Topic 7 Radioactivity and particles
Radioactivity
At the center of every atom is a nucleus It contains protons and neutrons
On the outside is energy levels also known as shells
Isotopes are atoms with diffrent numbers of neutrons
Radioactive decay is a random process
Unstable Isotopes: Nuclei of unstable isotopes decay randomly; you can't predict which will decay.
Spontaneous Decay: Each nucleus decays on its own schedule, unaffected by external conditions.
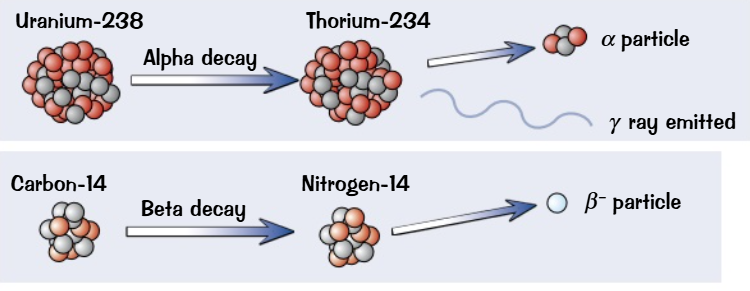
Types of Decay: When a nucleus decays, it can emit alpha (α), beta (β), gamma (γ) radiation, or neutrons.
Formation of New Elements: The decay process transforms the nucleus into a new element
Background radiation
Cosmic Radiation
Terrestrial Radiation
Radon Gas
Internal Radiation
Medical Procedures
Fallout from Nuclear Tests
Ionizing radiation
Nuclear Radiation: Causes ionization by knocking electrons off atoms, creating charged ions.
Damage Pattern: The further radiation penetrates an atom, the less damage it causes.
Detection: Ionizing radiation can be detected with a Geiger-Müller detector or photographic film.
