LAs Practicum + US
History of Local Anesthetics
Local anesthetics block nerve conduction, specifically pain signaling/conduction
First clinical local anesthetic (1880): Cocaine, classified as an amino ester.
Followed by Benzocaine, Procaine, Tetracaine, and Chloroprocaine.
In 1950, Lidocaine, an amino amide, was introduced offering lower allergic reactions and greater stability.
Summary: esters —> amides
Anatomy of Nerves and Conduction
Local anesthetics affect both the CNS and PNS, which are differentiated by the presence of Myelin sheath. With the axon functional unit surrounding the cell membrane/axolemma
PNS has myelinated nerve fibers, which is created by Schwann cells (1:1 ratio). Unmyelinated segments are located between Schwann cells, known as the Nodes of Ranvier; This is the site of conduction via axonal voltage-gated sodium channels exposed (d/t no myelin), and where LA MOA should occur.
Saltatory Conduction: Action potentials jump from node to node, “skipping” areas of the nerve. ONLY OCCURS WITH MYELINATED NERVES — Logic: Myelin acts as an insulator. Since ions can't easily flow through the fatty myelin sheath, the electrical signal "jumps" from one gap to the next, resulting in a much faster signal transduction. In unmyelinated axons, however, the sodium channels are distributed evenly along the entire length of the membrane; Without myelin to "throw" the signal forward, the depolarization has to happen continuously, like a falling row of dominos… This is known as continuous conduction, which is much slower than saltatory conduction.
**While cAMP and IP3 mediate intracellular metabolic changes, sodium acts as the physical messenger that propagates the action potential from one Node of Ranvier to the next in myelinated fibers.
CNS has white matter (myelinated axons) and gray matter (neuronal cell bodies and unmyelinated axons). The myelin here is created by oligodendrocytes (ratio is 1:50, so one oligodendrocyte can serve many axons).
CNS Toxicity: You do NOT want large amounts of LA in CNS, which is caused by LAST or rostral/cephalid spread of neuraxial anesthesia
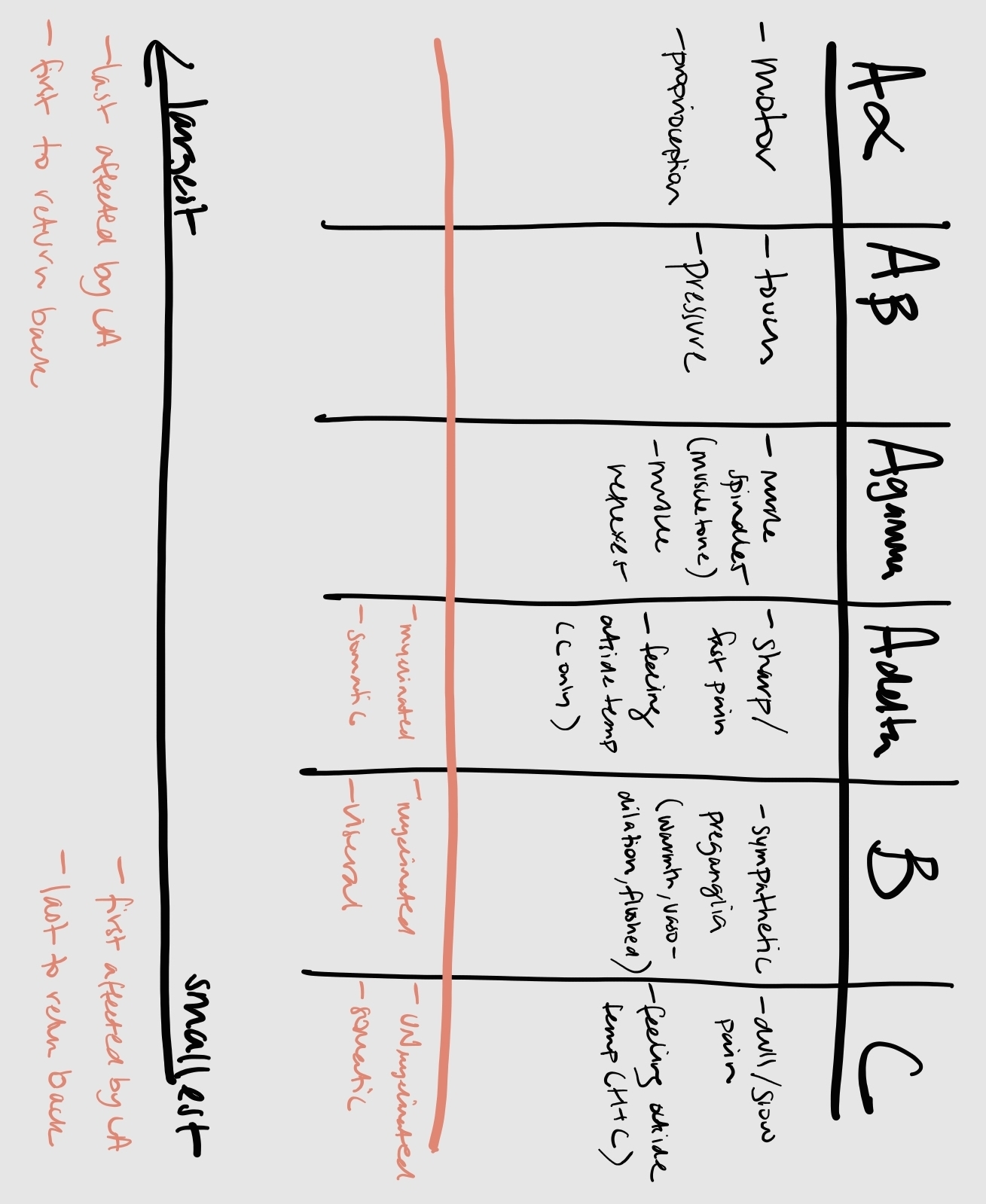
Classification of Nerve Fibers (by diameter, myelination, and function)
