Week 4 Pre-reading
9/17
Openstax 2.2.2,2.5; 2.3 is good background info, but we will not cover it in class
2.2 Evolution of Atomic Theory
J.J Thomson using a cathod ray, discovered the Cathode ray particke
They were attracted to + charge, repelled - charge
He suggested they were less massive than atoms and indistinguishable
This is what we call the electron <1/1000 mass of an atom
Mass of electron was found through known chage and charge-to-mass ratio
Mass of electron=1.602×10−19C×1kg1.759×1011C=9.107×10−31kg
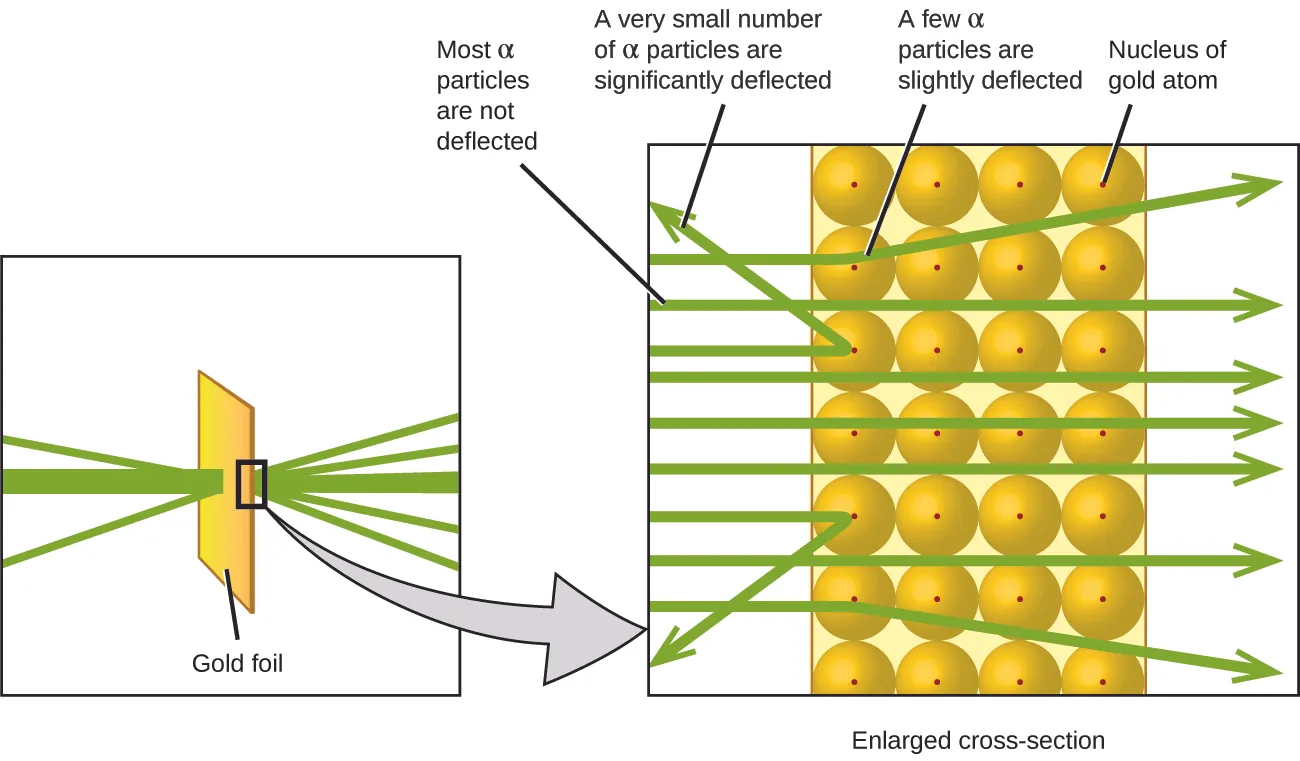
Here is what Rutherford deduced: Because most of the fast-moving α particles passed through the gold atoms undeflected, they must have traveled through essentially empty space inside the atom.
The volume occupied by an atom must consist of a large amount of empty space.
A small, relatively heavy, positively charged body, the nucleus, must be at the center of each atom.
Deduction of the proton
led the English chemist Frederick Soddy to realize that an element could have types of atoms with different masses that were chemically indistinguishable.
These different types are called isotopes—atoms of the same element that differ in mass.
The existence of the neutron also explained isotopes: They differ in mass because they have different numbers of neutrons, but they are chemically identical because they have the same number of protons.
2.3
When describing the properties of tiny objects such as atoms, we use appropriately small units of measure, such as the unified atomic mass unit (u) and the fundamental unit of charge (e).
The unified atomic mass unit is defined as exactly 1/12 of the mass of a single atom of the carbon-12 isotope (more on isotopes is provided later in this section) and is equal to 1.6605 × 10−24 g. The dalton (Da) is an alternative mass unit that is equivalent to the u.
The number of protons in the nucleus of an atom is its atomic number (Z)
The total number of protons and neutrons in an atom is called its mass number (A)
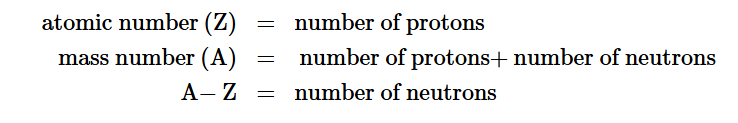
Atomic charge = number of protons − number of electrons
Ion- the atom is electrically charged
An atom that gains one or more electrons will exhibit a negative charge and is called an anion.
cations- Positively charged atoms,
Formed when an atom loses one or more electrons.
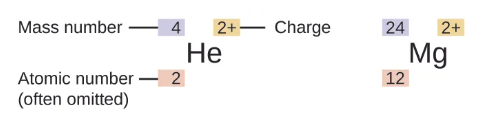
Atomic Mass:
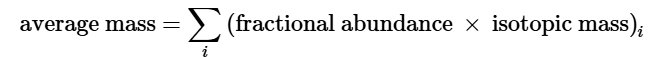
The Periodic Table
periodic law- the properties of the elements are periodic functions of their atomic numbers.
The elements are arranged in seven horizontal rows, called periods or series, and 18 vertical columns, called groups.
\metals (elements that are shiny, malleable, good conductors of heat and electricity—shaded yellow); nonmetals (elements that appear dull, poor conductors of heat and electricity—shaded green); and metalloids (elements that conduct heat and electricity moderately well, and possess some properties of metals and some properties of nonmetals—shaded purple).
the transition metals in the columns labeled 3–122; and inner transition metals in the two rows at the bottom of the table (the top-row elements are called lanthanides and the bottom-row elements are actinides
elements in group 1 (the first column) form compounds that consist of one atom of the element and one atom of hydrogen. These elements (except hydrogen) are known as alkali metals, and they all have similar chemical properties. The elements in group 2 (the second column) form compounds consisting of one atom of the element and two atoms of hydrogen: These are called alkaline earth metals, with similar properties among members of that group. Other groups with specific names are the pnictogens (group 15), chalcogens (group 16), halogens (group 17), and the noble gases (group 18, also known as inert gases).