apes semester 2
Semester 2
Unit 5: Land and Water Use
Module 29: Impacts of Mining
Lesson 29.1: How are natural resources extracted from Earth through mining?
Abundance of Ores and Materials
crustal abundance: The average concentration of an element in Earth’s crust
4 most common: oxygen, silicon, aluminum, iron make up most of crust
chemical composition highly variable in various locations
scientists study distribution + types of minerals to locate and manage extraction and conservation
ore: A concentrated accumulation of minerals from which economically valuable materials can be extracted
typically characterized by presence, but sometimes abundance
metals: An element with properties that allow it to conduct electricity and heat energy and to perform other important functions
copper, nickel, aluminum, varying concentrations and mixed w/ other metals
as demand for HEV increases, their manufacturing increases, and the demand for metals like lithium increase
ores formed by various geologic processes
some form in magma, some form in little/great abundance
nonmetallic resources like clay occur in concentrated deposits bc of physical/chemical separation from other materials by water
mine most accessible and high concentration first
less concentration and availability → more energy, disturbance, air pollution, etc.
difficult to quantify global mineral sources
reserve: In resource management, the known quantity of a resource that can be economically recovered (publicly known estimate)
economically recoverable resource → concentration high enough to be profitably mined
increased recycling and substitution will replace metals as they decrease
Mining Techniques
extract resources thru mining ore and separating other stuff from the sought metal
high concentration = less energy and waste
→ Surface Mining
strip mining: The removal of overlying vegetation and “strips” of soil and rock to expose underlying ore
overburden = overlying soil, rock, vegetation
increases erosion
mine parallel to surface → remove large volume of material, extract resources, return unwanted stuff
mine tailings: Unwanted waste material created during mining; chemical compounds and rock residues that are left behind after the desired metal or ore is removed
open-pit mining: A mining technique that creates a large visible pit or hole in the ground
close to surface, but extends horizontally and vertically, i.e. copper
mountaintop removal: A mining technique in which the entire top of a mountain is removed with explosives
deposit tailings in low elevation rivers
placer-mining: The process of looking for minerals, metals, and precious stones in river sediments
river water separates heavier materials from lighter sand/mud
→ Subsurface Mining
subsurface mining: Mining techniques used when the desired resource is more than 100 m (328 feet) below the surface of Earth
economically more expensive
horizontal dig into side of mountain, then vertical shifts with elevators
coal, diamond, gold
Lesson 29.2: What are the ecological and economic impacts of mining?
Mining Impacts our Ecosystems
several impacts on water, soil, biodiversity + economic and human health
construction of roads → erosion, bad waterways, fragmentation
mine tailings → health and eco consequences
coal mining → methane, greenhouse gas
can contaminate land with acids and metals
type of operation | effects on air | effects on water | effects on soil | effects on biodiversity | effects on human |
|---|---|---|---|---|---|
surface mining | dust and particles from equipment | water contamination from percolation via tailings | most soil removed from site, replaced if reclaimed | habitat destruction above mined surface areas | minimal, but nearby adverse effects from water and air quality |
subsurface mining | minimal dust, but significan fossil fuel emissions for equipment | acid mine drainage and percolation through tailings | habitat fragmentation from roads | occupational ahzards |
mountaintop removal for coal mining is much safer
companies make efforts to restore land, debate over efficacy of reclamation
damage to streams and groundwater
placer mining contaminates a lot of water and adjacent areas with sediment and chemicals
mercury is volatile and harmful
subsurface impacts less visible
acid mine drainage → water passes through tailings, becomes acidic, contains metals leached
pumps must always drain mines
lowers pH of nearby soil → another acid mine drainage
Mining affects Human Safety and the Economy
subsurface mining is a dangerous occupation
burial, explosion, fire, black lung disease, asbestosis
deposits of methane bw coal beds
dangerous to miners → explosive, suffocating
population growth → greater demand for mineral resources
demand for certain increases, but coal is decreasing
as easily mined stuff r depleted, efforts become more expensive economically and environmentally
more tailings
design systems that reuse and use mineral sources → protect environment and humans and lower costs for humans bc less healthcare needs
Unit 6: Energy Resources and Consumption
Module 35: Renewable and Nonrenewable Resources and Global Energy Consumption
Lesson 35.1: What are the characteristics of nonrenewable energy resources?
fossil fuels: Fuels derived from biological material that became fossilized millions of years ago
coal, oil, natural gas
anaerobic conditions → decomposers can’t break detritus, sediment, exposed to heat and pressure, transformed into things combustible easily, also adding co2 to atmosphere
nonrenewable energy source: An energy source with a finite supply, primarily the fossil fuels and nuclear fuels
unit of energy - Joule
calorie - Amount of energy it takes to heat 1 gram of water 1 °C; 1 calorie = 4.184 J → energy in food systems and food consumption
Calorie - Calories in food; 1 Calorie = 1000 calories = 1kcal = 4184 J; food labels, human food consumption
Btu - Amount of energy it takes to heat 1 pound of water 1 °F; 1055 J; Energy transfer in air conditioners and home water heaters
Kilowatt-hour (kWh) - Amount of energy expended by using 1 kilowatt of electricity for 1 hour; 3.6 mil J; Energy use by electrical appliances; often given in kWh per year
2 aspects
relatively large amount of energy in relatively small volume and mass
converted to useful heat energy quickly
best replacements → renewable, wind and solar
Lesson 35.2: What are the characteristics of renewable energy resources?
petroleum, natural gas, coal are finite and nonrenewable → depleted faster than replaced, uranium also finite
renewable energy sources: Sources of energy that are infinite
potentially renewable: An energy source that can be regenerated indefinitely as long as it is not overharvested
biomass, trees
nondepletable: An energy source that cannot be used up
solar, wind, geothermal, tidal, hydroelectric
humans have been using renewable energy sources for a while like wood, plants, manure, fish/animal oils before fossil fuels became available
still, in developing parts, rely on local biomass
Sub-Saharan Africa
Lesson 35.3: What are the trends of energy use worldwide and in the United States?
Worldwide Patterns of Energy use
oil, coal, natural gas (80%), renewable, nuclear makes up all energy use
600 EJ per year, 77 gj per person
hydroelectricity is largest renewable source, then wind and solar
US → 320 GJ per person per year
china great energy consumption, US greatest per capita
developed countries + urban areas more likely to use individual fossil fuels directly and indirectly
commercial energy sources: Energy sources that are bought and sold, such as coal, oil, and natural gas
subsistence energy sources: Energy sources gathered by individuals for their own immediate needs including straw, sticks, and animal dung
energy demand changes reflect industrialization level
more automobiles → more gasoline and diesel fuels, more electric vehicles → more electricity
more factories + more house fridges = more electricity
Patterns of Energy in the United States
wood predominant until 1875, then coal and natural gas 1900s
1960 → nuclear and hydroelectricity joined
1970 less oil resurge coal
recent increase in natural gas consumption, decrease in coal bc fracking reduces cost + more availability
renewable energy passes coal
energy based on inputs and outputs of big system, bounded by tech and politics
oil domestic and foreign, hydroelectric from within
output is work - transportation, residential, commercial, industrial
u.s. energy consumption and energy mix
total energy use about 100 ej per year with fossil fuels dominating
renewables now exceed coal in the energy mix reflecting recent change
most energy is domestically produced with industry and transportation as top users
energy usage varies regionally and seasonally
midwest/southeast → coal for electricity
west/northeast → electricity from nuclear, natural gas, hydroelectric
expanding domestic production → changing energy portfolio
type of energy applied based on ease of transport and amount of energy it contains
Quantities of Fossil Fuels in the United States and Worldwide
finite and important → assessed worldwide reserves of fossil fuels via geologic formations
DOE EIA and IEA estimate reserved and project how much underground, potentially recoverable, and usage rate changes
estimated reserves + projected consumption rate → number of years remaining for fuel
does it even matter that we know it’s finite?
indomitable human spirit → develop new energy sources and reduce demand for current
leveled off rn, slightly decreasing per capita
energy intensity: The energy use per unit of gross domestic product (GDP).
steadily decreasing → using energy more efficiently per dollar of GDP
→ The Hubbert Curve
fossil fuel usage isn’t sustainable bc can’t limit consumption to rate of formation → debates on economy being limited
shift from availability to consequences of fossil fuel combustion: The chemical reaction between any fossil fuel and oxygen resulting in the production of carbon dioxide, water, and the release of energy
hubbert curve: A graph that represents oil use and projects both when world oil production will reach a maximum and when world oil will be depleted.
upper and lower estimate of total world petroleum reserves
total reserves didn’t hugely influence time to use all
peak oil: The point at which oil extraction and use would increase steadily until roughly half the supply had been used up
future of petroleum use and energy transition
petroleum use is limited to a relatively short historical window regardless of total reserves (200 years)
societies switch fuels when better options appear, not when resources run out
focus is shifting from peak oil to identifying and developing next energy sources
The Future of Fossil Fuel Use
fossil fuel availability and shift toward energy transition
conventional oil and gas may be depleted within decades while coal could last centuries
projections are changing due to technology advances renewables and altered use patterns
climate change concerns are pushing focus from reserves to transitioning energy sources
Lesson 35.4: What is the importance of energy efficiency and conservation?
true sustainability = energy conservation + energy efficiency
energy conservation: Methods for finding and implementing ways to use less energy
energy efficiency: The ratio of the amount of energy expended in the form you want to the total amount of energy that is introduced into the system
least expensive and most environmentally sound, sometimes easiest to implement
Different Forms of Energy
best energy form depends on what its used for
transportation → liquid compact, high energy to mass ratio, oil diesel; harder w coal, carry much more
energy in batteries increase and price decrease, electricity is becoming more ideal
wood and coal vehicle starts slowly, no quick acceleration
large amount of air pollution per joule of energy and much more refining
Quantifying Energy Efficiency
both the process to get the energy and the process of converting it to fuel for work
second law of thermodynamics → energy transforms, ability to work diminishes bc some lost as heat
66% of coal energy ends up as heat
lost during extraction, transporting, process, combustion, electricity generation, disposal, transmission
energy return on energy investment: The amount of energy we get out of an energy source for every unit of energy expended on its production.
energy obtained from fuel/energy invested to obtain fuel
Module 37: Distribution of Natural Energy Resources: Fossil Fuels
Leeson 37.1: Why are fossil fuels and ores found only in certain locations?
Fossil fuel and ore distribution around the globe depends on the geology of the region
coal forms from remains of trees and ferns
organic matter to make coal must undergo the circumstances to stop it from decomposing → buried quickly without air exposure
tropical locations, wetlands deltas
coal locations today depend where coal formation circumstances were found 300k years ago
crude oil formed from remains of ocean phytoplankton died millions years ago
did not decompose normally
deposits of phytoplankton found where porous sedimentary rocks r capped by nonporous rocks, so after millions of years, crude oil fills the porous spaces
fossilized organic matter that becomes oil becomes less dense → up to highest point of porous, blocked by nonporous
sometimes flows similarly to how water comes from artesian wall, but mostly, need drill well and refined via pipeline
natural gas in crude oil separates naturally and floats to top
oil workers burn off natural gas to capture it for fuel
transportation, petrochemicals, plastics, lubricants
Lesson 37.2: What are the advantages and disadvantages of fossil fuels including oil extraction and fracking?