Larger Diameter = Faster Conduction: Myelination also enhances velocity. However, makes it HARDER for LA to penetrate through d/t thicker layers. 3 Connective tissue layers also make it harder (why you need a “3-node” block threshold for full analgesia)
From largest diameter to smallest diameter:
A-alpha fibers: motor function and proprioception
A-beta fibers: touch and pressure
A-gamma fibers: Muscle spindles (muscle tone) and muscle reflexes
A-delta fibers: pain (fast/sharp), and outside temperature (only cold)
These are myelinated, somatic
B fibers: Preganglionic sympathetic/autonomic (vasodilation, color, warmth, flushing)
There are myelinated, visceral
C fibers: pain (slow/dull), outside temperature (Hot AND cold)
These are unmyelinated, somatic
Block MOA of LAs
Summary: Resting state (Na channels are closed, appearing like a ‘V’) —> Activated state (Na channels are open, appearing like a ‘H’) —> Inactivated States (hyperpolarization, appearing like an ‘H’ but with a circle plug in the middle part) —> resting state
LAs can only work when the receptor is at least one of the two H shapes, as it needs something to actually bind to.
LAs do NOT affect the resting membrane potential; they only block Na influx which then prevents the neuron from reaching threshold potential
Phasic “use dependent” block: Describes how the drug binds based on specific nerve activity. LAs bind reversibly to THE ALPHA SUBUNIT on the intracellular side of the voltage-gated sodium channel. These channels exist in three states: closed (resting), open (active), and inactive (hyperpolarized). LAs bind preferentially to channels that are in the open (active) or inactive (hyperpolarized) states, NOT the resting (closed) state. This explains why LA target pain signals more aggressively than other nerve cell functions (e.g., motor pain signals are firing less often d/t patient immobilization vs getting the hip incised into)
Guarded (modulated) receptor hypothesis: Because the drug can only gain access to its binding site when the channel is open or inactive, the more frequently a nerve fires (repetitive depolarization), the more "opportunities" the drug has to bind and block the channel. LA block becomes stronger and develops faster when the nerve is actively stimulated/repetitively depolarized.
