6. Gene therapies
Gene Therapy- The insertion, alteration, or removal of genes within an individual's cells and tissues in order to treat disease
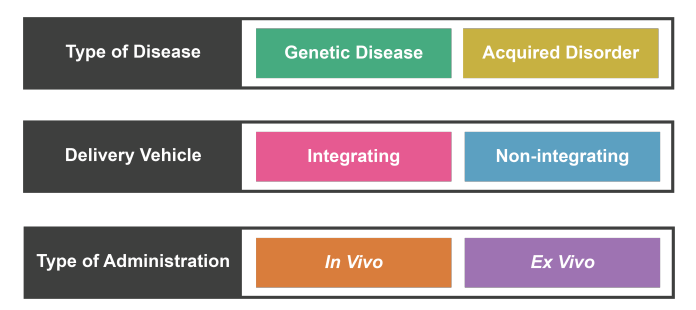
Classifying Gene Therapy
How you introduce into patient’s gene
Terminology
Transformation = transfer of naked DNA into bacteria.
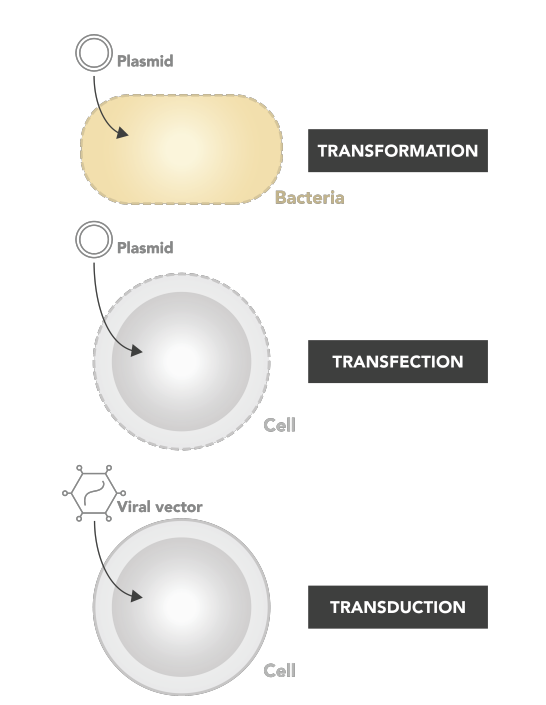
Transfection = transfer of naked DNA into cells.
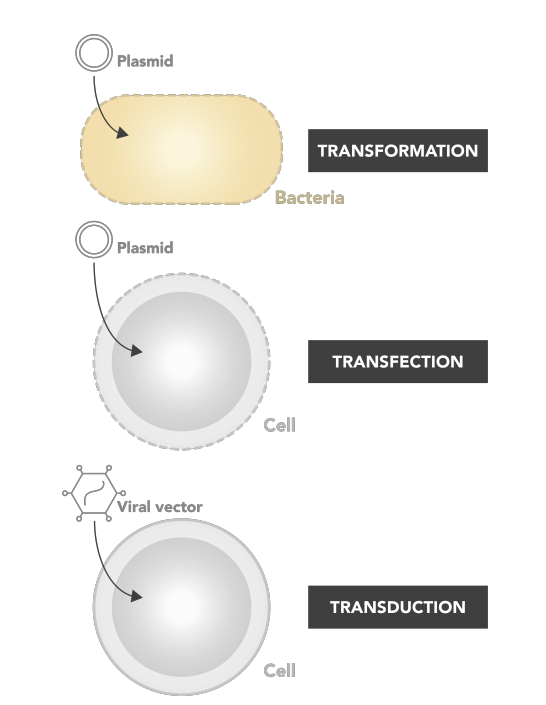
Infection = infect with a wild type virus.
