mol/mass calculations
Set 5 Q10, 11, 12
Define the ‘mole’ and Avogadro’s number.
Mole is the standard chemical unit for describing the amount of a substance in terms of the number of particles in the atom
Avogadro number 6.022 × 10²³ = 1
Calculate molar mass of elements in chemical formulae.
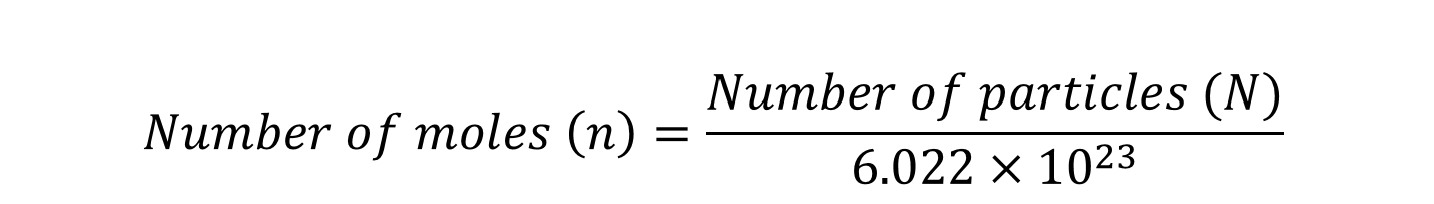
Calculate the moles of each type of atom/ion in a compound when the number of moles of the compound is known.
Molar mass, mass of one mole of its atom (g/mol)
atoms mol(xamount of each atom) + atoms mol (xamount of each atom) in substance= Molar mass
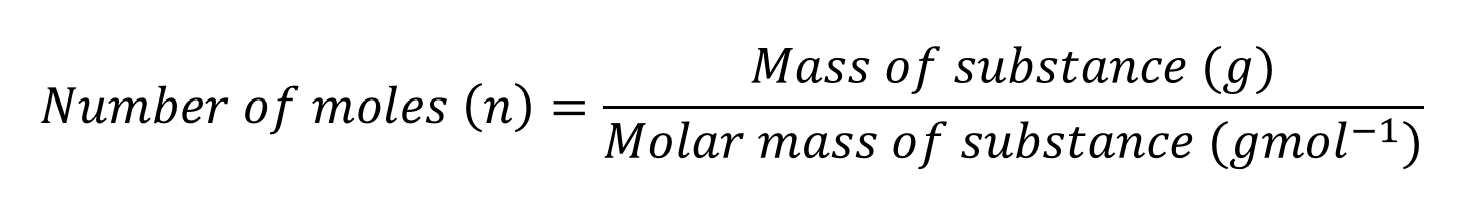
Mass calculations
Calculate the mass of a substance given the number of moles
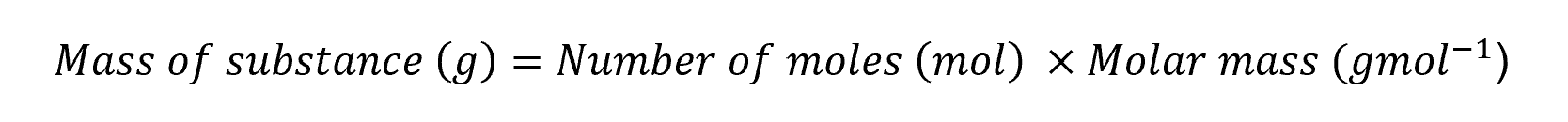
Perform different types of percentage composition calculations
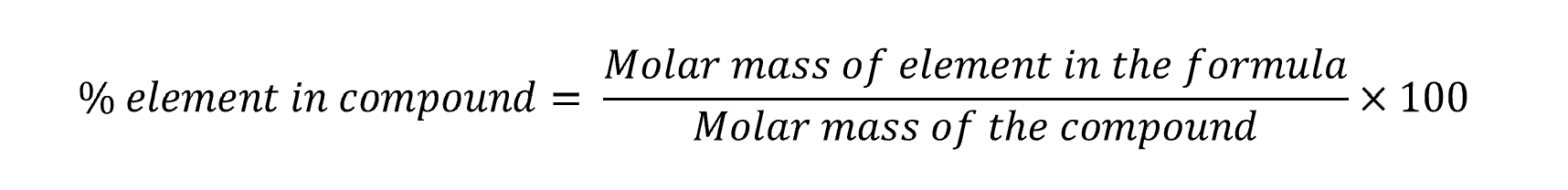
Percentage composition – Hydrated formulae
(Total molar mass of water / Molar mass of compound including water) x100
Percentage composition – Experimental measurements
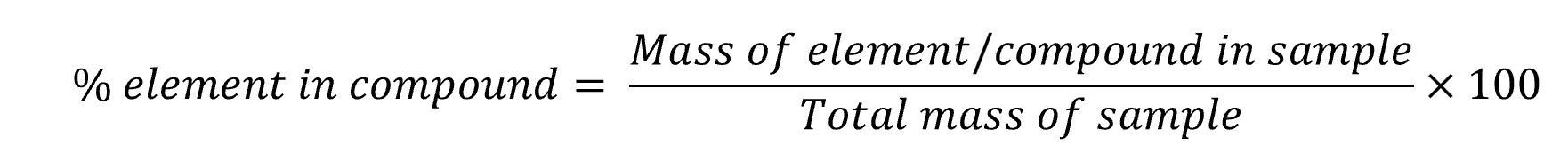
Mass of element or Compound in sample