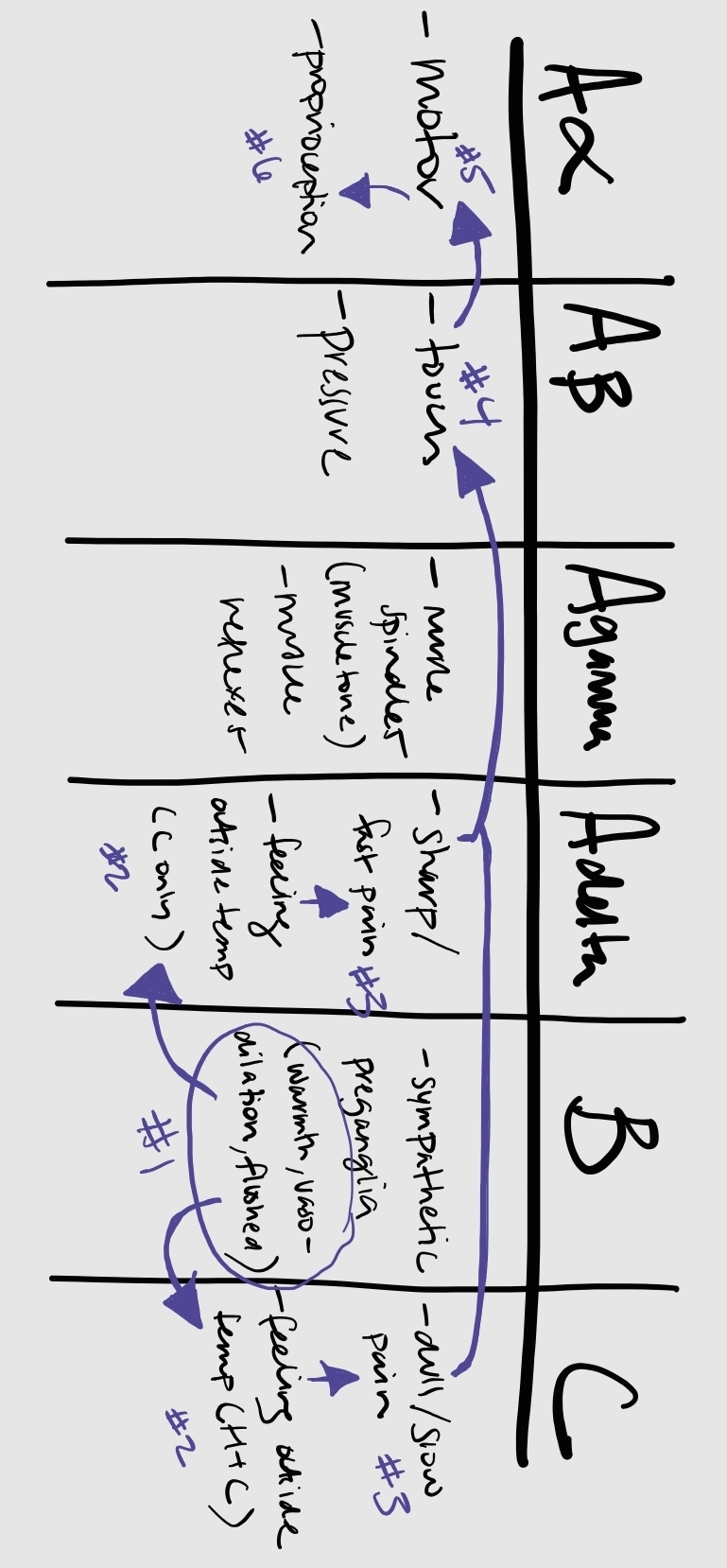
Differential Block: Describes how not all sensory or motor sensations are blocked equally, as Na and K channels exist in different concentrations in different nerve fibers. It also depends on the how well LA can reach the target nerve in adequate concentrations via diffusion through the tissues/neurons. Only 1-2% of injected LA ultimately penetrate the nerve (why accurate placement of LA is important). Depends on the amount of drug at target location. From first to last to be blocked (most sensitive to least sensitive):
autonomic (flushing/warmth d/t vasodilation) —> temperature (ability to detect hot/cold) —> pain —> touch —> proprioception/motor
OR: B fibers —> C/A-delta —> A-beta —> A-alpha
OR sympathetic/parasympathetic —> sensory —> motor
Tip: B-fibers are actually lost before C-fibers d/t myelination and the presence of very short Nodes of Ranvier between the small nerves, while C-fibers are smaller but are unmyelinated, and thus have no Nodes of Ranvier
Clinical Example #1: A Labor Epidural —> We use a low concentration (e.g., 0.125% Bupivacaine) to block the small pain fibers (C and A-delta) while allowing the mother to still move her legs (A-alpha). Basically, smaller nerve sizes need much lower concentrations than the larger nerve sizes to be blocked.