Advantages of Coal
energy dense and abundant
provides heat for industrial processes and electricity generation
low economic costs and tech demand (surface)
easy transportation, easy use
Disadvantages of Coal
subsurface mining has several negative impacts
burning of several impurities on coal releases emissions like sulfur
combustion can release air pollutants, or stay in ash
wash coal w organic compounds
leaks can contaminate water and cause residual ash
large deposits of ash near coal power plants can leak and release a muddy flow of ash
lots of air pollution
Advantages of Oil
easy transportation and use
slightly more energy dense and cleaner, good for mobile combustion engines
produces less co2
Disadvantages of Oil
contains trace metals that when burned, release greenhouse emissions
risk of leaks when extracting and transporting, contaminating water and wells
most oil spills occur from land runoff, airplanes, boats,
lots of debates of habitat consequences and domestic oil extraction
habitat interference, railing accidents
explorers say arctic wildlife council will yield so much liters, opponents mark
local air pollution problems, health, human rights
Advantages of Natural Gas
powers half of homes in the US
fewer impurities, emits less particles during combustion
emits less co2 than coal
Disadvantages of Natural Gas
releases less co2 when burned compared to other fossil fuels but
methane leaks are highly effective greenhouse gases and contribute to climate change
extraction methods like fracking cause environmental concerns
→ Fracking
fracking: Short for hydraulic fracturing, a method of oil and gas extraction that uses high-pressure fluids to force open existing cracks in rocks deep underground
can extract more at a lower gas cost
reliance on domestic energy sources and jobs
initially appeared more beneficial
large amounts of water used, potential contamination
many chemicals are added to fracking
nearby drinking water wells show higher natural gas contamination
wastewater injection linked to increased earthquake activity
steps of fracking
water acquisition
chemical mixing
well injection
flowback and wastewater
wastewater treatment
volatile organic compounds: An organic compound that evaporates at typical atmospheric temperatures
methane leakage and climate change
natural gas escapes during fracking and extraction as fugitive emissions
leakage estimates are uncertain and range from 2 to 9 percent
methane’s high heat trapping ability makes leakage a serious climate concern
Lesson 37.3: How are fossil fuels used for electricity generation?
Fuel is converted to electricity and releases carbon dioxide and heat energy
coal is burned in a boiler
energy of combustion fuel is transferred to water to become steam
energy of steam transfers to turbine: A device that can be turned by water, steam, or wind to produce power such as electricity
shaft turns generator, generates electricity
distributed to other places, used for heat, light
after passing through turbine, condenses back to water → cooling tower or discharged in water body
as coal is burned, some of its energy is lost as heat
energy losses from plant to light bulb
transmission lines lose about 10 percent of electrical energy
incandescent bulbs convert only 5 percent of electricity into light
overall efficiency is found by multiplying each stage’s efficiency
calculating energy efficiency
coal to electricity x transport of electricity x light bulb efficiency = overall efficiency
Energy Quality
energy quality: The ease with which an energy source can be used to do work
high-quality = concentrated, convenient
energy quality of gasoline versus wood
gasoline has high energy concentration and easy transport
infrastructure allows efficient conversion to work and heat
wood has lower energy density and greater usage challenges
Module 38: Nuclear Power
nuclear power: Electricity generated from the nuclear energy contained in nuclear fuel
Lesson 38.1: How is nuclear energy used to generate electricity?
Nuclear reactors use fission to generate electricity
same basic process as electricity from fossil fuels
fuel releases heat, converts water to steam, turns turbine, turns generator for electricity
uranium-235 isotope is used as fuel
nuclei of atom stability based on mass of isotope and number of neutrons
radioactivity: The emission of ionizing radiation or particles caused by the spontaneous disintegration of atomic nuclei
transferred to surrounding environment, hotter, to create steam
fission: A nuclear reaction in which a neutron strikes a relatively large atomic nucleus, which then splits into two or more parts, releasing additional neutrons and energy in the form of heat
chain reaction fissions
2-3 million times more energy than coal
harness kinetic energy from additional neutrons to produce self-sustaining nuclear fission chain reaction
byproduct = radioactive waste for millions of years
fuel rods: A cylindrical tube that encloses nuclear fuel within a nuclear reactor.
uranium processed into pellets then put here
heat transferred to circulating loop of water, then follows same process for other electricity generators
harness heat from fission for steam, but slow fission to allow collisions to happen appropriately
moderator like water slows down neutrons
control rods: A cylindrical device inserted between the fuel rods in a nuclear reactor to absorb excess neutrons and slow or stop the fission reaction.
rods uncontrolled → meltdown
Concentrating the Uranium Ore
uranium mining and nuclear fuel preparation
900 kg of uranium ore must be mined and processed to produce 3 kg of nuclear fuel
uranium mining requires fossil fuel energy and generates mine tailings though less material is needed than coal for electricity
remove lots of host rock, extract and concentrate, leave remaining in tailing pilings
most uranium contains U238 (not ez fission) and not U235 → chemically enriched to increase concentration of U235 (>3% = suitable nuclear fuel_
Lesson 38.2: What are the advantages and disadvantages of nuclear power?
no air pollution during operation
achieve independence from imported oil
france, china, russia, south korea, canada, ukraine
1/5 electricity USA from nuclear
early proponents → too cheap to meter
new construction is now expensive bc public protests of nuclear accidents and waste, legal battles, delays
nuclear reactors and environmental considerations
number of us nuclear reactors has declined since 1990 but interest is increasing
overall emissions from nuclear power is 10% of coal even accounting for fuel and construction
main environmental concerns are radioactive waste disposal and accident risks
Lesson 38.3: What are radioactivity and radioactive waste?
Nuclear power depends on radioactivity but as a result, it generates radioactive waste
less fuel required to operate nuclear plant than coal plant
fuel stays radioactive even after not useful for electricity
radioactivity - emission of radiation or particles from spontaneous decay of atomic nuclei
Radioactive Isotopes Undergo Radioactive Decay
radioactive decay: When a parent radioactive isotope emits alpha or beta particles or gamma rays
half-life: The time it takes for one-half of an original radioactive parent atom to decay
determine length of how dangerous a radioactive element will be
Radioactive Waste: The By-Product of Electricity Generated from Nuclear Power
radioactive waste: Nuclear fuel that can no longer produce enough heat to be useful in a power plant but continues to emit radioactivity
high level - used rods → ionizing radiation to cause immediate human damage
low level - contaminated items during plant maintenance → damage human DNA, cancer, tumor, eyes, reproduction
mine tailings - residue after uranium mined and enriched
Measuring Half-Lives
radioactive half life and nuclear waste disposal
spent fuel rods lose efficiency as radioactive isotopes decay
u-235 has an extremely long half life (700 mil) causing slow reduction in radioactivity
long half lives make safe long term disposal of nuclear waste a major concern
becquerel (Bq): A measurement of the rate at which a sample of radioactive material decays; 1 Bq is equal to the decay of one atom per second
curie: A unit of measure for radiation, a curie is 37 billion decays per second
Radioactive Waste Disposal
store spend rods at plant itself, first in pools of water to act as a shield
run out of space → lead-lined dry containers, eventually moved to permanent waste disposal facility
radioactive waste disposal challenges
waste cannot be destroyed or dumped or in space without risking environmental contamination
long term storage on earth must prevent leakage protect groundwater and limit human exposure
sites must be secure remote and safe during storage and transport and prevent terrorists bombing it
→ Long-term Storage
yucca mountain seems like a nice place
protests and controversy, released report conforming soundness of its safety, later abandoned efforts to license waste repository
nuclear energy and sustainability debate
nuclear power releases low co2 emissions and none electricity generation
it creates large amounts of long lived high level radioactive waste → is it sustainable?
will there ever be a safe enough place?
Lesson 38.4: What are the three major nuclear accidents?
Three Mile Island, Chernobyl, and Fukushima are the three nuclear accidents
march 28 1979 Three Mile Island Pennsylvania
cooling water valve closed one day → lack of cooling → partial meltdown, reactor core severely damaged, contaminant structure became radioactive plus unknown radiation released
children and pregnant women evacuated
lots of anxiety, potential explosive gas bubble
nothing really happened, but still concerns and claims of mortality and cancer
april 26 1986 chernobyl
tried testing (not allowed) disconnecting emergency systems and removed control rods → reactions uncontrolled and overheated, explosion and fire, deaths from immediate impacts and burns
wind blew radiation across Europe, contaminated food, evacuations, thyroid cancer and deaths in children and adults from radioactive iodine
March 11, 2011, Fukushima
earthquake → tsunami → flooding damaged plant, hydrogen gas explosions, released radioactive gases, fires
thousands killed, evacuated
no deaths from radiation itself
Nuclear Power Compared with Other Fuels
future of nuclear energy in the united states
nuclear energy offers low emissions but raises concerns about accidents and waste containment
us nuclear construction has resumed slowly with a few plants approved, halted, or planned since 2006
future expansion is uncertain, with limited growth in the us and more rapid development in countries like china
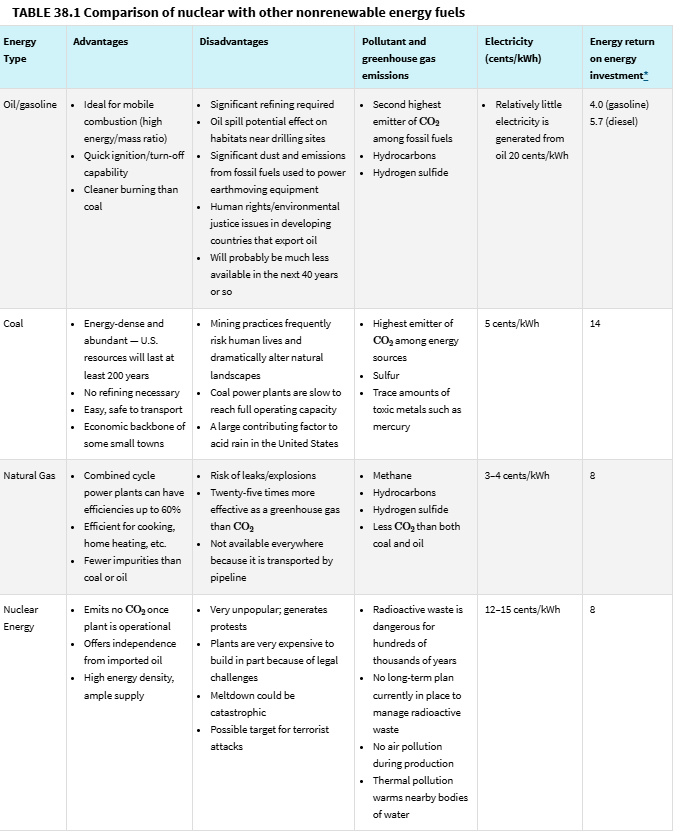
Module 40: Geothermal Energy and Hydrogen Fuel Cells
Lesson 40.1: How do humans harness geothermal energy?
geothermal energy: Heat energy that comes from the natural radioactive decay of elements deep within Earth
convection brings hot magma to Earth’s surface → groundwater becomes heated → drives to surface as geysers or hot springs, or drill it
clean renewable from US, Indonesia, Philippines, Turkey
little GW (15)
Harvesting Geothermal Energy
can be piped directly into household radiators to heat homes
heat exchangers collect heat by circulating cool liquid, heat flows in, goes to surface → Iceland
generate electricity, but steam comes from water evaporated by internal heat
heat from this is basically nondepletable during human time
groundwater can be depleted
long harvesting periods → unsustainable, so return it to the ground
25% of electricity in Iceland from geothermal energy
US → only 1%, but largest producer of geothermal electricity cs uses so much electricity, mostly west/southwest
less growth potential cs not easily accessible + expense of drilling
hazardous gases and steam can release → hydrogen sulfide, methane
Ground Source Heat Pumps
ground source heat pumps: A technology that transfers heat from the ground to a building
underground temp constant bc retains heat better than air → heat and cool residential and commercial buildings
technically solar instead of geothermal → nondeplatable
ground source heat pumps do not remove steam or hot water from ground
cycles fluid through pipes underground
winter → fluid absorbs heat
slightly warm fluid compressed to increase temperature → distributed through house → expands and cools, repeat again
summer → fluid is cooled underground and pulls heat from house
→ Hot Water Heat Pumps
hot water heat pumps and home energy efficiency
extract heat from air in a basement or garage and transfer it to domestic hot water (showers, dishwashing)
efficiency exceeds 100 percent because it adds heat from surrounding air to the electrical energy used
increasingly popular in the us with financial incentives, reducing fuel use, co2 emissions, and costs
Lesson 40.2: What is a hydrogen fuel cell and what is its potential for providing electricity?
