Chapter 10 - Energy Changes in Chemical Reactions
10.1 Exothermic and Endothermic Reactions
Chemical Reactions
Occurs when particles collide and are rearranged to form new particles
Involve energy changes
Reactant particles rearrange, chemical energy of reactants change
Energy from a system can be released to surroundings, or the system can absorb from the surroundings
Chemical Energy
Chemical energy is stored in the chemical bonds between atoms and molecules. Cannot. be directly observed, only experienced through the effect of changes. This energy comes from;
attractions between electrons and protons
repulsion between nuclei
repulsion between electrons
movement of electrons
vibrations and rotations around bonds
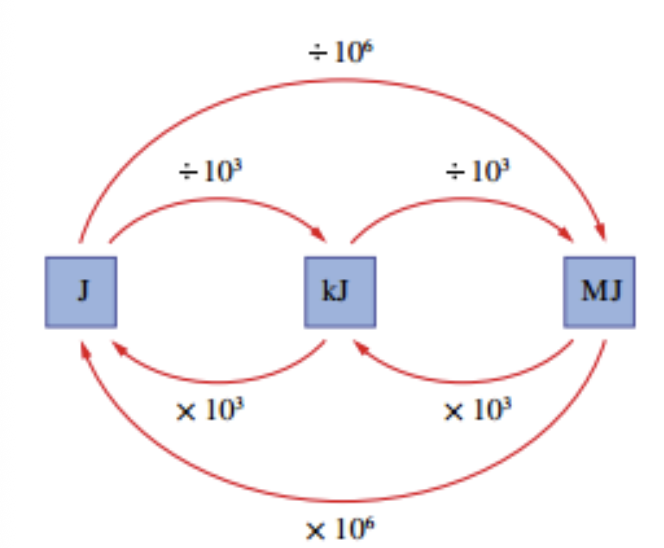
SI Units
Measured as Joules (J)
1 MJ = 1 000kJ = 1 000 000 J
1 kJ = 1000 J
Energy Conservation
Law of Conservation of Energy; energy cannot be created or destroyed. It can change forms (transform and transfer). Amount of energy stays the same.
Systems and Surroundings
System - Chemical reaction
Energy is released or absorbed by a system → referring to energy changes when bonds are broken and formed
Surroundings - everything else
Energy leaves the system and enters the environment/surroundings, or leaves surroundings and enters the system
Energy Changes during Chemical Reactions
Reactant in chemical reaction have certain amount of chemical energy stored in bonds
Products formed as a result of rearrangement of reactants
Energy is released + absorbed
Energy is needed to separate particles from each other.