Types of Radiation: Understanding requires knowledge of three different types of ionizing radiation.
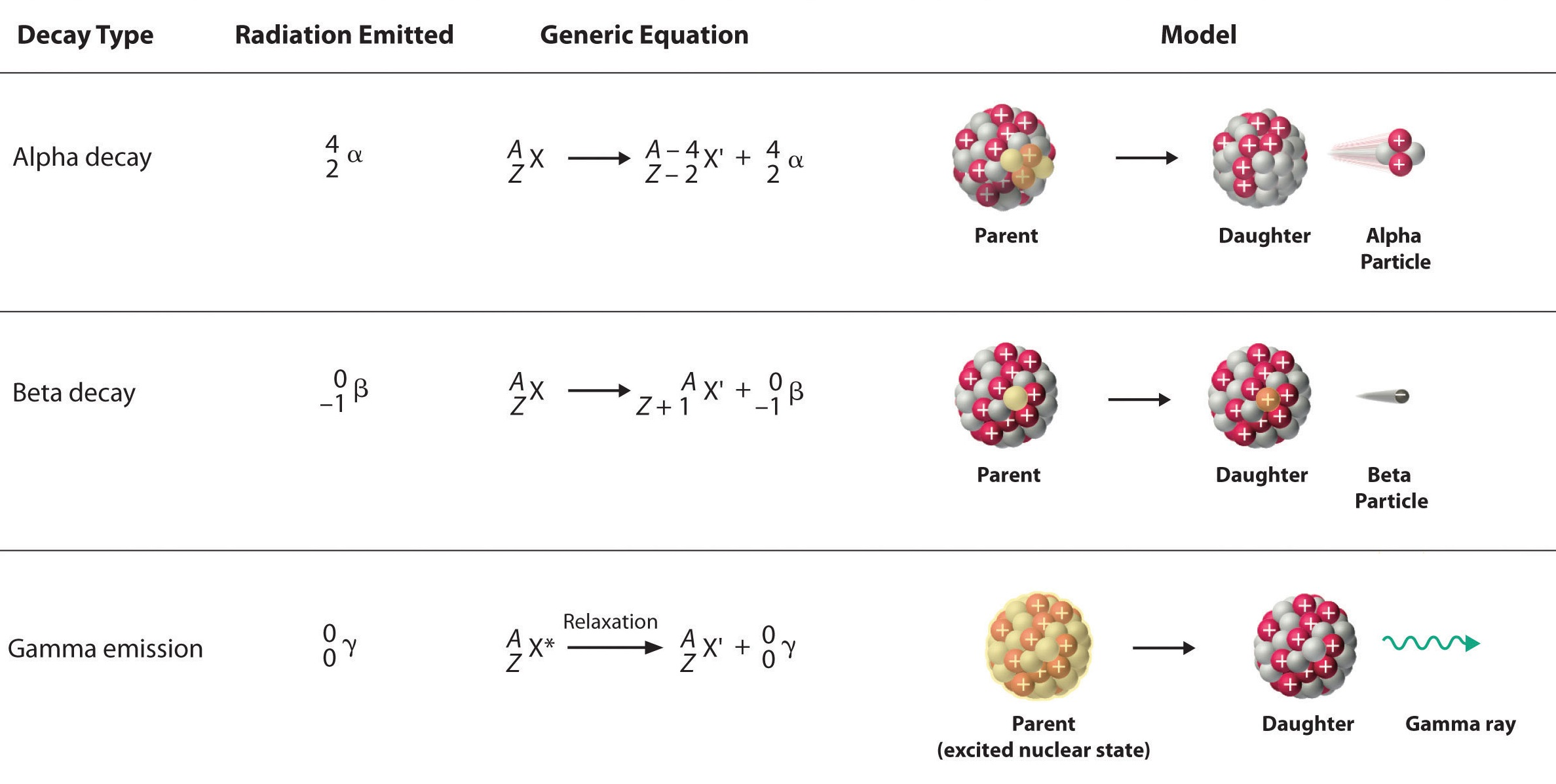
Alpha Particles |
|
Beta Particles |
|
Gamma Rays |
|
Investigating radiation & Nuclear equations

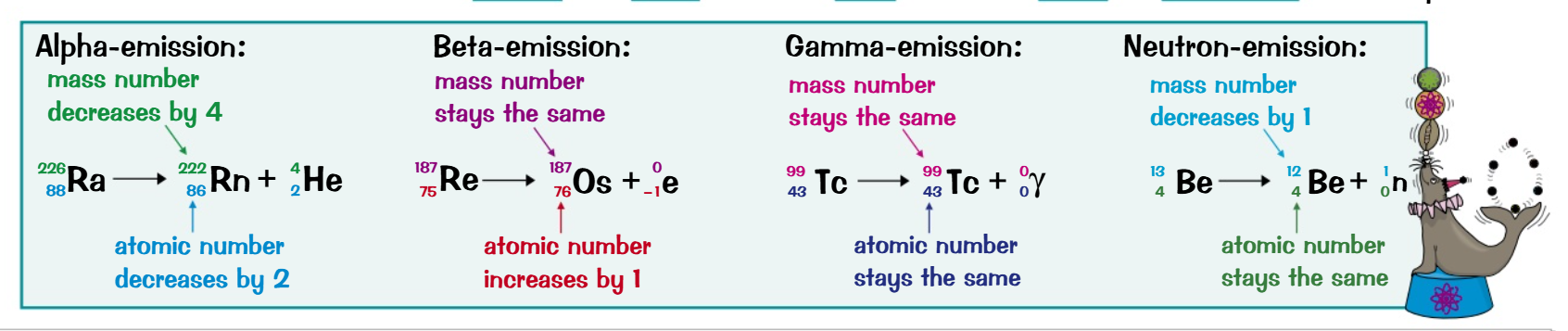
Nuclear Reaction Equations: Similar to chemical reactions.
Conservation: Charge and mass are conserved.
Balance: Atomic and mass numbers must be equal on both sides.
Half - Life
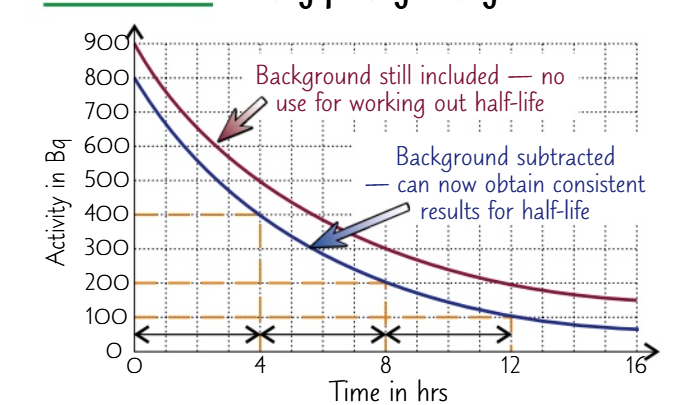
Half - Life the time taken for half of the radioactive atoms now present to decay
Decay Process: Each time an alpha or beta particle is emitted, a nucleus decays.
Unstable Nuclei: As unstable nuclei decay, the number decreases over time.
Activity Measurement: The rate at which activity drops varies; some isotopes take years, while others take just a few hours.
Half-Life Definition: Half-life is the time for half of the radioactive atoms to decay.
Decay Rate: Short half-life indicates rapid decay; longer half-life means slower decay.