Hydrogen fuel cells use hydrogen as an energy source and are almost pollution free
fuel cell: An electrical-chemical device that converts fuel, such as hydrogen, into an electrical current
similar to a battery - electricity generated by 2 chemical reactants in a closed container w no additions, used up, dies
cell → reactants continuously added, alternate to nonrenewable
2H2 + O2 → energy + 2H2O (NO POLLUTANTS!)
hydrogen molecules split into protons and electrons in upper reaction layer
protons across membrane
electrons go another way to form an electric current
oxygen split and combine with protons and electrons to form water lower reaction layer
constant supply of hydrogen required, but free hydrogen is rare and unsafe
separate it from water or gases → energy-intensive process that releases co2
electrolysis: The application of an electric current to water molecules to split them into hydrogen and oxygen.
no fossil fuels or emissions
hydrogen acts as an energy carrier
wind and sun energy sources can’t produce electricity constantly, but the electricity produced can be used to generate hydrogen → stored
generate electricity for electrolysis using clean nondepletable energy source → hydrogen could be sustainable energy carrier
when warm, use sun, when cold, use stored hydrogen via powerplant
The Viability of Hydrogen
hydrogen fuel cells and energy potential
convert hydrogen and oxygen into electricity with about 80 percent efficiency and water as the only by-product
currently limited use in the us with 160 fuel cells at 100+ power plants, less than 0.2 percent of electricity generation
potential applications in vehicles, offering zero co2 and pollutant emissions, but widespread adoption faces skepticism
diadvantages
obtain hydrogen without expending more fossil fuels than we save → renewable source
distribution network to safely deliver to consumers, stored as liquid or gas
full cell vehicle → large gas tank with high pressure → tank rupture risk
sustainable bc uses electric motor just like electric vehicles
more efficient than internal combustion engines
hydrogen from natural gas → total amount used to move electric vehicle will be less than gasoline, even lower if hydrogen from renewable sources
hydrogen fuel cell vehicles and geothermal energy
toyota offers a hydrogen fuel cell vehicle, but fueling stations in the us are limited to about 40, mostly in california
both hydrogen fuel cells and geothermal energy are currently small contributors to total energy use
both have potential to expand in the us energy mix with minimal co2 and air pollutant emissions
Unit 7: Atmospheric Pollution
Module 43: Photochemical Smog, Thermal Inversions, Atmospheric CO2 , and Particulates
Lesson 43.1: What is photochemical smog and how can we reduce it?
Photochemical smog is a complex combination of compounds and it can be reduced by decreasing emissions of its precursors
4 in 10 americans today live in counties w bad air quality
sulfur, nitrogen, co have been reduced since the Clean Air Act, but controlling secondary pollutants like smog and ozone is hard
daytime increase in ozone bc of smog formation and atmospheric behavior when weather changes
In most cities, mid- to late-afternoon ozone concentrations are higher than the daily average as a result of an increase in nitrogen oxides in the early morning hours
Chemistry of Ozone and Photochemical Smog Formation
smog is combo of oxidants and particulate matter, reducing sunlight and increasing haziness; 2 categories
photochemical smog: Smog that is dominated by oxidants such as ozone. Also known as Los Angeles–type smog; brown smog
sulfurous smog: Smog dominated by sulfur sulfur dioxide, sulfate compounds, and particulate matter. Also known as London-type smog; gray smog; industrial smog
economic harm since poor visibility = less tourism revenue and income for hotels and restaurants
complex problem still unsolved bc number of pollutants → based on intensity/duration of sunlight, water, and VOCs
chemical process of photochemical smog
during the day, UV warms the atmosphere
lots of NOx (little VOCs) → splits into NO2 and free oxygen O
energy from sun makes O combine with O2 into ozone O3
so as nitrogen dioxide increases, ozone accumulates → highest early/mid afternoon during summer
even when sunlight intensity decreases, nitrogen oxide is still present, and the ozone combines with NO to make O2 and NO2
ozone destruction natural at night, but some compounds can interfere with this
VOC chemicals easily released into air, either evaporate or sublimate
evaporate:The process of converting from liquid to a gas or vapor
sublimate: The process of converting from a solid to a gas or vapor
can come from tree fires or gasoline or formaldehyde: A naturally occurring compound that is used as a preservative and as an adhesive in plywood and carpeting
if large amounts of VOCs are present, they combine with nitrogen oxide to a stronger bond and can’t combine with ozone
ozone isn’t broken down → larger accumulations in urban areas and rush hour
→ The Importance of Temperature
emissions of VOCs and evaporation of liquid VOCs increase as temperature increases
NO emission too bc more cooling
→ Smog and Human Health
harms human health a lot
itchy eyes, irritates respiratory system, risk of infection bc inflamed
aggravates asthma emphysema bronchitis
decreased lung capacity, more admissions for asthma
apps tell u air quality, but poor people dont have a choice
Lesson 43.2: What are thermal inversions and how do they relate to air pollution?
Thermal inversions trap air pollutants close to Earth’s surface
normally, as altitude decreases, temperature increases
thermal inversion: An atmospheric condition in which a relatively warm layer of air at mid-altitude covers a layer of cold, dense air below
inversion layer: The layer of warm air that traps emissions in a thermal inversion
esp in cities bc vehicle exhaust and industrial emissions
lead to problems resulting from other pollutions
a temperature inversion trapped carbon monoxide and particulate matter from coal-burning stoves after a cold spell, where the city shut down municipal heating and made houses burn their own coal
thousand people suffered carbon monoxide poisoning or respiratory illness and few people died
thermal inversions are natural, investigating whether climate change is increasing their frequency and worsening both anthropogenic and natural pollution effects
Lesson 43.3: What are the natural sources of particulates and CO2?
Volcanoes, forest fires, respiration, and decomposition are natural processes responsible for particulate emissions and carbon dioxide
volcanoes, lightning, fire, and plants also release pollutant compounds, not just human activity
lightning → nitrogen oxide, forest fires → PM, NOx, COx, fragrant terpene VOCs from fires
natural VOCs created smog in mountain ranges
Natural Sources of Particulates
large agricultural fields can release PM when plowed, like Dust Bowl
SOx 30% natural, NOX 40% natural, VOCS 89% natural
places like NA make anthropogenic contribution bigger
effects depends on wind direction
westerlies distributes PM and SOx from st helen explosion, lowering rain pH
PM comes from volcanoes forest fires dust storms, ranging from 0.01 to 100 micrometers
>10 micrometers is nose filtered
PM10: Particles smaller than 10 μm are called Particulate Matter-10 and are not filtered out by the nose and throat and can be deposited deep within the respiratory tract
PM2.5: Particles of size 2.5 μm and smaller can travel further within respiratory tract and are of even greater health concern
PM scatters and absorbs sunlight
particulate matter and sunlight reduction
high concentrations after forest fires or volcanic eruptions can reduce solar radiation enough to limit photosynthesis
haze results from light scattering by particulate matter, with ozone and photochemical oxidants also contributing indirectly to haze formation
Natural Sources of Carbon Dioxide
natural sources of carbon dioxide in biological and chemical processes
aerobic and anaerobic respiration, decomposition, and combustion, all of which convert biomass or sugars into energy, water, and carbon dioxide
decomposition with or without oxygen and the combustion of biomass in fires are major natural global sources of carbon dioxide
anaerobic decomposition: sugar → carbon dioxide + methane + less energy
Module 44: Indoor Air Pollutants
Lesson 44.1: What are the major indoor air pollutants and where do they come from?
indoor air pollutants: Compounds that adversely affect the quality of air in buildings and structures
indoor air pollutants are compounds that reduce air quality in buildings and affect occupant health and comfort
some pollutants, like carbon monoxide and particulates, are shared with outdoor air, while others, such as radon and asbestos, are primarily indoor concerns
sources include combustion inside buildings, off-gassing from building materials, and the transfer of outdoor pollutants into indoor spaces
Carbon Monoxide
carbon monoxide as an indoor air pollutant
extremely dangerous indoors, binding to hemoglobin more effectively than oxygen and causing oxygen deprivation, headaches, dizziness, nausea, loss of consciousness, or death
sources and risks in developed countries
malfunctioning natural gas heaters or furnace exhaust systems release colorless, odorless carbon monoxide into living spaces
lack of detection can allow dangerous accumulation, especially while occupants are asleep, making carbon monoxide detectors essential
sources and risks in developing countries
indoor carbon monoxide exposure arises from burning biomass in open-pit fires with little or no ventilation
poor combustion and absence of exhaust systems make CO and particulate matter major hazards, with children at higher risk due to faster respiration rates
Particulates
→ Smoke
indoor particulate matter and smoke
smoke and soot from fires inside or outside buildings, as well as tobacco smoke, are major sources of indoor particulate matter
prolonged exposure can cause respiratory infections, bronchitis, pneumonia, and cancer
second-hand smoke exposes non-smokers to high concentrations of particulate matter, increasing health risks
→ Dust and Mold
dust and mold as natural indoor pollutants
household dust contains particulate matter, pollen, bacteria, and dust mite waste, fueled by millions of shed human skin cells each day
dust mites live in bedding, pillows, flooring, and carpets, and their body fragments and droppings contribute to asthma and allergic reactions
mold grows on damp organic matter indoors, often hidden behind walls, and exposure can trigger allergies, lung inflammation, and asthma
Asbestos
ASBESTOS: A long, thin, fibrous silicate mineral with insulating properties, which can cause cancer when inhaled
used historically for insulation and building materials, all harmful when inhaled
respiratory diseases such as asbestosis and mesothelioma, especially among miners and workers
safe when undisturbed, but old or damaged materials release fine fibers into the air, requiring careful removal by qualified abatement personnel
Radon
radon-222: radioactive gas that occurs naturally from the decay of uranium and is an indoor air pollutant
can enter buildings through foundation cracks or contaminated groundwater
decays into polonium-210, which can attach to dust and be inhaled, increasing the risk of lung cancer
second leading cause of lung cancer, and recommends testing, ventilation, mitigation systems, and sealing cracks to reduce indoor exposure
Volatile Organic Compounds
building materials, furniture, glues, paints release (VOCs), with formaldehyde being one of the most toxic → homes and wood
irritation, asthma, cancer
detergents, dry-cleaning fluids, deodorizers, plastics, fabrics, paints, synthetic carpets, and outdoor emissions from nearby chemical or waste facilities
Lead
primarily comes from old lead-based paint, last used in the u.s. in the 1960s by demolishing these buildings
can come from outside soil or air, but ingestion of paint chips remains the greatest exposure risk for young children
Other Pollutants
NOX and SOX, common outdoor pollutants, can also accumulate indoor via poorly insulated or loosely sealed buildings
NOX from indoor combustion sources like cooking, while proximity to outdoor pollution sources strongly influences indoor air quality
Lesson 44.2: How does indoor air pollution differ in the developing and developed world?