Separated particles have more chemical energy than when together.
When together, the particles bonded together will have less energy than separate particles.
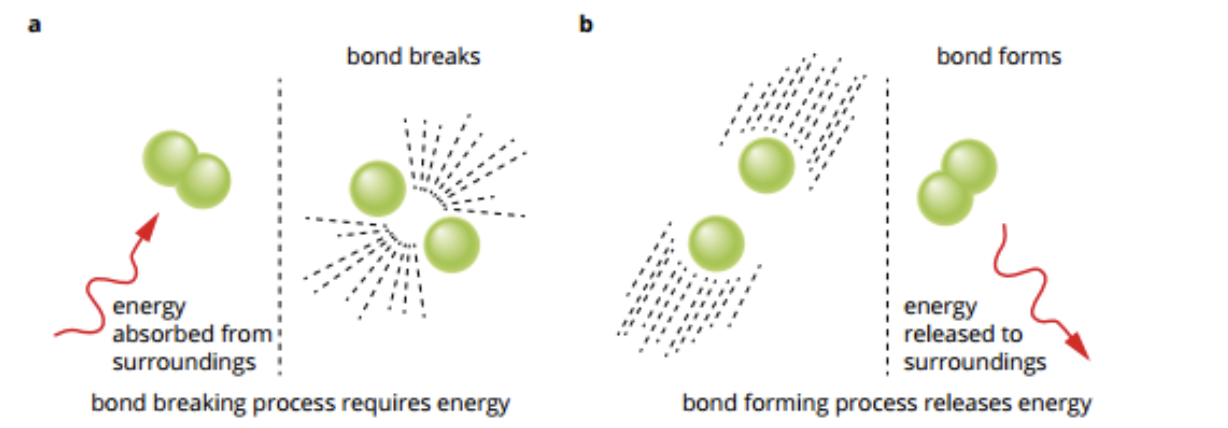
Energy can be released + absorbed as;
Thermal (heat)
Kinetic (light, electricity + movement)
Exothermic and Endothermic Systems
Exothermic Reactions
Definition - Energy is released (exits) in a reaction
Total chemical energy in products < total chemical energy in reactants
Lost energy is released into surroundings
Shown in an equation as ‘energy"‘
Hydrogen gas + Oxygen gas → Water + Energy
2H2 (g) + O2 → 2H2O (l) + Energy
Endothermic Reactions
Definition - Energy enters the system/reaction
Total chemical energy of products > total chemical energy of reactants
Energy is absorbed from surrounding environment
∴ Process that absorbs energy gets colder
Energy gained by the system is unobservable stored chemical energy → the energy is absorbed from surroundings which loses heat
Decomposing equation
Water + Energy → Hydrogen + Oxygen
2H2O (l) + energy → 2H2 (g) + O2 (g)
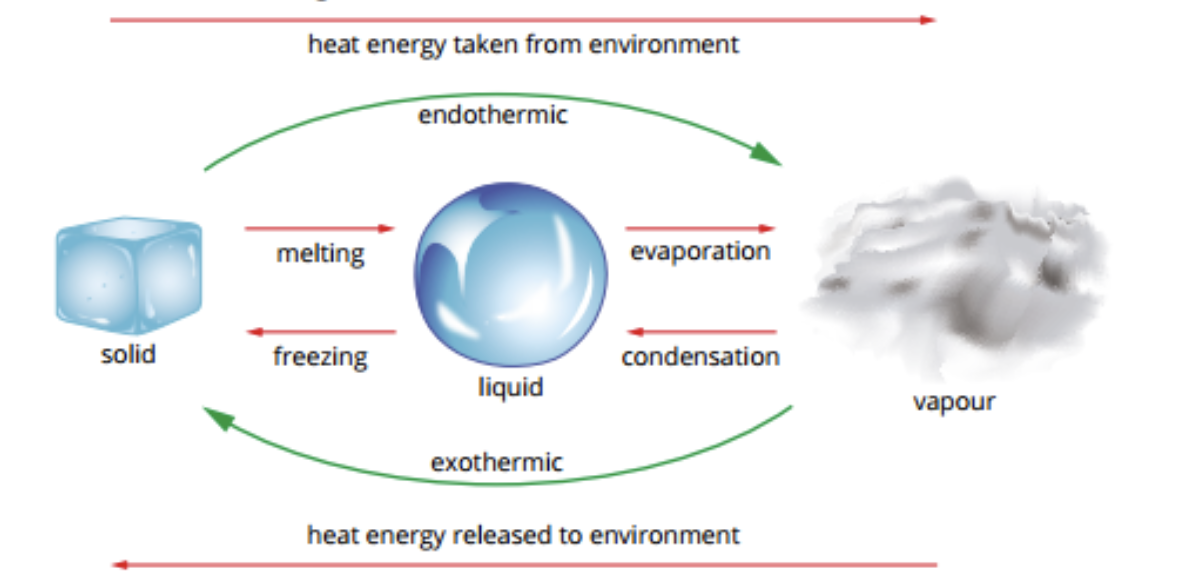
Changes of State
Also phase changes are a physical change, but also involve energy being absorbed and released
10.2 Thermochemical Equations, Energy Profile Diagrams and Enthalpy
Thermochemical equations - balanced chemical equation that includes the molar enthalpy change (\(\Delta H\)) of the reaction, indicating the amount of heat energy absorbed or released. It shows physical states (solid, liquid, gas, aqueous) of reactants and products because enthalpy depends on these states.
