Unit 4 Chemical Compounds
Law of Definite Proportions/ Constant Composition
Joseph Proust
All pure compounds contain the same elements in the same proportion by mass/ Atoms of elements have the same ratios
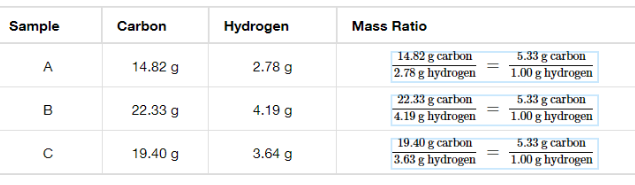
Percent Composition
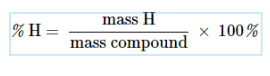
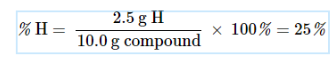
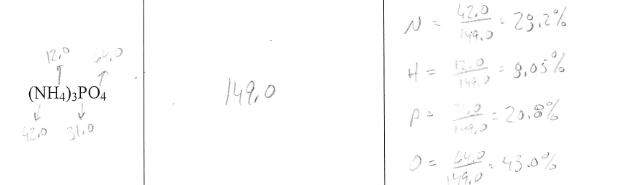

Ionic Compounds
Metal (Cation) and Non-Metal (Anion)
generally, the metal is positively charged (cation) and the negatively charged one (anion) is the non-metal
1st Metal ____
Metals = Left side to middle
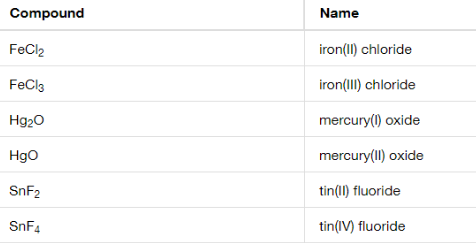
Binary Ioninc Compounds: means only two diff elements
-ide is added at the end of the second element
look in packet
the roman numeral is on the opposite one
Ex. FeCl2 = Iron (2) Chloride

General Special Names for Oxyacids
+1 O: Per- ____ - ate
Original: ____ -ate
-1 O: ___ ite
-2 O: Hypo- ____ -ite
Hydrates
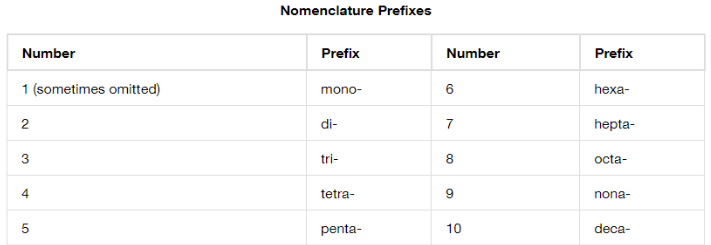
Prefix for multiples
Ex. Sodium Carbonate Decahydrate is Na2CO3 × 10H2O
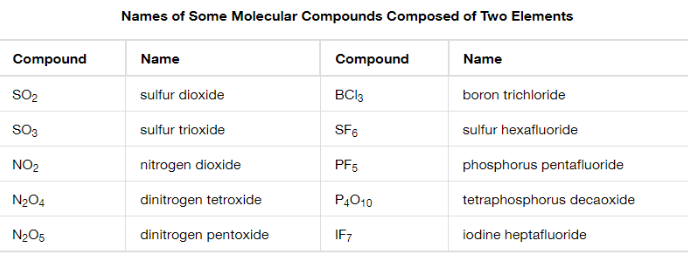
Molecule (Covalent) Compounds
1st nonmetal ____
Always use prefix for 2nd part
Only use prefix for 1st when it’s a multiple
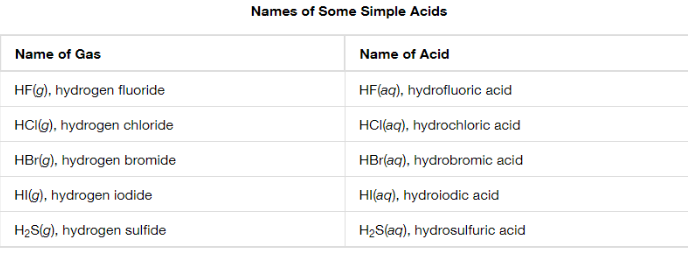
Binary Acids
Hydrogen
Hydro- turned to prefix
Other nonmetallic element name modified with suffix -ic
“acid” as second word
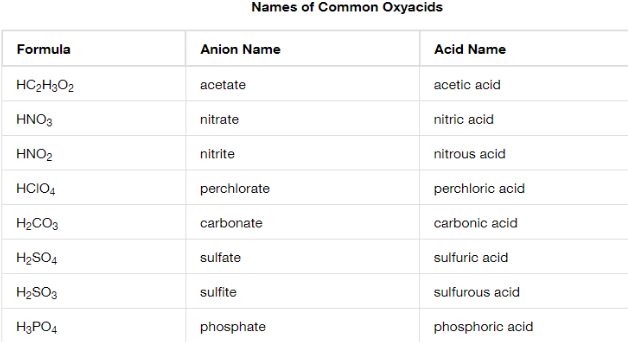
Oxyacid
Omit “hydrogen”
Start with root name of the anion
-ate replaced with -ic
-ite replaced with -ous
Add “acid”
Graham’s Law of Effusion

when given an amount and time, divide the amount/time to get the effusion rate
unit: g/mol (grams per mole)