Notes for Topic 2.1: Quantum Numbers and Atomic Orbitals
Electrons as Waves and the Wave-Particle Duality
Electrons are best understood as waves and particles (wave-particle duality).
Waves & Particles as the correct framing.
This dual nature is supported by experiments showing wave-like behavior for electrons.
Quantum Mechanics to the Rescue
Evidence that electrons have wave properties:
Electrons produce a diffraction pattern
Electrons produce an interference pattern
Electrons have allowed states and forbidden states (quantized energy levels)
Four Quantum Numbers and Orbitals
Solving Schrödinger’s equation gives energy levels and wave functions for electrons (orbitals).
Four quantum numbers describe each electron: n, l, , .
Principal quantum number (n): Determines size and energy; number of nodes = (n - 1).
Azimuthal (orbital angular momentum) quantum number (l): Describes orbital shape (s, p, d, f); possible values depend on n.
Magnetic quantum number (): Describes orientation on Cartesian plane; orientation of the orbital.
Spin quantum number (): Describes spin state of the electron.
Principal Quantum Number, n
Determines size and energy of the orbital.
Number of nodes =
Specific values: (positive integers).
Notation examples:
Azimuthal Quantum Number,
Also known as orbital angular momentum quantum number.
Describes the shape of an orbital.
Possible values depend on n: .
Specific value mappings: = 0 → orbitals, → p orbitals, → d orbitals, → f orbitals.
Example: Which orbital does not exist?
Based on the rule , an orbital with and (2d) does not exist.
Magnetic Quantum Number,
Describes orientation of the orbital (along x, y, z coordinates).
For a given , possible values:
Example: If (p orbital), .
Spin Quantum Number,
Describes the intrinsic spin of the electron.
Possible values: .
Pauli Exclusion Principle
No two electrons in an atom can have the same set of four quantum numbers
Consequence: electrons in the same orbital must have opposite spins (paired spins).
Note: While the principle implies opposite spins for electrons in the same orbital, the general statement is about distinct quadruples of quantum numbers across all electrons in an atom.
Which orbital corresponds to this quantum number set? (Example 2.1.4 b)
For (b) 1, 1, 0, -1/2: Not valid, does not exist.
Reason: n = 1 allows only (1s); would require
Shapes of Atomic Orbitals: n = 1
At
Number of nodes
, so only s orbital.
only one orientation.
Represented as .
The depicted wavefunction is an oversimplification – the real wavefunction is three-dimensional.
Representations of the 1s Orbital
1s orbital representations include various views of the electron density or probability density map.
Electron probability density is given by
Shapes of Atomic Orbitals: n = 2
At :
There is one node.
and p orbitals.
The wavefunction is three-dimensional.
The 2s Orbital
2s orbital: or .
(only one orientation).
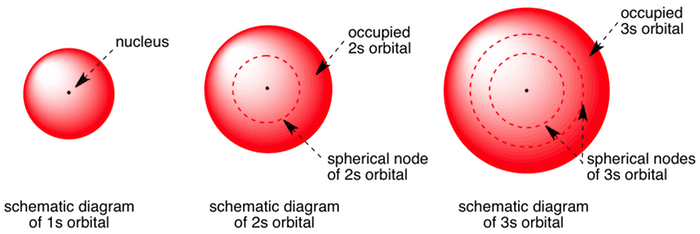
Summary and Notation
Orbital labels depend on n and Orbital notation: with spin notation included when considering multiple electrons.
Viability of an orbital requires n > l and within .
For a given n, the number of available subshells is n (i.e., s, p, d, … up to ).
Connection to Concepts and Real-World Relevance
Quantization of energy levels explains discrete emission/absorption spectra in atoms.
The shapes of orbitals reflect electron density distribution, influencing chemical bonding and molecular geometry.
Understanding spin and Pauli exclusion is essential for predicting electron configurations and periodic table structure.
The 3D nature of orbitals highlights why electron density is distributed in space rather than along a single path.
Practice and Application Hints
When evaluating a set ( ):
Check range: 0 ≤ l ≤ n − 1.
Check range: .
Check values: ±1/2.
For a given : the orbital is labeled as nℓ with labeling the specific orientation (e.g., 3p with = 0 corresponds to one orientation).
The number of nodes in an orbital is : this helps gauge complexity of the orbital's radial distribution.
Course Context and Study Tips (From the Transcript)
Topic 2.1 Team Module: Find or join a team on OWL Groups (Communications). Complete before Test #1. Ensure you work in your own OWL account and submit the completion quiz.