Clinical Example #2: Always warn your patients "You might feel me moving you around or feel some pressure and tugging, but you should not feel anything sharp or cold.”
Remember though: you lose the small —> large sized nerve functions, and gain back large —> small sized nerve functions. BUT, theoretically, it is correct to say that the “first thing you lose is the last thing you get back”, since your full function of it will take the longest to diffuse completely away since it requires the least amount of LA to remain blocked
pKa of LAs
pKa: The pH at which 50% of the drug is ionized (water-soluble) and 50% is non-ionized (lipid-soluble). THIS IS NOT THE SAME AS LIPID SOLUBILITY.
Important differences:
pKa determines the speed of onset
protein binding determines the duration of action
lipid solubility can also impart effect on duration of action bc higher lipid solubility = highly protein bound
lipid solubility determines the potency
Ion trapping: Occurs when a drug (usually a weak base like a local anesthetic or an opioid) crosses a membrane and enters a more acidic environment, where it becomes ionized (charged), gets "stuck," and cannot easily diffuse back out. NOT the same thing as LAST though
Similar to if the patient becomes acidotic (previously normal pH): the weak base LA is now further away from the pH, so it is more ionized (bases are more basic below) and will have slower onset as a result. Vice-versa, the higher the pKa OF A LA, the more ionized it becomes and slower onset, too, as it is further from the physiologic pH.
Occurs with BBB, placenta, and acidic wound infections
Unique fact: All LAs are weak bases, but in the body they exist in equilibrium between non-ionized and ionized forms. This means the non-ionized form is what penetrates the nerve sheath through the lipid membrane barrier, while the ionized form binds the receptor inside the channel for its main blocking effects.
Impact on Speed of Onset:
Agents with a pKa closer to physiological pH (7.4) have quicker onset because they are the least ionized. These are generally in the amide class, with lidocaine being the closest to physiological pH
Agents that are more ionized (further from physiological pH) have a slower onset. These are generally in the ester class
Chloroprocaine is an exception to this rule; despite the highest pKa, it has a fast onset d/t low toxicity meaning you can give it at very high concentrations (3%)
Classifications of LAs
LAs have a characteristic structure, comprising:
Benzene Ring (lipophilic); looks like a hexagon
Quaternary Amine (hydrophilic); positively charged NH group as an AA makes a weak base
Tip: think of quaternary amines for NMBDs chapters that means they do NOT cross BBB; however, if there are high concentrations, it can build up and diffuse into the brain
These are bound by an Intermediate Chain, which is either ester or amide (which is the main way that LA are classified, regulating the duration of action, metabolism, and allergy potential)
Esters vs Amides
Esters:
Intermediate chain contains CO bond
Metabolism: Rapidly hydrolyzed by plasma cholinesterases (except Cocaine, which is the only ester to go through liver metabolism). **in plasma, NOT CSF. Must diffuse from CSF to blood before getting metabolized.
Because so rapidly metabolized, there is a shorter duration of action
Also typically have lower levels of protein binding and lipophilic properties
Esters experience slowed/delayed metabolism in conditions like pregnancy, liver disease, increased BUN, atypical plasma cholinesterase, some chemotherapeutic drugs, and decreased hepatic blood flow (e.g., giving volatile anesthetics)
Higher allergy potential due to PABA metabolites, specifically Type 1 hypersensitivity reaction (pruritis and erythema, NOT anaphylaxis)
There IS cross-sensitivity (do not give any other esters with a true ester allergy, and instead give an amide)
EX: Procaine, Chloroprocaine, Tetracaine, Cocaine, Benzocaine.
