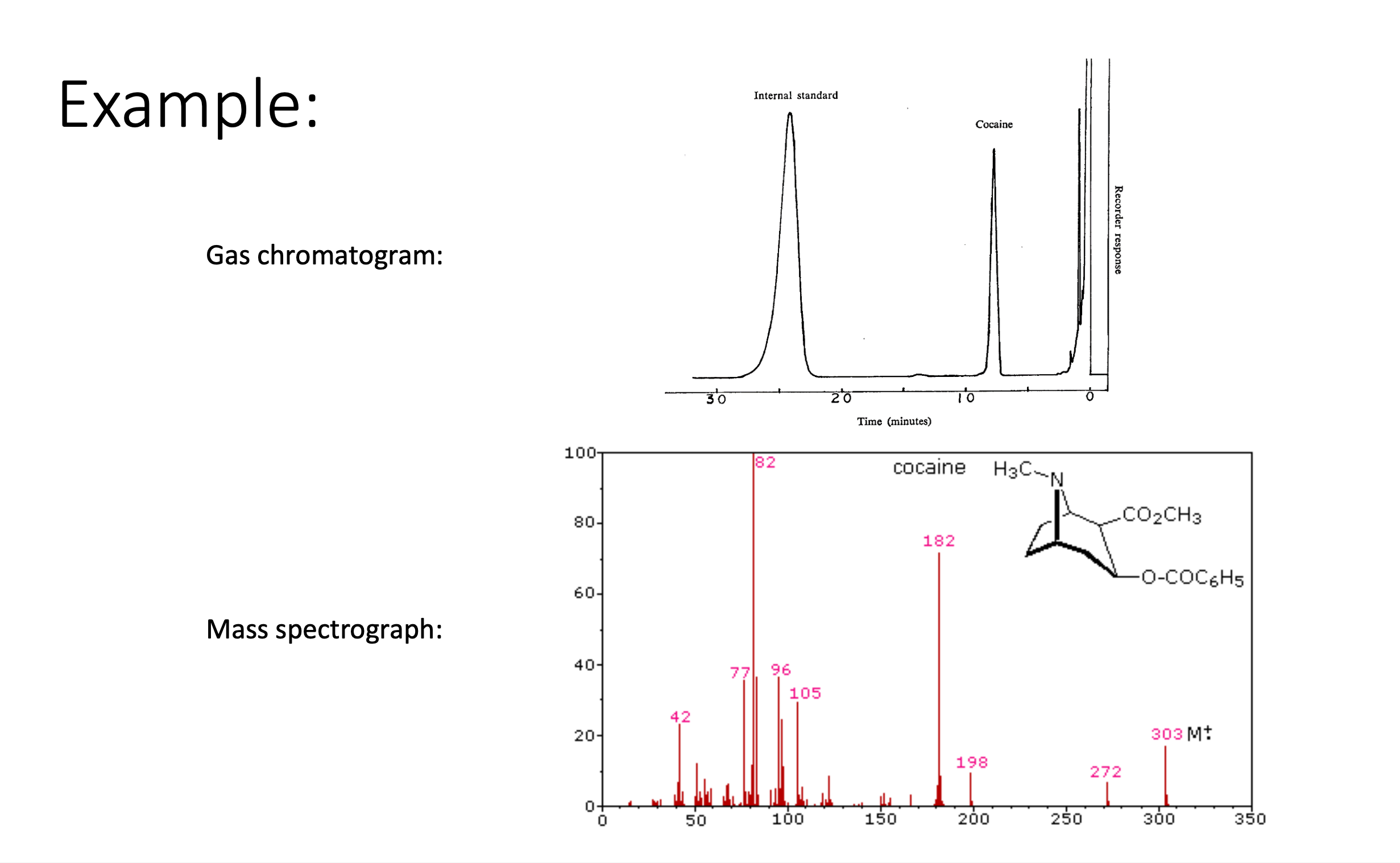
GC-MS Demonstration (Cocaine)
Gas chromatogram separates analytes by retention time; internal standard ensures quantitation accuracy.
Mass spectrograph displays ion fragments ( values 303, 272, 182, 198, etc.) — unique “fingerprint” verifying cocaine identity.

Common Pitfalls and Sources of Error
False Positives
Immunoassay cross-reactivity: structural analogues, over-the-counter meds.
Endogenous acetone in diabetics falsely elevating breath alcohol devices.
Legal drug metabolites identical to illicit drug metabolites (e.g.
dextromethorphan vs.
PCP screening).Post-mortem redistribution: higher cardiac blood concentrations; choose alternative matrices when possible.
False Negatives
Sample dilution (water loading, diuretics) masks urine drug levels.
Sampling too soon after ingestion (insufficient absorption) or long after (compound cleared).
Individual metabolic variation, rapid detox (e.g.
pseudocholinesterase hyperactivity with succinylcholine).Wrong analytical method for compound class.
Adulteration/tampering: bleach, vinegar, soap, lemon juice in urine.
Elevated lactate interfering with enzymatic assays.
Ethical, Legal, and Practical Considerations
Chain of custody: strict documentation to ensure evidentiary admissibility.
Consent & privacy: especially for workplace or sports testing.
Public health impact: data informs legislation (drink-drive limits, industrial exposure limits).
Resource allocation: vast chemical universe necessitates prioritising high-risk substances.
Evolving cultural standards influence intoxication definitions (e.g.
cannabis legalisation, medically assisted dying).
Interconnections to Other Disciplines
Pharmacology: therapeutic drug monitoring, antidote selection.
Analytical chemistry: development of new high-resolution MS, miniaturised sensors.
Pathology: correlation between toxicology findings and histopathological lesions.
Environmental science: tracking pollutants from industrial spills to human exposure.
Law & criminology: establishing mens rea, negligence, or intent via toxicological evidence.