worldwide health hazard, ~4 million deaths yearly → pneumonia, heart disease, pulmonary disease mostly from low/middle income countries
Developing Countries
indoor air pollution from indoor combustion in developing countries
burning wood, animal manure, or coal indoors for heat and cooking produces high concentrations of particulate matter near occupants, super high PM
poor building insulation and ventilation may allow some pollutants to escape, but if outdoor air quality is also poor, ventilation provides only limited improvement
Developed Countries
indoor air quality and sick building syndrome in developed countries
modern lifestyles and building practices have increased the importance of indoor air quality, as people spend more time indoors and buildings are more tightly sealed for energy efficiency, trapping pollutants inside
widespread use of materials containing VOCs, formaldehyde, plastics, and petroleum-based products leads to off-gassing of chemicals, contributing to high indoor pollutant levels
sick building syndrome: A buildup of toxic pollutants in weatherized spaces, such as newer buildings in the developed world
when ventilation is inadequate and contaminants accumulate, causing headaches, nausea, eye/throat irritation, and fatigue, with causes including indoor sources (glues, furniture, cleaning agents), outdoor pollution, and molds or pollen
Unit 8: Aquatic and Terrestrial Pollution
Module 47: Sources of Pollution, Human Impacts on Ecosystems, and Endocrine Disruptors
Endocrine Disruptors
endocrine disruptors: A chemical that interferes with the normal functioning of hormones in an animal’s body
low concentrations move through blood stream and “bind” to specific cells → causes them to produce a response relating to body function: growth, metabolism, reproductive organ development
endocrine disruptors bind/mimic receptive cells and cause responses
wastewater: The water produced by livestock operations and human activities, including human sewage from toilets and gray water from bathing and washing of clothes and dishes
exposure to hormones → male organisms become feminizied - low sperm, male and female organs, less production of estrogen to testosterone
Chesapeake Bay → estrogen causes 23% largemouth and 80% smallmouth fish to have both male female organs
grow eggs in male organs
pesticides can mimic animal hormones → disrupt endocrine system
worries about humans → developmental disorders, low sperm, increased breast cancer risk
Module 48: Human Impacts on Wetlands and Mangroves, Eutrophication, and Thermal Pollution
Lesson 48.1: What are the human impacts on water availability?
humans have creatively moved water by channeling flow of flood water, block river flow to store water, transport it far away, extract fresh water from salt water
Levees and Dikes
before human intervention, periodically overflowed banks → nutrient rich water made fertile floodplains
now, we want to prevent flooding for commercial/residential use
levee: An enlarged bank built up on each side of a river
largest system in Mississippi
levees have many problems
1) reduce fertility of floodpains bc no sediment deposition
2) sediments further downstream and settle where river meets ocean → delta
3) force floodwater downstrean to cause worse flooding
4) levees means development in floodplains, but these areas still flood → human and economic risk
high floodwaters → levees collapse from pressure or erode hole in levee → flooding
levees couldn’t handle Hurricane Katrina, super expensive and human death
dikes: Structure built to prevent ocean waters from flooding adjacent land
Northern Europe → lots of farmland below sea level esp Netherlands
pumps move intruding water back to ocean → ppl can farm and live in areas previously uninhabitable
originally with windmills, now electricity and diesal
Dams
dam: barrier that runs across a river or stream to control the flow of water
reservoir: The water body created by damming a river or stream
human consumption, generate electricity, flood control, recreation
largest in Missouri River
turn waterwheels at grain mills or modern turbines for hydroelectric plants (little percent)
built for recreation, flood control, hydroelectricity, or aesthetics
example: Three Gorges Dam, Yangtze River
good: generates large amount of hydroelectric power
reduces fossil fuel usage
prevent deadly seasonal floodings
bad: human and environmental costs
large amount of energy and materials
displacement, flooding, forced relocation
disrupted free flowing rivers, only free flow in remote areas
interruption of natural water flow organisms have adapted to
migrating fish like salmon can’t breed → bears can’t eat them
fish ladders: A stair-like structure that allows migrating fish to get around a dam
alleviates and allows them to migrate
kills many fish when water passes through turbines
seasonal flooding scours pools and shorelines
favors colonization of certain plants/animals
people try to experiment and simulate seasonal flooding, promising
dams can be removed when no longer necessary
Klamath river used to be very successful for salmon migration
now, damned for electricity, irrigated for farming → less salmon, fishermen and natives suffered
hydroelectric company removed the four dams
farmers conserved water
better river flow → salmon population recovers
largest dam removal in history
with dam removal, water flow and ecology restores
dramatic increase in plant species along riverbanks
Aqueducts
canals, ditches, or pipes used to carry water from one place to another
ancient r made of limestone, now made with concrete canals and pressurized steel pipes → efficient water carriers
older aqueducts lose 55% from evapotranspiration, not good in Levant
NYC and LA rely on aqueducts for their daily needs
Colorado River aqueduct and Catskill Aqueduct
costs and benefits
clean supply of water
expensive construction, disturbs natural habitats
drying rivers
water diversion projects can have international impacts
india made large scale project to divert a lot of water from north, but worries about reducing water flow in bangladesh
reduce fresh water in estuaries → salty water further north
Chinese super dam
primarily for hydroelectricity, worried about using it for agriculture n taking from bangladesh and india
most infamous water diversion was soviet union aral sea
less freshwater, more saltwater, split into north and south
without moderating effect of water, more extreme conditions
world bank funded kazakhstan to stop north from flowing into south aral
depth increased, fishing increased, economic boom
south aral sea drying too expensive, dried completely
Humans are converting saltwater into freshwater by desalination
desalination/desalinization: The process for obtaining fresh water by removing the salt from salt water
used in waterpoor countries like Middle East (50% of world’s desalinated water)
slowly reduced costs bc of tech
ocean or salty lakes
two common techniques
1) distillation: A process of desalination in which water is boiled and the resulting steam is captured and condensed to yield pure water
lots of energy to boil and condense water → financially and environmentally expensive
2) reverse osmosis: A process of desalination in which water is forced through a thin semipermeable membrane at high pressure
new tech → efficient, less expensive
remaining liquid is brine → high salt concentration, cant be deposited in ground or coastal area bc impacts wildlife → dump in middle of ocean
all water management systems require large investment to build maintain repair
1.2 billion people live in water scarcity areas
Lesson 48.2: What are the human impacts on wetlands and mangroves?
human activities impact waterbodies, visible in wetlands
wetlands → standing water, water-saturated soil during some part of the year
hydric soils
many ecosystem services
purify water by removing pollutants and nutrients
absorb floodwaters and buffer shorelines
habitat for migrating birds and other organisms
protection for young fish
major carbon sinks
historically, drained for irrigation and to reduce diseased mosquito populations
wetlands converted to development of homes and businesses → pollution
others into lakes for flood control or recreation
commercial fishing declines fish population → alters food webs by affecting dispersal of seeds
huge loss of wetlands
hurricanes, rising sea levels, human development
disappearing faster than forests
less water purification and flood buffering
Lesson 48.3: What are the causes and consequences of eutrophication and sediments?
eutrophication: Excess nutrients from human activities that make their way into waterbodies; it causes nutrient pollution that alters food webs and harms water quality
Algal Blooms
phosphorus and nitrogen enter water and can cause algal blooms/red tides → water becomes red
bad treated sewage, fertilizer runoff, storm runoff
rapid increase in abundance of algae in water bc of super high water fertility
bad for swimming, recreation, some species produce toxins that harm organisms
populations decompose and die
bc of low concentrations of oxygen → hypoxic water makes species migrate or die
5 → 87 deadzones
increased concern of freshwater algal blooms
oligotrophic → low nutrient, clear water, high oxygen, less harmful algal blooms
eutrophic experience natural blooms → inputs can make them more hypoxic and harmful
Oxygen Sag Curves
similar oxygen concentration dip in sewage or other organic pollutants that quickly decay
measure O2 concentration in water in diff places to see if there’s a point source input
measure water near sewage pipe because bacteria consume the oxygen
downstream → less sewage → oxygen normal
oxygen sag curve: The relationship of oxygen concentrations to the distance from a point source of decomposing sewage or other pollutants
Sediments
some inputs of nutrients = sediments eroding from landscapes and traveling in water
ex: Mississippi River and other fast-moving streams dump water into lakes
sediment accumulation → harder for boats to navigate bc shallow water
dredging → dig up sediments to let boats move
impacts on organisms
reduce sunlight penetration in water → stunt growth or die
harder for visual predators to find prey + clog gills = less oxygen
bury organisms that r stationary at the bottom
Lesson 48.4: What are the sources of thermal and noise pollution?
thermal pollution: Occurs when humans cause a substantial change in the temperature of a water body
noise pollution → elevated sounds that harm organisms
Thermal Pollution
water bodies near logged forests → more sunlight
outside range of tolerance + less dissolved oxygen for organisms
industry removes cold water to absorb heat for cooling processes and returns warm water
thermal shock: A dramatic change in temperature that can kill many species
warm water = less dissolved O2 = suffocation
steps to reduce include pump heated water into outdoor holding pools where it can cool before being returned
EPA regulates how much heated water returned to natural bodies
hard to comply esp during summer → high demand for electricity = high demand for cooling water despite low river depth and high temp
solution: cooling towers use evaporation to reduce temperatures (like for nuclear energy); built closed systems that cool hot water in a tower and recycle it → neither extract nor release
Noise Pollution
noise is a type of water pollution
sounds from ships and submarines hinder animal communication
loud sonar negatively affects low-frequency long distance communication animals like whales
concerns raised by naval operations
navy conducts training like ship sonar, torpedoes, detonating bombs underwater
negligible effect on fish and whales according to NOAA
over years, though, estimate 30% of marine wildlife will be negatively affected → ships with quiet propellers
Module 49: Persistent Organic Pollutants (POPs), Bioaccumulation, and Biomagnification
Lesson 49.1: How do persistent organic pollutants affect ecosystems?