Transduction = A viral vector with foreign DNA introduced into another cell
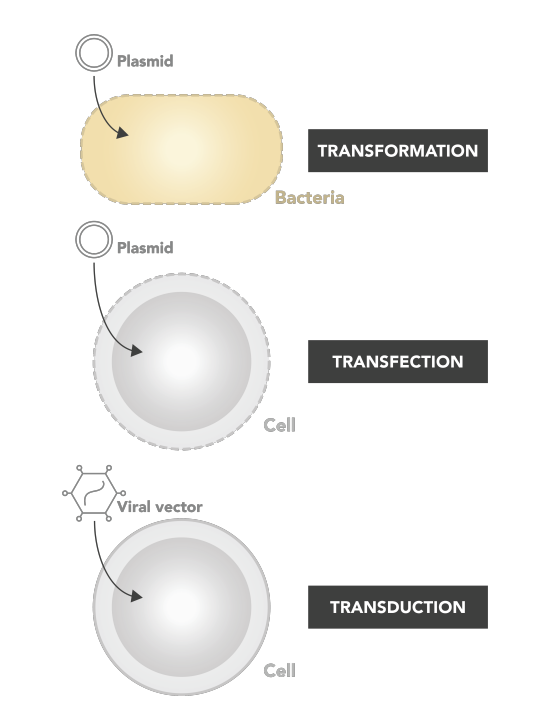
Titer = way of expressing concentration of viral particles (Infectious Units per ml, IU/ml)
Genetic background of diseases
How disease can occur
- Environmental factors: infectious diseases
- Genetic factors: inherited disorders
- Monogenic (caused by one gene)
- Polygenic (requires many genes to manifest) ← uncommon
- Environmental factors + genetic factors: cancers, chronic diseases
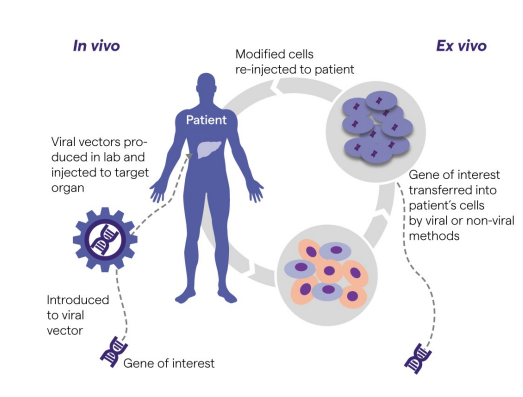
Ex vivo or In vivo
Ex vivo: Gene of interest transferred into patient’s cells. Ex vivo means "out of the living" in Latin.
In vivo: Viral vectors produced in the lab and injected into the target organ. In vivo means "within the living" in Latin.
Ex vivo: Allogenic and autologous
Allogeneic: from a donor
Autologous: from the patient itself
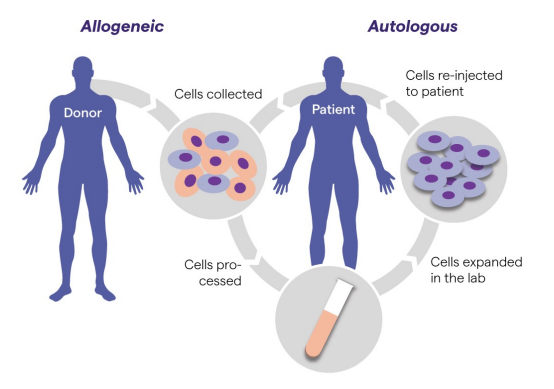
Autologues-
Advantages:
•Patient-specific
•No rejection by the immune system
•No risk for graft-vs-host disease
•Repeated doses possible
Challenges:
•High costs for manufacturing and quality testing
•Starting material variability (donor variability)
Allogenic-
Advantages:
•Larger-scale manufacturing possible
•Donor can be screened for desirable characteristics
•More reproducible manufacturing (less donor variability)
•Banking possible (immediate availability)
Challenges:
•Risk for graft-vs-host disease needs to be minimized through additional steps (cost increase)
•Risk of rapid rejection as cells are still recognized as foreign cells
Plasmids
- Circular double stranded DNA molecule, can be propagated in bacteria
- Naked DNA (i.e. no surrounding membrane, outside the nucleiod)
- Can replicate independently of chromosomal DNA in the cell
- Excellent gene carriers
Plasmid production
Bacterial transformation is a process of horizontal gene transfer (transmission of DNA between different genomes) by which some bacteria take up foreign genetic material (naked DNA) from the environment.