Uses of nuclear radiation
Medical Tracers: Beta or gamma radiation sources are employed in medical diagnostics. These tracers are injected into the patient and emit radiation, allowing doctors to visualize organs and detect issues through imaging techniques.
Cancer Treatment: Radiation therapy uses targeted radiation to destroy cancer cells. While effective in shrinking tumors, it must be carefully managed to minimize damage to surrounding healthy tissue.
Food Irradiation: This process involves exposing food to ionizing radiation, which kills harmful bacteria, parasites, and insects. It helps extend shelf life and improve food safety without significantly affecting nutritional value.
Industrial Applications: In industries, radiation is used in tracers to monitor processes and locate leaks. Thickness gauges utilize radiation to measure material thickness in manufacturing, ensuring quality control and consistency in production.
Risks from nuclear radiation
Tissue Damage: Ionizing radiation, such as alpha and beta particles, can penetrate the skin and damage delicate cells and tissues. This can lead to harmful effects, including cancer.
Irradiation: Exposure to radiation is termed irradiation. It signifies that radiation has interacted with the body, potentially causing cellular damage.
Contamination Risks: If radioactive materials contaminate the skin or clothing, there is a risk of internal exposure if they are ingested or inhaled.
Radioactive Waste: Disposal of radioactive waste poses significant challenges. It must be managed carefully to prevent environmental contamination. High-level waste requires secure storage for thousands of years due to its long half-life and potential harm.
Safety Measures: It is crucial to implement safety protocols to minimize exposure, including using protective equipment, maintaining distance from radiation sources, and ensuring proper waste management.
Nuclear fission and fusion
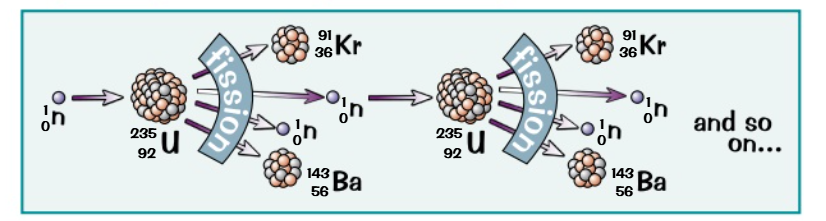
NUCLEAR FISSION CHAIN REACTION
Nuclear Fission: The splitting of a nucleus (like uranium-235) releases energy, which can happen spontaneously or be induced.
Chain Reactions: A neutron absorbed by uranium-235 causes it to split, releasing more neutrons that trigger further fissions.
Energy Production: Each fission event releases energy, which is converted into thermal energy to produce steam.
Radioactive Waste: Fission creates radioactive byproducts that pose disposal challenges.
Electricity Generation: The steam drives turbines, converting thermal energy into electricity.
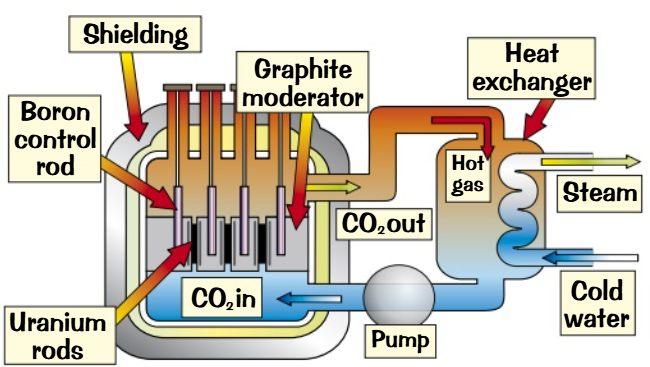
Nuclear reactors have to work safely
Neutron Moderation: Neutrons are slowed down using a moderator like graphite or water.
Control Rods: Boron control rods absorb excess neutrons to regulate fission.
Radiation Shielding: Thick concrete shields protect against high-energy neutrons and gamma rays.
Heat Absorption: A substance like carbon dioxide absorbs heat from fission and transfers it to water.
Electricity Production: The heated water turns to steam, driving a turbine that generates electricity.
NUCLEAR FUSION JOUNUNG SMALL NUCLEI
Nuclear Fusion: The process where two light nuclei (e.g., hydrogen) collide at high speed to form a larger nucleus, releasing energy.
Energy Release: Fusion releases more energy than fission for a given mass of fuel, making it a powerful energy source.
Conditions Required: Fusion requires extremely high temperatures and pressures (around 10 million °C) to overcome the repulsive forces between positively charged nuclei.
Challenges: Current fusion reactions are difficult to sustain due to the extreme conditions needed, making them complex and costly to develop for energy production.