Chemical Persistence
persistence: The length of time a chemical remains in the environment
based on pH, temp, etc., often measured by half life
persistent organic chemical pollutants (POPs): Synthetic, carbon-based molecules that break down very slowly in the environment
can travel long distance via water and wind, or stored in fat tissues
harmful chemicals even worse if they r persistent → modern chemicals are short-lived
Persistent Organic Chemicals of High Concern
PCBs are lethal, carcinogenic, and highly persistent; though banned, still persist
we can reverse it though (Hudson River and General Electric)
PFAs ruin immune and reproductive systems → “forever chemicals” 1k years
some towns only bad groundwater → install water filter systems in every home or installing public water lines for every home
Lesson 49.2: How do routes of exposure and solubility determine the concentration of chemicals that organisms experience?
to understand effects of chemical concentrations that organisms experience, we must understand the following
Routes of Exposure
routes of exposure: The way in which an individual might come into contact with an environmental hazard, such as a chemical
complex - consumption, water, air, etc.'
some can be tracked, like for a baby - bisphenol A from toys, food containers, bottles
also important for animals
pesticides from ingestion or droplets or gills
some animals r permeable and exposed to pesticides
Solubility of Chemicals
solubility: How well a chemical dissolves in a liquid
herbicides in water, insesticides in fats/oils
highly soluble in water → washed off as run off
soluble in fats → no runoff, bound to soils under water
Lesson 49.3: How can bioaccumulation and biomagnification increase the concentrations of chemicals in organisms?
Bioaccumulation
bioaccumulation: The selective absorption and concentration of a chemical within an organism over time
chemicals soluble in fats accumulate in fatty tissues
accumulation of mercury is why govt recommends to not eat meals of tuna more than once a month
pesticides after WW2 bioaccumulate
modern ones are designed to not be in tissue
rate of accumulation = concentration, rate of intake, rate of chemical breakdown, rate of excretion
Biomagnification
biomagnification: The increase in chemical concentration in animal tissues as the chemical moves up the food chain
best example: insectide DDT seemingly only killed pests without any impact on humans
however, not water soluble → binds to particulates like algae, then zooplankton, becomes even more concentrated
biomagnification causes birds to make thin shelled eggs
DDT still used in africa for high malaria concerns
similar effects in humans
advise mercury PCB POPs consumption
cancer, reproduction, nervous issues
development abnormalities, long-term learning and motor skill issues
Module 51: Waste Reduction Methods
Lesson 51.1: What are the three Rs that divert materials from the waste stream?
reduce, reuse, recycle: A popular phrase promoting the idea of diverting materials from the waste stream. Also known as the three Rs
most environmentally beneficial to least, most energy to least energy use
Reduce
most energy efficient → optimal way to start decline in solid waste generation
less material → less to discard
source reduction: An approach to waste management that seeks to cut waste by reducing the use of potential waste materials in the early stages of design and manufacture
increases energy efficiency bc less waste, less disposal processes, less resources = economic benefit
applied by individuals, companies, institutions
electronic copies > printing
manufacturing companies can do source reduction in many ways
new packaging, same protection, less material
decreased CD wrapping bc using online music is an example of source reduction for companies, downloading online music is for individuals
source reduction by substituting materials
ex: switching from paper cups to personal reusable mugs reduces MSW, despite requiring energy to clean and use water
break-even point for plastic is lower than ceramic cs less energy used
switched from oil-based paint with toxic lead to nontoxic paint
reduce material use, solid waste, without spending extra materials or energy
Reuse
reuse: Using a product or material that would otherwise be discarded
used longer before wasted; ideally, no additional energy/resources for reusing
others may involve repair - costs time, labor, energy, materials, but less waste than discarding
energy required sometimes to transport for reuse
ex: milk/soda bottles shipped to factory - washed, sterilized, refilled
super common esp in USA
reuse in other ways, like newspapers for bedding/art
universities w surplus equipments donate items
thrifting, flee markets
expenditure of energy and generates other waste - like washing milk bottles creates wastewater
Recycle
recycling: The process by which materials destined to become municipal solid waste (MSW) are collected and converted into raw materials that are then used to produce new objects (two categories)
closed-loop recycling: Recycling a product into the same product
ex: aluminum cans collected, melted, made into new cans
prevents waste indefinitely
open-loop recycling: Recycling one product into a different product
soda bottle → jacket; prevents from going into landfill, but doesn’t reduce demand for raw material (petroleum) for new bottles
not new, but since 2000 it’s been really encouraged and embraced by individuals and cities bc belief in environmental quality
both waste and recycling rates increased, sharp increase since 1985
1/3 MSW today recycled
56% rate in germany
helpful cs extracting resources takes more time energy labor and money and generates pollution
zero-sort recycling programs → mix all recyclables in one container that they deposit on the curb or transfer
saves time for residents + more likely to recycle
at the sorting facility, workers organize materials by highest demand → best economic return
volatile glass/paper markets → recycle when financially worthwhile
always demand for copper and aluminum bc less expensive than extracting
easier to make new bottles from petroleum than using existing plastic bottles
degraded plastic from recycling, not degraded metal from recycling
time, processing, cleaning, transport, modification → most energy used
high recycling costs can influence cities to suspend glass recycling, which was a major shift from recycling as much as possible
sorting glass and plastics from recyclables was not cost effective
much recycled material shipped to China for production into new products
2018 - china stopped accepting recycled paper and plastic products, so smaller market for purchasing recycled items → companies charge more for picking up recycled items
rising costs → cities reduce or end recycling programs → more MSW in landfills
Lesson 51.2: How does composting reduce materials entering the waste stream?
fourth approach to 3 Rs focused on organic material like food and yard waste
these can break down anaerobically → methane gas more potent than carbon dioxide
avoid landfills by composting: The breakdown of organic materials into organic matter (humus)
vegetables, cornstalks, grass, manure, leaf litter, paper fiber are great
no meat and dairy - don’t decompose well, smell, and attract other animals
simple pile of food/waste in a yard or sophisticated boxes and drums rotated for mixing and aeration
30:1 carbon to nitrogen ratio best for microbial activity
layer “brown material” like leaves or dried cut grass with wet kitchen vegetables/green material
frequent turning ensures aerobic process and maintains proper moisture; nice odor of fresh compost within 2-3 months
humus enhances soil structure, cation exchange capacity, and land fertility
large-composting facilities may exist in cities to divert organic material from waste streams
waste is dumped and separated into compostable and non compostable material (delivered to landfill)
compostable material is aerated and turned at least once for a period of 30-365 days using rotating blades or tractors - high respiration kills any pathogenic bacteria; turn more to reduce heat
composed material cures and when finished, transported for use
outdoors is convenient, but can be done inside in a room, kitchen, or basement
compost kitchen waste w red wriggler worms → small recycling bin and if properly maintained, no odors
takes time and space
separating materials is difficult and storing materials before adding to pile attracts organisms
pile itself attracts unwanted animals
high organic matter → high cation exchange capacity and nutrients → soil quality
Lesson 51.3: How do life-cycle analysis and integrated waste management reduce municipal solid waste?
how do we decide what waste management techniques r the best? use life cycle analysis and a holistic approach
Life-Cycle Analysis
life-cycle analysis: A systems tool that looks at the materials used and released throughout the lifetime of a product—from the procurement of raw materials through their manufacture, use, and disposal. Also known as cradle-to-grave analysis
in theory, helps community determine if incinerator or landfill is better, but has limitations; difficult to determine overall environmental impact
is the production of particulates and NOx worse than production of methane?
is SO2 from paper cup or VOC from plastic cp worse?
city begins analysis from point of receiving solid waste to final point of disposal
economically, compare costs of disposal methods
better to receive 5 dollars for a ton of glass from a bottle manufacturing plant than paying $50 fee for landfill deposit
also consider low cost of nearby landfill versus further glass plant
always some cost to waste disposal
some covered by taxes, others individuals pay
disposing recyclables (lower tipping fee or revenue) usually cheaper than landfills bc always tipping fee, but can depend on fluctuating prices of recyclables
energy use wise, considers energy content of gasoline/diesel and pollution in trucking material
monetary, energy, pollution savings if new glass made from old glass vs new materials
super debatable, so now people take holistic approach
Integrated Waste Management
integrated waste management: An approach to waste disposal that employs several waste reduction, management, and disposal strategies in order to reduce the environmental impact of MSW
source reduction + any combo of three Rs, composting, landfills, etc
start with source reduction during manufacturing and identify behaviors for less waste and desired outcome (no forcing)
pay for incinerator - u need lots of people to use it to make up for the cost, detracting incentive for recycling/landfill
free to consider all options - efficient, cost-effective, least harmful
McDonough’s Cradle to Cradle favors minimizing waste generation before, during, and after manufacturing
some industries design future recycling into their products
ex: automobile manufacturers build cars they can be easily taken apart w different materials of composition ez to separate for recycling
design carpets so when they wear out, it’s easy to recycle for new carpeting
turtles create shells without making toxic waste - observe turtles to make production with less toxic materials
upcycle something of lesser value to something w more value
Module 52: Sewage Treatment
Lesson 52.1: What three major problems are caused by wastewater pollution?
three major reasons for wastewater pollutant concerns
decomposition by bacteria created large oxygen demand in water
nutrients released from decomposition can make land fertile
carries lots of pathogens
Oxygen Demand
as waste increases, microbes increase, increased oxygen demand
biochemical oxygen demand: The amount of oxygen a quantity of water uses over a period of time at specific temperatures
measure initial dissolved O2, incubate 5 days 20 C and measure second dissolved O2 → 2nd - 1st = amount of consumption done
low BOD → less pollution from wastewater
large microbial demand → less oxygen for other organisms
positive feedback as more fish die and get decomposed
Nutrient Release
cultural eutrophication: An increase in fertility in a body of water, the result of anthropogenic inputs of nutrients
wastewater contains phosphorus and nitrogen nutrients
algal blooms → rapid growth of algae
dies, microbes digest dead algae, consuming bunch of dissolved oxygen → dead zones
most caused by human activities
Disease-Causing Organisms
worldwide, drinking water source same as bathing, washing, sewage → pathogens via contact or ingestion
major waterborne diseases - cholera, typhoid fever, hepatitis, dysentery
hepatitis in USA from lack of restaurant sanitation
¼ population has insufficient supplies for access to drinking water
2.3 billion don’t have proper sanitation, half in India and China
Monitoring for Wastewater Contamination
fecal coliform bacteria: A group of microorganisms that live in the intestines of humans, other mammals, and birds that serve as an indicator species for potentially harmful microorganisms associated with contaminated sewage
public water supplies routinely tested for e coli
acceptable e coli amount for swimming higher than drinking (0)
beaches thus can close from animal feces
Lesson 52.2: How do we treat wastewater to prevent pollution?