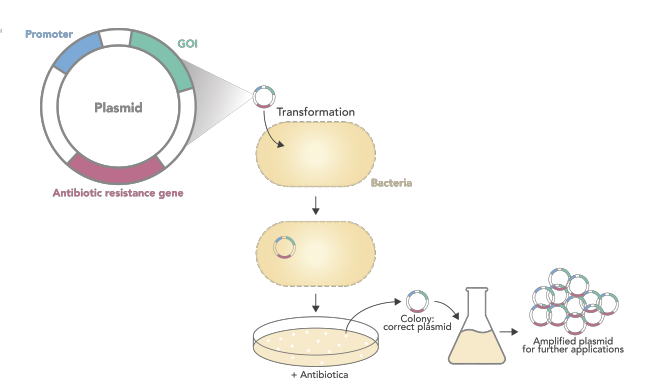
Plasmid mediated gene delivery
• Low gene transfer efficiency
• Only transient expression (non-integrating)
• Difficult with primary cells
→ This why people have moved onto Viral vector
Delivery Vehicle: Viral
Two main classes
- Integrating
- Retrovirus
- Lentivirus
- Non-integrating
- Adenovirus
- Adeno-associated virus
Adenovirus
- Class of virus with double-stranded DNA genomes
- infect dividing and non dividing cells
- DNA is not integrated into host cell genome
Pros:
- High efficiency of transduction
- High levels of transgene expression
- Rarely integrates into hest cell genome
- Infect both dividing and non-dividing cells
- Easily produced in high amounts
Cons:
- Causes common cold symptoms in human
- Difficult to achieve persistent gene expression - since non integrating
Adeno-associated virus (AAV)
- Class of small, single-stranded DNA virus
- transduce both dividing and non-dividing
- DNA is not integrated into host cell genome (it can integrate but at low frequencies)
Pros:
- Not pathogenic to humans
- Low immunogencity
- Low cytotoxicity
- Smaller than Adenovirus (1/3 of the diameter), have a relatively small packing capability
Cons:
- Difficult to achieve persistent gene expression - since non integrating
Retrovirus
- Class of virus that can create double-stranded DNA copies of their RNA genomes.
- These copies can be integrated into the chromosomes of host cells.
- The vector genome integrates into the host cell genome, whereby persistent transgene expression is expected.
- Most retrovirus can only infect dividing cells.
Pros:
- Persistent transgene expression is expected
- Non-dividing cells resistant
Cons:
- Due to the integration, there is a potential risk of insertional mutagenesis
Lentivirus (e.g.HIV)
- Sub-species of Retrovirus
- Unique ability among retroviruses of being able to infect non-dividing cells
- Successful transduction of host cells by lentiviral vectors does not require active cell division because of nuclear transport mechanisms (not require dividing cells due to that transport)
Pros:
- Persistent transgene expression is expected
- Less likely to activate adjacent oncogenes
- Tends to integrate more within active genes, but less frequently upstream of transcriptionally active promoters.
Cons:
- Due to the integration, there is a potential risk of insertional mutagenesis, although less than with gammaretroviral vectors, because it is more random
Clinical example: ADA-SCID
• A rare disorder caused by the retrovirus inserted near a proto-oncogene promoter.
• Caused by a mutation that results in the absence of protein called adenosine deaminase (ADA), which is required for the production of lymphocytes.
• Children born with ADA-SCID do not develop a healthy immune system so cannot fight off
everyday infections, which results in severe and life-threatening illness.
CAR-T cell therapy- CAR-T cell therapy involves re-engineering a patient’s own T cells to recognize and destroy cancer. T cells are currently being explored as carriers for chimeric antigen receptors (CARs)
- A specific cell type (e.g. T, γδ Τ) is isolated from the patient’s own blood.
- These T cells are genetically altered to express a TCR (T cell receptor) that binds to a specific antigen on the patient’s tumour.
- Expansion of these cells and re-infusion follow.
- CAR-T cells attack cancer cells
Drawback: tight control by natural cytotoxicity receptors and KIRs, which therefore might be educated over time to avoid tumour recognition
Pros:
- When using patient’s own cells, no risk of graft-versus-host disease
- Potential for lasting immunity even after a single infusion
Cons:
- High cost (personalized gene therapy)
- Length of time required for T-cell processing and modification (a delay can be detrimental)
- Adverse events including cytokine release syndrome (CRS)
- Natural cytotoxicity receptors and KIRs tightly regulate the activity of γδ T cells, which therefore might be educated over time to avoid tumour recognition
Functional Challenges in CAR T cell therapy (solid tumor)
- hard to get the T cells to traffic into the tumor cells
- hard to make the functional for a long time