Tip: One ‘i’
Usually slower onset than amides d/t pKa >8.0; exception if chloroprocaine because not very toxic and can give high concentration
Amides:
Intermediate chain contains NHC bond
Metabolism: liver metabolism
Slower metabolism means there is a longer duration of action and more chances of systemic buildup (aka, LAST)
Also typically have higher levels of protein binding and lipophilic properties
However, this also means rate of elimination will be prolonged/delayed if liver dysfunction present or if there are abnormal protein levels
Allergies are rarely seen compared to esters.
If there is a true allergy, there is NO cross-sensitivity (you can either give another amide or an ester)
Usually, the allergy is “due to epi” or actually the preservative (Methylparaben)
EX: Lidocaine, Mepivacaine, Prilocaine, Bupivacaine, Ropivacaine.
Tip: Two ‘i’s; usually preferred compared to esters
Usually faster onset than amides d/t pKa <8.0
**NOTE: LAs actually have significant pulmonary (lung) first pass elimination, more than liver for initial decrease in serum levels
Pharmacodynamics/Pharmacokinetics
Risk for LAST is most directly related to potency
Higher Local Anesthetic Concentrations = quicker action onset.
Importance: giving more volume but lower concentration will be SLOWER than giving lower volume but high concentration
Vasodilation Effects: LAs generally cause vasodilation; vasodilation not only shortens the duration of your block (because the drug leaves the site faster), but it also increases its own peak plasma concentration in the blood, bringing you closer to the toxicity threshold (LAST).
Exceptions: Cocaine is a vasoconstrictor
**Ropivacaine and lidocaine are also somewhat vasoconstrictive compared to the other LAs
Additionally, this explains why Epi is frequently added to LA to cause vasoconstriction
Total Dose: Linearly responsible for plasma levels, regardless of concentration or volume. Basically, this refers to how once the LA is absorbed from the tissue into the bloodstream, the blood doesn't care if it started as a "concentrated" or "diluted" liquid. It only "counts" the total number of milligrams circulating. This then creates the “maximum safe dose” numbers we need to know to prevent LAST.
Slower rate of absorption = reduced LAST risk; faster speed of absorption = increased LAST risk. This is referring to circulating BLOOD absorption
This can be caused by accidental IV injection, highly vascularized sites, and lack of vasoconstrictors
Fastest to slower absorption rate, based on tissue vascularity (aka, highest to lowest risk of LAST):
Blood (highest, bc straight into systemic circulation, arterial > venous injection) > Tracheal > Interpleural > intercostal > Caudal > Paracervical > Epidural > Brachial > Sciatic/femoral (legs) > SQ > topical (lowest); “BICEPS” kind of helps
Remember, we inject LA directly to the site of action, so systemic absorption (which is higher in more vascularized areas) actually DECREASES the duration of action. This can increase risk of LAST
Duration of action: related to protein binding
More protein binding = longer duration of action (same as with the vasoconstrictor/vasodilation concept; by having more protein binding, the LA stays at the effector site for longer instead of being washed away by the blood)
Most to least protein binding (basically the same ranking also for lipid solubility/potency from most to least): levobupivacaine > bupivacaine > ropivacaine > tetracaine > mepivacaine > lidocaine > prilocaine > procaine (6%) > chloroprocaine (0%)