all solutions follow same basic approach
bacteria used to break organic matter into CO2 and inorganic compounds → N, P
harmful pathogens outcompeted by nonharmful
Septic Systems
septic system: A relatively small and simple sewage treatment system, made up of a septic tank and a leach field, often used for homes in rural areas
septic tank: A large container that receives wastewater from a house as part of a septic system and buried underground adjacent to the house
wastewater enters one end, leaves another
three layers form
top: scum
middle: septage: A layer of fairly clear water found in the middle of a septic tank; can contain lots of bacteria and N/P
bottom: sludge: Solid waste material from wastewater
leach field: A component of a septic system, made up of underground pipes laid out below the surface of the ground
small holes → septage seeps out of pipe, slowly absorbed and filtered by surrounding soil
harmful bacteria outcompeted by nonharmful bacteria or get degraded, organic matter break into CO2 and inorganic nutrients
soil filtered water taken by plants or aquifer
no electricity needed to run system bc they rely on natural gravity to make water flow downhill to the tank
sludge must be pumped every 5 yrs and taken to a plant
Sewage Treatment Plants
septic systems not feasible for urban areas w little open land
developed → use centralized sewage plans from multiple houses via underground pipes
wastewater gets primary and secondary treatment
primary treatment involves physical removal of large objects by filtering through grates → settles sludge layer
secondary treatment → bacteria breaks down 90% of organic matter in remaining wastewater, converts it to CO2 and inorganic nutrients
aerates the water and adds oxygen → more aerobic bacteria, less bad smell than anaerobic
treated water settles, settled particles added to sludge
before sludge taken away from plant, exposed to bacteria so it’s lighter n less disease
landfill, burned, or fertilizer pellets
very effective at breaking down into CO2 and nutrients
bad effects on waterways they r released into
algal blooms from nutrients
tertiary treatments - physical, chemical treatments to remove inorganic compounds (nutrients, metals) and pathogens often via precipitating or mesh filters or disinfection
Legal Sewage Dumping
even in developed countries, raw sewage can directly be dumped into water bodies
treatment plants handle local house and industry wastewater
older plants get water from stormwater drainage systems; when it rains, it overwhelms the capacity → pump water into adjacent body
concentrated in older cities in northeast and midwest
contaminates water, beaches, fishes/shellfish → human illness
straightforward and expensive answer: modernize systems to prevent influx of stormwater
expensive long tunnels are dug for such things
Animal Feed Lots and Manure Lagoons
small scale → animal manure contaminates local water sources w hormones n antibiotics
farms w lots of animals use manure lagoons
after bacteria break down manure w the same processes, manure spread onto farm as fertilizer
leak in lining risk → groundwater/soil contamination
overflow into nearby water risk → disease in organisms
if used as a fertilizer, runoff
Module 53: Lethal Dose 50% (LD50) and Dose-Response Curves
Lesson 53.1: How are dose-response curves used to estimate lethal doses of chemicals?
three techniques to determine harmful concentrations
Dose-Response Studies
dose-response studies: A study that exposes animals or plants to different amounts of a chemical and then looks for a variety of possible responses, including mortality or changes in behavior or reproduction; 1-4 days for efficiency
acute studies: An experiment that exposes organisms to an environmental hazard for a short duration
chronic studies: An experiment that exposes organisms to an environmental hazard for a long duration
most commonly measure mortality responses; graphs follow an S-curve typically
threshold - dose at which an effect can be detected
LD50: The lethal dose of a chemical that kills 50 percent of the individuals in a dose-response study
estimate by draw horizontal line from 50% y-axis until it intersects the curve
assesses relative toxicity of a chemical for a certain species, like comparing LD50 of new chemical to older chemicals
chronic studies done from birth to maturity
sometimes, scientists measuring in other stuff like teratogens, carcinogens, neurotoxins
sublethal effects: The effect of an environmental hazard that does not kill an organism but which may impair an organism’s behavior, physiology, or reproduction
ED50: The effective dose of a chemical that causes 50 percent of the individuals in a dose-response study to display a harmful, but nonlethal, effect
no-observed effect level (NOEL): The highest concentration of a chemical that causes no lethal or sublethal effects
LD50 Studies
Toxic Substances Control Act of 1976 allowed EPA to regulate chemicals, but not food cosmetics, and pesticides (separate law for pesticides 1996)
won’t cause unreasonable bad effects
scientists developed a method of testing the most sensitive bird, mammals, fish, and invertebrates
regulations to protect the most sensitive apply to the rest → extrapolate results from mice and rats, representing all mammals
pigeons/quails for birds, trout for all fish, water fleas for all invertebrates
no separate standards for reptiles/amphibian bc nobody cared
aquatic → fish, terrestrial → bird
LD50 and ED50 levels determine concentrations in the environment that do no harm
usually, LD50/10 is a safe concentration
regulatory agencies r conservative in setting concentrations for humans
LD50/ED50 values for mice, divide by 10 for safe mice/rats, divide by 10 again bc humans r more sensitive, and divide by 10 again for extra caution
Lesson 53.2: How do we estimate potential harm of chemicals in the environment?
three steps to risk analysis
Risk Assessment
Identify hazard
Characterize toxicity (dose/response)
Determine exposure extent
Risk Acceptance
Determine acceptable level of risk
Risk Management
Determine policy with input from private citizens, industry, and interest groups.
Risk Assessment
risk analysis seeks to find a potential hazard and determine how bad it can harm
two types of assessment: qualitative and quantitative
environmental hazard: Anything in the environment that can potentially cause harm
substances like pollutants/chemicals, human activities like driving, natural catastrophes like volcanoes
qualitative judgments → low, medium, high risk
personal risk assessments, e.g. slowing down on a highway, are not quantitative and often don’t match actual risk
make sure perception of risk matches actual risk
data on probability of death from various hazards
can determine probabilities quantitatively
however, our perception messes w this; when we see news and media talking a bunch abt nuclear meltdown damage, we think it’s more dangerous than an airplane
we downplay risks that give us cultural, political, economic advantages
→ Quantitative Risk Assessment
Risk = probability of being exposed to hazard * probability of being harmed if exposed
→ A Case Study in Risk Assessment
noticed PCB might have harmful effects - live damage, impaired learning
now, start risk assessment
exposed rats to diff concentrations of PCBs; examined cancer cases of workers w PCBs
determine concentrations people experience → examine current concentrations in air, soil, water and half-life
persistent → high probability of PCB contact; contamination routes include eating fish, drinking water, breathing air
eating fish > water > breath → policies in Massachusetts: warned about not eating fish caught
risk of developing cancer is low, but high enough to dredge Hudson River and remove lots of settled PCBs
Risk Acceptance
second step - level of risk that can be tolerated
conflict bw those willing to have some risk and those who want no risk
EPA - 1 in 1 million is acceptable for most environmental hazards
some think too high, some think the minimal risk from nuclear deaths is worth less compared to electricity generation
Risk Management
integrates 1st and 2nd steps w economic, social, ethical, political issues
always trade offs when deciding amount of chemical allowed in environment; regulatory activity carried out by government
example: arsenic in drinking water for assessment v. management
even if 50ppb killed arsenic, FDA kept max at 50 ppb; then lowered it to 10ppb, which economically hurt mining companies and western states w high arsenic water
returned to 50 ppb, but new analysis determined 5ppb, so then they settled at 10ppb
Lesson 53.3: What are the major philosophies of regulating chemicals in the environment?
key factor to determining type of chemical regulation is guided by innocent-until-proven guilty principle or precautionary principle
innocent-until-proven guilty principle: A principle based on the belief that a potential hazard should not be considered an actual hazard until the scientific data definitively demonstrate that it actually causes harm
beneficial chemicals added sooner, but chemicals can affect wildlife for a while before scientists determine enough evidence for harm
precautionary principle: A principle based on the belief that when a hazard is plausible but not yet certain, we should take actions to reduce or remove the hazard.
scientific basis and intervention in proportion to hazard → fewer harmful chemicals enter environment
if chemicals indicates plausible risk and it proves harmless but beneficial, introduction for use can be delayed → less financial motivation for manufacturers to invest in research for new chemicals
great safety and slow addition vs greater potential risk and fast addition
most of world and Europe uses precautionary, USA uses innocent until proven guilty
benefit of precautionary principle example: asbestos
first mined → no evidence of harm 1879; deaths in 1906 → test mice 1911 shows harmful effects → 1930s lots factory workers suffered → 1955 high lung cancer isk → 1965 rare cancer
despite growing evidence, little was done
EU didn’t ban until 1998 → extra PPE → 250k-400k people today will die from past exposure to asbestos, could’ve been prevented with precautionary principle
netherlands banned in 1965 → less deaths and wayyyyy less expensive
International Agreements on Hazardous Chemicals
stockholm convention: A 2001 agreement among 127 nations concerning 12 chemicals to be banned, phased out, or reduced.
DDT pesticides, industrial PCBs, certain by-product chemicals of manufacturing
all known endocrine disruptors and banned or declining in use
all countries tg in forum to discuss controlling most harmful chemicals - huge achievement
REACH: A 2007 agreement among the nations of the European Union about regulation of chemicals; the acronym stands for registration, evaluation, authorization, and restriction of chemicals
embraces precautionary principle → more responsibility on chemical manufacturer
enacted bc many chemicals didn’t have rigorous risk analyses
phased in thru 2018 to allow sufficient time for required testing
Module 54: Pollution, Human Health, Pathogens, and Infectious Diseases
Lesson 54.1: How can we establish cause and effect between pollutants and human health?
other than testing on mice and extrapolating, another way to test effects of chemicals is studying populations already exposed everyday
two ways of conductive this research
1) Retrospective Studies
retrospective studies: A study that monitors people who have been exposed to an environmental hazard such as a harmful chemical at some time in the past
two groups usually - exposed to in past, never exposed, monitored for several years
helps determine immediate and chronic effects on development, births, body systems, etc.
help identify harmful effects of many common pollutants
exposed to vs not raw sewage → lots of bad chemicals and pathogens → emphasis on good sewage treatment worldwide
asbestosis!
tropospheric ozone → lung damage
2) Prospective Studies
prospective studies: A study that monitors people who might become exposed to an environmental hazard, such as a harmful chemical in the future
ask lots of participants to keep track of their habits for many years → determine if associated with future health problems
difficult cs lots of other risk factors, like socioeconomic status, included
synergistic interactions: A situation in which two risks together cause more harm than expected based on the separate effects of each risk alone
asbestos and smoking
lead poisoning in children is usually prospective
studied lead on children’s intelligence
accounted for other factors like mother’s IQ, tobacco exposure, intellectual development in home
high lead exposure = lower IQ
Lesson 54.2: What are the different types of human diseases?
¾ of death caused by disease: Any impaired function of the body with a characteristic set of symptoms
infectious disease: A disease caused by a pathogen
¼ of all deaths
pathogens include everything yk + parasitic worms helminths
4 types of infectious disease make up ¾ of all infectious disease deaths
respiratory infections, HIV/AIDs, tuberculosis, diarrheal diseases
acute diseases: A disease that rapidly impairs the functioning of a person’s body
chronic diseases: A disease that slowly impairs the functioning of a person’s body
Risk Factors for Chronic Diseases in Humans
risk factors very diff between low and high income countries
low income → malnutrition, poor sanitation, underweight, bad drinking water
high income → less young age deaths and more lifestyle diseases - tobacco, less activity, poor nutrition, overeating → obesity and blood pressure
Lesson 54.3: What historic human pathogens have cycled through the environment?
historically, pathogens have big toll on human health and mortality
epidemic: A situation in which a pathogen causes a rapid increase in disease
pandemic: An epidemic that occurs over a large geographic region, such as an entire continent
diseases associated w bad sanitation and water → cholera, hepatitis
dysentery: An infection of the intestines that causes diarrhea, which results in dehydration and can cause death; often caused by cholera
hepatitis can be caused by direct transmission bw 2 people, contaminated food
3 historically important diseases cycled bw environment and host
Plague
plague: An infectious disease caused by a bacterium (Yersinia pestis) that is carried by fleas
transmitted by flea bites or handling rodents
swollen glands, black spots, bad pain
still small outbreaks bc of few rodents; modern antibiotics work very well
Malaria
malaria: An infectious disease caused by one of several species of protists in the genus Plasmodium.