Enthalpy
Chemical energy stored in a substance (H)
All chemicals have a certain amount stored
The exchange of heat between the system and its surroundings = enthalpy change (△H)
For a chemical reaction (reactants → products) the enthalpy is calculates by
△H = H products - H reactants
Enthalpy Change in Exothermic Reactions
Enthalpy in products < enthalpy in reactants = energy is released from system into surroundings (exothermic)
System has lost energy ∴△H is negative
Enthalpy Change in Endothermic Reactions
total enthalpy of products > total energy of reactants energy is absorbed from surroundings (endothermic)
System has gained energy ∴ △H is positive
Thermochemical Equations
Shown by writing △H value to the right of the chemical equation
Unit = kJ mol-1 (Kilojoules per mole)
Means…amount of energy signified by the △H value corresponds to the mole amounts specified by the coefficients in the equation
Respiration Equation
C6H12O6 (aq) + 6O2 (g) → 6CO2 (g) + 6H2O (l) + energy
△H = -2803 kJ mol-1
Thermochemical equation tells you have; 1 mole glucose reacting with 6 moles oxygen to produce 6 moles carbon dioxide and 6 moles water, with 2803 kJ released to surroundings
C6H12O6 (aq) + 6O2 (g) → 6CO2 (g) + 6H2O (l) + 2803 kJ mol-1
⭐️ energy is on right side as a product in this exothermic equation
Enthalpy changes per mole of Reactant
Combustion of hydrogen gas in oxygen has the equation…
2H2 (g) + O2 (g) → 2H2O (l) △H = -572 kJ mol-1
To compare enthalpy changes you need to compare equal quantities of the substance
In this equation, you need everything to be 1 mole, therefore everything is divided by 2
H2 (g) + ½ O2 (g) → H2O (l) △H = - 286 kJ mol-1
Physical Changes
Thermochemical equations can be written for physical changes
Eg → Melting ice. Endothermic (heat needs to be applied to solid ice in order to break the bonds to turn into a liquid)
H2O (s) → H2O (l) △H = +6 kJ mol-1
△H is positive (endothermic)
H2O (s) +6 kJ mol-1 → H2O (l)
Effect on △H of Reversing a Chemical Equation
Reversing a chemical equation changes the sign but not the magnitude of △H
Eg; Methane reacts with oxygen gas to produce carbon dioxide gas and water (exothermic)
CH4 (g) + 2O2 (g) → CO2 (g) + 2H2O (l) △H = -890 kJ mol-1
When reversed…
CO2 (g) + 2H2O (l) → CH4 (g) + 2O2 (g) △H = +890 kJ mol-1
⭐️ demonstrates the law of conservation of energy is true for all reversible reactions
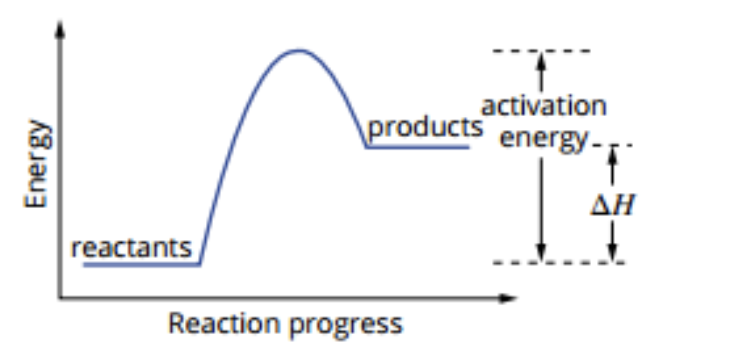
Activation Energy
Definition - Energy required to break the bonds of reactants so that a reaction can proceed. Energy barrier that must be overcome so a reaction can start
Exists for exothermic and endothermic
Activation energy low = not much energy required. Chemical reaction can be initiated as soon as the reactants contact (reactants have sufficient energy)
Energy Profile Diagrams
Exothermic Reaction
Energy is released
△H is negative
Absorption of energy
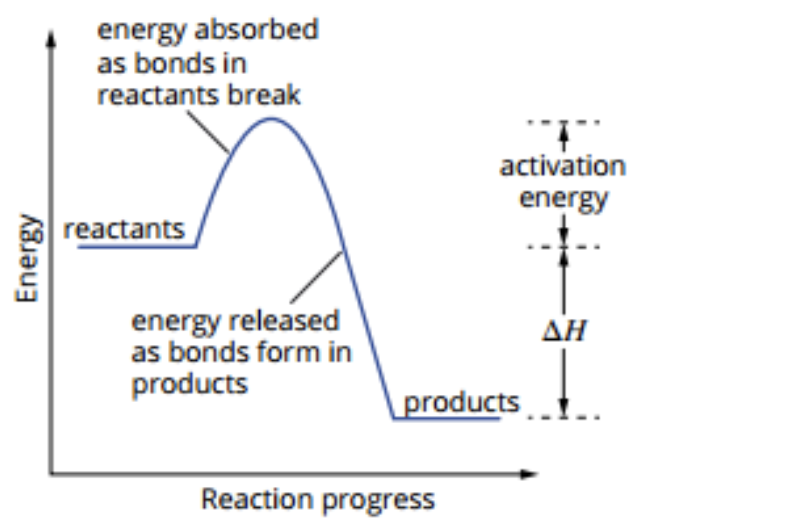
Endothermic Reaction
Energy is absorbed
△H is positive
Release of energy