**There is a nearly linear relationship between lipid solubility and protein binding.
LA bind primarily to alpha1-acid glycoproteins; some secondary binding to albumin may occur as well
Distribution: Once systemically absorbed, LAs distribute rapidly through the body
Rapid distribution = lower plasma concentration and first goes to the vessel-rich groups (heart/brain), can cause CV and CNS toxicity
Once equilibration occurs, LA leaves highly vascular tissue and shifts to less perfused tissue; muscle tissue receives the greatest amount of LA from redistribution effect
Book Notes
Absolute contraindications to LA: Patient refusal, infection at site, severe coagulopathy (d/t risk of spinal hematoma in case everything goes wrong)
Relative contraindications: pre-existing neuropathy (risk of “double crash'“ phenomenon), liver disease, systemic bacteremia
Dense block means there is no motor function left; light/walking blocks means there is analgesia but some preserved motor function
Dense block always occurs with spinals/C-section, while epidurals are light/walking blocks. You control dense or light block by the concentration
Opioids (morphine and fentanyl) provide a denser block on the initiation of the LA, but once LA wears off, there is no loss of motor function with CONTINUED pain relief
Precedex, clonidine, toradol? can also be added to provide faster onset and significantly prolonged duration of action
Peripheral regional blocks last longer than spinals, but epidural that are continuous last the longest
EMLA cream: 50/50 mix of lidocaine and prilocaine, producing analgesia in 1 hour with peak effect 2-3 hours/ A dressing should be applied after the cream is applied.
self limiting skin layer is the stratum corneum
Transdermal lidocaine patch will achieve effect in 30 minutes
US Physics:
High Frequency (>10 MHz): High resolution, shallow depth
d/t shorter wavelength
Low Frequency (<5 MHz): Lower resolution, deep penetration
D/t longer wavelength
Transducer array configurations:
linear: parallel tip; high frequency. EX: nerve blocks.
curvilinear: convex tip; medium frequency. EX: abdominal US’s, obese/pregnant patients.
phased array: small square tip; low frequency. EX: echocardiograms.
Gain = brightness; increased gain = increased brightness
Depth: The vertical field of view/window
You want the object of interest to fall into the middle of the screen monitor
Measured in cm on the R side of the screen monitor
4 cm for most deep structures
1-2 cm if superficial structures (e.g., Radial A-line insertion)
Echogenicity: amplitude of the returning echoes, based on different tissue types
anechoic/echolucent: No returning sound waves, so the structure will be black color. EX: blood, free air, fluids, vessels
hypoechoic: only some returning sound waves, so the structure will be a grey color. EX: muscles, adipose, cartilage, skin, solid organs
Nerves will typically appear as a hypoechoic round/triangle/oval area encircled by a relatively hyperechoic area; similar to a honeycomb pattern. They are noncompressable, nonpulsatile, and do not change in size as you move along an axis. There is always a triad (nerve next to an artery for sure, and a vein should be somewhere nearby)
hyperechoic/echogenic: large amount of returning sound waves, so the structure will be white in color. EX: bone, fascia
Arteries vs veins: Arteries are more defined circular structure, pulsatile (when compression is attempted), and cannot be easily compressed; Veins are nonpulsatile and can be easily compressed
In-Plane vs. Out-of-Plane:
In-plane: needle parallel to long axis of the probe, which allows visualization of the entire needle shaft. Typically used for deeper invasive techniques or as confirmation method
out-of-plane: needle perpendicular to long axis of the probe, which shows the needle as a dot (only a cross-section). Typically used for regular US IV cannulation (aka, difficult stick)
Probe maneuvers:
Macro/gross movements are used when doing initial sweep over a large area of interest
sweep: move along short axis of transducer
slide: move along long axis of transducer
tip: think about slides being long in appearance, while imagining a broom for sweeping
compression: less or more pressure in a stationary position
increased compression help improve US image resolution by pushing air away (either in abdomen or US gel) and reducing the distance between the anatomical target and transducer. The most common reason for a black screen is not enough gel!
micro/fine movements are used for pinpointing/decision making
rocking: while in stationary position, change angles along long axis of transducer
Think about the rocker on ice skates
fanning: while in stationary position, change angles along short axis of transducer
rotation: turn transducer clockwise or counterclockwise
Usage guidelines:
There is an orientation marker on the probe (a protruding/colored line), which correlates with the indicator symbol on the Top L/R corner of the US screen.
The Golden Rule: The notch on the probe should always correspond to the indicator icon on the ultrasound screen for direct Correlation (aka, if you move the probe to the left, the image on the screen also moves to the left)
Always insert the IV into the center line on the probe; you can add a visual line marker on the screen as we
Hold the probe close to the tip for stability instead of closer to the top/cable