one stage of life in mosquitoes, next stage in humans
recurring flulike symptoms
hardest in subsaharan africa, asia, middle east, central/south america
USA malaria usually from travel
traditionally, insecticides with DDT were used to eradicate mosquitoes; ineffective
Tuberculosis
tuberculosis: highly contagious disease caused by the bacterium Mycobacterium tuberculosis that primarily infects the lungs
spreads thru coughing and droplets, persists in air, infects via inhalation
weakness, cold sweats, coughing blood
1/3 of world has infection, but few million get disease and die
year long antibiotics usually fixes infection in USA → huge drop in cases and deaths
leading cause of death by disease elsewhere in the world → medicine not accessible, not properly taken for full duration
pathogens quickly rebuild, last few bacteria most drug resistant
drug resistant strains r becoming new concern for new medicine
Lesson 54.4: What are the major emergent infectious diseases in humans?
emergent infectious diseases: An infectious disease that has not been previously described or has not been common for at least 20 years
at least 1 emergent disease per year; usually from diseases in animal hosts that happen to accidentally go to humans → rapidly mutate, new genotype for infecting humans
usually local, then spread across world
animal to human diseases include: hiv/aids, ebola, mad cow, bird flu, SARS, west nile virus
concern bc of increased movement of ppl and cargo
HIV/AIDs
Acquired Immune Deficiency Syndrome: An infectious disease caused by the human immunodeficiency virus (HIV)
Human Immunodeficiency Virus: A type of virus that causes Acquired Immune Deficiency Syndrome (AIDS
spreads through sex and blood transfusions - mother to kid, shared needles
hypothesized to have come from contact and consumption of chimps in africa
new antiviral drugs help maintain low viral levels + expand life expectancy
combinations of drugs to avoid evolving resistance → expensive and hard for low-income, but still increasing in availability and distribution
Ebola Hemorrhagic Diseases
ebola hemorrhagic fever: An infectious disease with high death rates, caused by several species of Ebola virus
first DRC → central africa; big issue bc has high mortality once infected
fever, vomiting, in and ex bleeding
kill other primate at high rates; they can’t have it, where is it from? → fruit bats
2019 first ebola vaccine approved
Mad Cow DIsease
mad cow disease: A disease in which prions mutate into deadly pathogens and slowly damage a cow’s nervous system.
prions: A small, beneficial protein that occasionally mutates into a pathogen
lose control of nervous system :(
transmits to humans by eating beef
prions hard to destroy even with cooking
CJD disease
transmits only through consumption of another cow w disease
1980s → cows ate other ground up cow, spread disease rapidly, spread to humans
can exist for many yrs before causing symptoms
hugely decreased cs no more cattle to cattle!
Swine Flu and Bird Flu
swine flu: A type of flu caused by the H1N1 virus
sometimes jumps from pigs to humans → may be lethal
vaccines help
bird flu: A type of flu caused by the H5N1 virus
people w close contact to birds; not deadly in wild birds but yes in domesticated birds
no evolutionary history w H5N1 → little defenses, high mortality
declined, but worried abt future mutation
SARs, MERS-CoV, and SARS-CoV-2
severe acute respiratory syndrome: A type of flu caused by a coronavirus
first in southeast asia → similar respiratory symptoms
wild animal infects person, then spreads
MERS-CoV: A coronavirus that causes the disease known as Middle Eastern Respiratory Syndrome
camels
SARS-CoV-2: A coronavirus that causes the disease known as Covid-19
traveled and rapidly spread globally → rapidly developed vaccines had good defense + social distancing + protective masks
West Nile Virus
west nile virus: A virus that lives in hundreds of species of birds and is transmitted among birds by mosquitoes
most birds survive, mosquitoes bite horses n humans n then it gets bad
1999 in NYC → spread to USA
now, ez to combat mosquito populations and protect against bites, declined
Lyme Disease
Lyme Disease: A disease caused by a bacterium (Borrelia burgdorferi) that is transmitted by ticks
hatches on forest floor → first attached to birds and small rodents → if infected, tick can also get infected cs feeding on → attach to larger mammals in winter → kiss humans n infect them
infections in northeast USA
red bullseye mark, flu, arthritis symptoms, neurological disorders
modern antibiotics work, some problems may persist
first discovered in schoolchildren in lyme
Zika Virus Disease
zika virus disease: disease caused by a pathogen that causes fetuses to be born with unusually small heads and damaged brains
bitten thru mosquito or sex w infected person
mostly mild effects of fevers and rashes weeks after infection, but huge risk of transferring to fetus while pregnant
rapid spread over time → no cure, just limit mosquitoes
Future Challenges to Human Health
combat diseases in low income
nutrition, more availability of clean drinking water, better sanitation
high income
more physical activity, balanced diet, limit tobacco and alcohol
educate everywhere
pathogen resistance is a big concern, esp when developing country people pause their medication
changing climates bring mosquitoes and diseases to new areas and infect new people
no experience to new diseases and no prediction → must develop rapid response plans
better notification and quarantine strategies → slow spread, give time for researchers
Lesson 54.5: What laws protect human health from pollutants and pathogens?
two important laws for aquatic pollution and pathogen protection in USA
The Clean Water Act
Clean Water Act: Legislation that supports the “protection and propagation of fish, shellfish, and wildlife and recreation in and on the water” by maintaining and, when necessary, restoring the chemical, physical, and biological properties of surface waters
not groundwater, passed in 1972
Federal Water Pollution Control Act 1948 → first major water quality bill
1960s → water quality poor, growing awareness encouraged series of laws
traditionally focused on chemical aspects of water, new attention to biological aspects like species
acceptable limits of pollutants in waterways → EPA and state controls how much pollution cities and industries dump
The Safe Drinking Water Act
safe drinking water act: Legislation that sets the national standards for safe drinking water.
maximum contaminant levels: The standard for safe drinking water established by the EPA under the Safe Drinking Water Act for 77 things in surface/ground water
account for harm and ease of reducing concentration
arsenic has 10ppb, e coli has 0
subjective to economic and political pressures
overall very successful
bodies of water based on purpose, determine if a waterway fully supports designated uses
municipal water systems are generally safe bc water regulations eliminated big point sources, nonpoint sources not covered and fracking too
Water Pollution Legislation in the Developing World
developed countries underwent industrialization and had horrible air and water quality, but then enacted legislation to clean up areas and prevent future problems
developing countries are still industrializing → hard to afford water-quality investments + political instability and corruption
polluting industries outsourced to developing nations; benefit from new jobs associated with their creation, but still pollution sucks
water pollution prevalent problems in africa, asia, latin america, eastern europe
china and india industrialized n sm people - uh oh
more affluence → more interest in addressing environmental issues, i.e. Tiete river
Unit 9: Global Change
Module 55: Stratospheric Ozone Depletion and its Reduction
Lesson 55.1: How does stratospheric ozone form and what benefits does it provide?
Formation of Stratospheric Ozone
O2 + UV-C → O + O
very short duration bc O mostly stays as O2
then, a free oxygen atom from that reaction meets with O2
O + O2 → O3
UV-B and UV-C can break this ozone into atomic and compound oxygen
O3 + UV-B or UV-C → O2 + O
indefinitely occurs as long as there is UV
normal conditions → ozone amount stays constant
Benefits of Stratospheric Ozone
ozone filters out harmful UV radiation
all types of UV can damage tissue of organisms
UV-B specifically increases skin cancer, cataracts, bad immune response, less photosynthesis in plankton → food chain impacted
stratospheric ozone is good, tropospheric ozone is bad
Lesson 55.2: What has caused the depletion of stratospheric ozone?
The Chemical Reactions between CFCs and Ozone
chlorofluorocarbons: Chemical that can be used for cooling refrigerators and air conditioners
also in aerosol spray cans and Styrofoam, essential to modern life + multibillion industries
seemed safe bc nontoxic and nonflammable
CFCs released into the air in use and in landfills, and they release chlorine that can react with ozone
O3 + Cl → ClO + O2
chlorine monoxide reacts with a free oxygen
ClO + O → Cl + O2
ozone and free oxygen make 2 oxygen molecules, but chlorine starts and ends as free chlorine
CFC Destruction of the Ozone Layer
stratospheric ozone has been overall decreasing, depleting most at the poles
occurs august through november → '“ozone hole”
key cause is CFCs bc when they r exposed to UV, they break into Cl
Cl reacts with ozone to make O2
manufacturing more CFCs → more chlorine in stratosphere → destruction of ozone each spring (southern hemisphere))
as ozone decrease, UV-B radiation reaching earth increased, esp in Antarctica
Lesson 55.3: What efforts have been made to reduce ozone depletion?
montreal protocol: A commitment by 24 nations to reduce CFC production by 50 percent by the year 2000
new coolers and propellants developed → HFCs, which are still a greenhouse gas
CFC concentration in atmosphere has been declining and chlorine too
depletion of ozone should decrease in following decades, and decrease skin cancer maybe
Module 57: Increases in the Greenhouse Gases and Global Climate Change
Lesson 57.1: How have CO2 concentrations changed over the past 7 decades?
IPCC - Intergovernmental Panel on Climate Change
understand effects of and mitigate effects of climate change
new understanding of greenhouse gas and temp link, esp CO2
Measuring CO2 Concentrations
earlier, most assumed excess CO2 absorbed by oceans and vegetation, and it’s so little it’s hard to measure
Charles Keeling was the first to overcome hard measuring of CO2
u dont need 2 separate measurements years apart, so Keeling measured CO2 in hawaii for a year → varied seasonally, increased each year
seasonal variation bc each spring, plants turn green and increase absorption of CO2
also, warmer bodies of water → algae more active
CO2 Emissions Among Nations
most carbon dioxide produced in the developed world
however, as developing nations industrialize, this number is changing
2009 → developing passed developed in carbon dioxide production, esp in China and India
however, lower per capita emissions in developing, more in developed
Lesson 57.2: How have temperatures and greenhouse gases varied historically?
since 1880, there’s been enough direct land and ocean temp measurements to generate a global temperature change over time graph
increased 1.1 C F, seven warmest years 2014-2020
not even distributed globally
antarctica and other areas cooler, some areas unaffected, but extreme northern latitudes have 1 to 4 C increase - northern ice cap melting
gradual warming over last century, but it’s also part of a natural warming
scientists estimate temp changes based on preserved protists (foraminifera) → major periods of cooling and warming
ancient air bubble isotopes with increasing and decreasing co2 concentrations
1958-present, CO2 concentrated went from 300ppm to 415ppm!!
methane and nitrous oxide dramatically increased bc of industrial revolution
Climate Models Predicting Future Global Temperatures
determine how well a model approximates by applying it to a time in the past where we have accurate data on temperature, vegetation, co2, ice coverage, etc.
modern models r fairly accurately produce temperature fluctuations over large spatial scales
not 100% accurate but huge confidence
if multiple models predict similar changes, increased confidence
generally agree temp rise 1.8 to 4 C by 2100, depending on CO2 emissions
expect climate change
more extreme weather
change in precipitation and ocean currents
some regions benefit from more yields and recharged aquifers
but also flooding, soil leaching, erosion
less precipitation elsewhere - famine, drought, water
Lesson 57.3: How has global climate change affected the environment?
