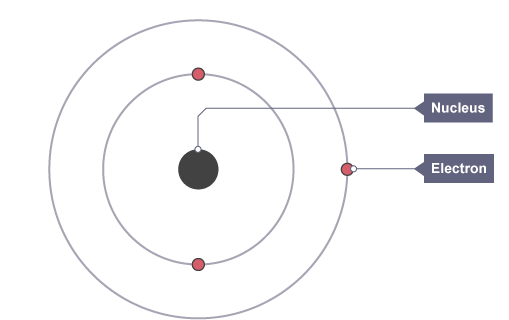
Electrons
ALL matter consists of atoms
Atoms contain three types of smaller particles
Protons are positively charged;
Electrons are negatively charged;
Neutrons have no charge – they are neutral
so atoms of neutrally charged with the same number of electrons and protons
Protons and neutrons are found inside the nucleus while electrons orbit the nucleus
Ions:
If it loses an electron, it becomes a positively charged ion.
If it gains an electron, it becomes negatively charged ion.
-charged atoms are called ions
Key points
An electric current is a flow of charged particles in one direction.
Metals are good conductors of electricity because they contain free electrons.
Insulators such as plastic and rubber do not generally contain free electrons and so are poor conductors of electricity
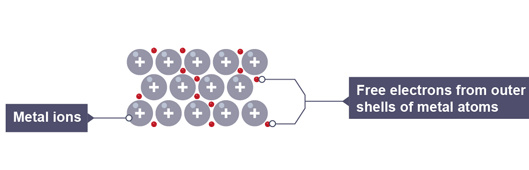
What are free electrons?
-particles in a metal are held together by strong metallic bonds
Metals atoms have loose electrons in the outer shells, which form a 'sea' of delocalized or free negative charge around the close-packed positive ions.
These loose electrons are called free electrons.
They can move freely throughout the metallic structure
Direction of flow of free electrons
Energy is required to make the free electrons travel in one direction.
An electric cell (often called a battery) can supply this energy and make free electrons move in a metal conductor connected between its two terminals
Key points
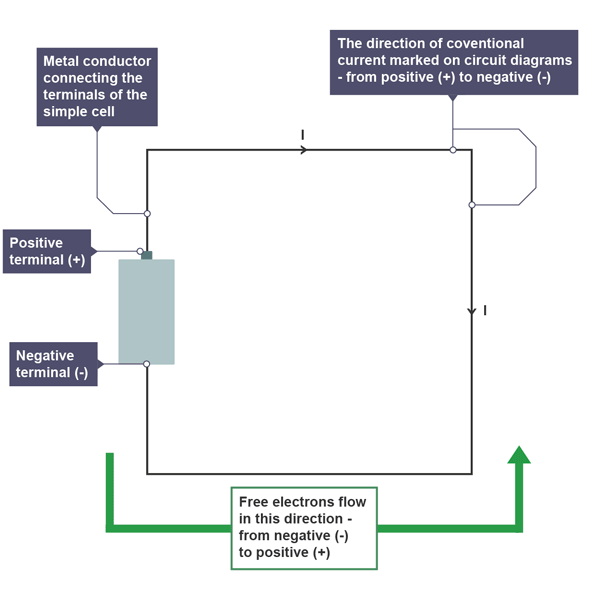
The direction of conventional current is from the positive terminal, through the conductor, to the negative terminal.
The direction of free electron flow is from the negative terminal, through the conductor, to the positive terminal.
The direction of conventional current is the direction marked on all circuit diagrams.
The symbol for electric current is I.