Melting Polar Ice Caps
ice cap significantly reduced, collected each september
thinner remaining ice - vulnerable to future melting
arctic expected to warm 4 to 7 more C → large openings in sea ice
ice melting in greenland and antarctica
ice cap loses thickness, so overall mass reduced
sea levels rise
Melting Glaciers
glaciers dramatically disappearing
loss of ecosystem services
recreation
clean water source - if melts too fast during summer, none in winter
more exposed soil, less albedo, more warmth, positive feedback loop
Ocean Currents
more fresh water released from ice, changing currents and heat distribution
more melting from greenland and north ice cap dilutes thermohaline salty cold water to stop it from sinking in greenland, cutting off thermohaline circulation
europe gets way colder temp
Warming Soils and Permafrost
high temps → more activity in carbon packed soil → increased decomposition → more CO2
melting permafrost makes overlying lakes smaller as water drains deeper
problems w buildings built on permafrost like houses and oil pipelines
lots of organic matter in tundra permafrost will decompose
wet, low oxygen conditions → decomposition releases a lot of methane
melts more, more methane positive feedback loop
Rising Sea Levels
rising temp affects sea levels 2 ways
water from glaciers and ice sheets adds to volume of water
as water expands, the ocean becomes warmer
endangers coastal, low lying cities
saltwater intrusion in aquifers, more soil erosion
some countries use dikes on shoreline to mitigate flood
poor countries can’t respond as well
new habitats for aquatic organisms along flooded shorelines
deep water species no longer in photic zone
Lesson 57.4: How is global climate change affecting populations?
temperature-induced changes in plant flowering and animal behavior + ability of plants and animals to find more hospitable habitats
northern hemisphere plants’ growing season increased in highest latitudes w most temp change
everything breeds and plants and comes earlier
grape growers in France having hard time bc too warm and diff rain, but in England, the warmth is helping them
effects on human health and agriculture
disease vectors shift north → affect human health and crop pests make food decline
people move
rapid changes harm organisms if they don’t adapt or move in time
historically, organisms migrate - so temp changes r not usually catastrophic
now, habitat fragmentation allows global warming to promote species extinction
flycatcher example
bc of global warming, trees produces leaves earlier and peak caterpillar abundance is two weeks earlier than normal
however, bird hatching still the same, so when they hatched, there wasn’t enough caterpillars and they suffered :(
arctic experiencing extreme effects
polar bears hunt seal blubber (good energy) by searching for holes in ice and pouncing on seals that come up
rest of carcass important for arctic fox
fewer weeks polar bears can stand on ice to hunt cs ice melts earlier → polar bears lost sm weight
less seal predation, less carcasses for other organisms
polar bear in hudson bay declining, increasing in or stable in arctic
new skipping lines - reduce distance ships have to travel
lots of oil and natural gas under polar cap, but fossil fuels bad
Module 58: Ocean Warming and Ocean Acidification
Lesson 58.1: How is ocean warming altering ocean ecosystems?
2 C warming in bodies of water
Impacts on Marine Species
alters species distribution bc each species has diff optimized factors
others migrate to better temp, those in higher latitudes can’t really
southern fish moved to northern sea bc of warmer temps there
incoming species worth less, departing fish species worth more
corals at risk bc low temperature range
coral bleaching → stressed colors eject mutualistic algae that provide coral w energy → coral turns white
long lasting → death
Lesson 58.2: How does climate change affect ocean pH?
more co2 absorbed by oceans
CO2 + H2O → H2CO3 → releases positively charged hydrogen atoms (protons)
CO2 quickly reacts with H2O to form H2CO3 (acid) that releases H+
equilibrium reaction
ocean acidification: A process in which an increase in ocean causes more to be converted to carbonic acid, which lowers the pH of the water.
problem for species that build shells and skeletons made of calcium carbonate
pH decreases, calcium carbonate dissolves and saturation point for calcium carbonate declines → less material for shells and skeletons + break down
decline of corals = high temp + low pH + more coral disease
affects behavior of organisms
less ability to detect predators
sea urchins grow smaller, while lobsters grow larger and thicker shells as a response
some species perform better
affects human health and food supply
declining pH → huge monetary loss in shellfish industry
threatens jobs
algae produce more toxins harming humans
Lesson 58.3: What are the international agreements on global climate change?
nations must work tg → developing nations have it worse cs worse effects and less benefits
Kyoto Protocol: An international agreement that sets a goal for global emissions of greenhouse gases from all industrialized countries to be reduced by 5.2 percent below their 1990 levels by 2012.
varying emission reduction per country; developing nations didn’t bc they argued that the poorest didn’t contribute but have been exposed
countries w historically most emitting co2 must pay costs to reduce co2
based off precautionary principle
enough evidence to see humans altering earth → stabilize gg concentrations by reducing or removing them
first option) increase fuel efficiency or switch to better options
second option) carbon sequestration
taking co2 out of atmosphere, like storing in soils, former farms, forests, and pastures
atmospheric carbon returns as plant biomass and soil carbon
cost effective ways to capture atmospheric and power plant co2 being researched → compressed and pumped into old wells or ocean
developed countries → reducing co2 means we gotta change a lot of things, huge expenses and economic impact
kyoto protocol not legally binded to the US because Senate voted that US won’t agree to any international agreement that lacked restrictions on developing nations
192 countries ratified this (most developed and developing), though 100 developing like india and china are exempt
US is the only unratified developed one
US gov takes more steps to regulate co2 emissions
2007 → SCOTUS said EPA can regulate greenhouse gases thx to Clean Air Act + REQUIRED to do so, so 2009 EPA started regulating
automakers increased fuel efficiency → invest in new tech, less fossil fuels, save money
mixed co2 emissions
germany, russia, and USA have significantly decreased bc of fuel efficiency or natural gas use
canada increased
Paris Climate Agreement: A pledge by 196 countries to keep global warming less than 2°C above pre-industrial levels. Also known as the Paris Climate Accord
each nation decides itself how they wanna contribute, went into effect 2020
no enforcements by UN
Module 59: Invasive Species, Endangered Species, and Humans Impacts on Biodiversity
Lesson 59.1: What are the threats posed by invasive species?
native species - historical range, exotic species out
exotic species that quickly spread across large areas, decline native species, harm ecosystems, or negatively impact humans - invasive species
r selected, generalist species - mosquitoes, Norway rats
Great Lakes lots of invasive species introduced from other ships
zebra mussel from black sea quickly grow - outcompete native mussels, clams, etc.
clog pipes, impede flow of industrial water
most r too numerous to be controlled, arriving on every continent
most arrive accidentally, some intentionally introduced like honeybees from Europe
good for honey and pollination, but decline native populations
kudzu vine from japan and southeast china
planted in southeast to reduce erosion, great climate rapid growth
grows so fast, overshadows sm flowers and trees
silver carp from Asia spread quickly thru rivers
may outcompete native species for algae
randomly jumps out and attacks boater
act as predators, pathogens, competitor
Controlling Invasive Species
remove aquatic plants when they r first observed and low in abundance
introduce enemy from their native range
cactus introduced from south america to australia and outcompeted plants with sheeps grazed on
cactu moth eats prickly pear and reduces abundance
best way to reduce is to prevent - prohibit untreated wood crates to prevent pests
Lesson 59.2: Why are species becoming endangered?
The Percentage of Endangered Plants and Animals
extinct species existed in 1500 but no longer exist today
endangered species: Species that are likely to go extinct in the near future
threatened species: likely to go to endangered soon
near-threatened to be threatened later
least-concern r widespread and abundant
International Union for Conservation of Nature had 50k
1/3 are threatened / endangered
higher for reptile and mammals, highest conifers and amphibians
Cause of Endangered Species
human activities make it harder to survive and adapt fast enough
overharvest, clear habitat, competitors
Lesson 59.3: How are human activities affecting genetic biodiversity?
low genetic diversity - cant survive cs theres not enough phenotypes and not enough mutations
cheetahs have little genetic diversity, florida panther
Declining Genetic Diversity of Domesticated Species
38 species of livestock come from 7 mammals and 4 birds
bred for diff characteristics to survive their location
farmers breed based on economic productivity
green revolution - fewer crop varieties w only high yield ones
lost 75% of crop diversity
very vulnerable if environmental conditions change
store seed diversities in special warehouses for the future - Svalbard Global Seed Vault Norway
designed to resist global warming and natural disaster, but at risk of destruction from warfare
Lesson 59.4: What are the causes of declining biodiversity?
underlying cause for less biodiversity is human increase - less habitats, more invasive species, population growth, pollution, climate change, and overexploitation
Habitat Destruction
human development removes natural landscapes, replaces with homes, industries, fields, malls, roads
specialists r prone to population declines
forests have been destroyed for lumber and agriculture historically, humans replanted them but not as diverse
developing areas - accelerated clearing
woodland/shrubland in mediterranean also lost a lot of land, and wetlands
carribean losing a lot of coral - warming oceans, pollution, removal
species can decline without full destruction
no cooler areas to go during winter - animals cant go
some larger species need more undisturbed land
increased interactions with harmful species
Invasive Species and Human Population Growth
increased demand for resources from nature
Pollution
toxic contaminants, endocrine disruptors, nutrient pollution, thermal pollution
huge oil spill, used chemical dispersant to cleanup but thats also harmful/
Climate Change
species have varying responses to diff climate change
Overexploitation
most direct - used faster than can be replenished, recent past
more legal protections
billions of passenger pigeons shot/caught at once
tried replenishing, but lay one egg at a time
increased federal regulations
poaching: harvesting outside of seasonal time
international treaties r very successful
however, not enforced or illegal everywhere, esp for rare sought after species
Lesson 59.5: How do we conserve biodiversity?
Conservation of Single Species
one species at a time
increase ideal conditions, or if very few, capture and breed them
major functions of zoos and aquariums
many laws
→ The Lacey Act 1900
Lacey Act: A U.S. act that prohibits interstate shipping of all illegally harvested plants and animals
→ The Maine Mammal Protection Act
marine mammal protection act: A 1972 U.S. law that prohibits the killing of all marine mammals in the United States and prohibits the import or export of any marine mammal body parts
→ The Endangered Species Act 1973
Endangered Species Act: A 1973 U.S. law designed to protect plant and animal species that are threatened with extinction, and the habitats that support those species
authorized US fish and wildlife service to categorize species and prohibit their harming
purchase habitat to conserve these species
as of 2022, 1600 species r endangered or threatened
controversial bc restricts human usage of their land - less construction, limits people’s jobs
lack of personnel and funds to enforce law
→ Convention on International Trade in Endangered Species of Wild Fauna and Flora
CITES: A 1973 treaty formed to control the international trade of threatened plants and animals.
each country has agency to monitor and regulate spread of species on Red list
still occurs
Conserving Entire Ecosystems
worried about mass sixth extinction - preserve biodiversity hotspots, increased protection rates
→ Configuring Protected Areas
apply theory of island biogeography
protected areas are far apart - less species travel, so if lost from one ecosystem, hard to recolonize
small areas should be close enough for species to move between
single large area supports lot, but a species is more likely to survive if scattered in multiple small areas
SLOSS - single large or several small
due to human development, several small may be the only viable option
also determine how much edge habitat we should care about
→ Biosphere Reserves
we want to preserve pretty areas but not have tm human degradation - UNESCO made biosphere reserves: protected areas of zones that vary in permissible human impact
buffer zone surounds core areas - modest tourism, environmental education, scientific research
farther transition area - sustainable logging agriculture residence for locals
→ The Restoration of Habitats
restoration ecology - florida everglades and chesapeake bays
restore water flow, nutrient inputs