Biology H Unit 3 - Organic Chem
Carbon
All living things are made up of carbon-based molecules. Carbon enters the biosphere through photosynthesis. Carbon can make up to 4 covalent bonds (as it has 4 valence electrons); it may form almost limitless numbers of carbon skeletons as it readily bonds with other atoms as well as itself. Its bonds may be single, double, or triple and its chains may be straight, ringed, or branched.
Organic Compounds
- made up of carbon and hydrogen atoms
Inorganic Compounds -
do NOT contain carbon and hydrogen atoms
Functional Group
Hydrocarbon - an organic molecule with ONLY carbon and hydrogen
Substituted Hydrocarbon - hydrocarbon with 1+ hydrogen atoms substituted by another atom/group of atoms also known as a functional group.
Substituted Hydrocarbon = Functional Group
Function groups are specific configurations of atoms which are usually attached to carbon skeletons
They always behave the same
They give molecules unique properties
Typically cause chemical reactions
Hydroxyl (-OH)
Compound Name: Alcohol
Names usually end in: -ol
Polar
Attracts water molecules
Helps dissolve organic compounds
Singled-bonded
Examples: Ethanol, Methanol

Carbonyl (-CO)
Compound Names: Aldehyde and Ketone
Aldehyde - When the functional group is at the END of a carbon change; its characteristics depend on the rest of the molecule. Names usually end in -al
Methanal
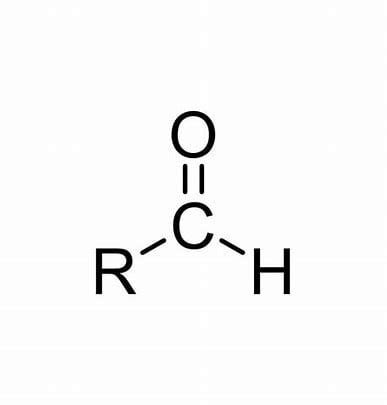
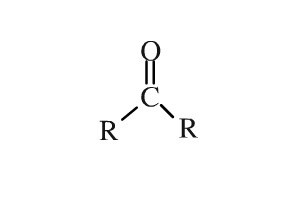
Ketone - When the functional group is somewhere WITHIN the carbon change; characteristics also depend on the rest of the molecule. Names usually end in -one
Testosterone
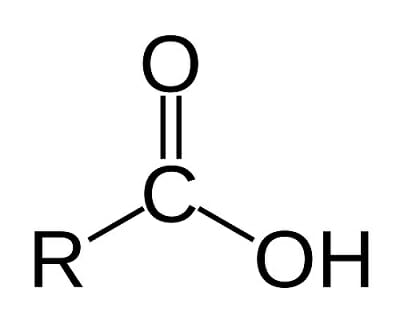
Carboxyl (-COOH)
Compound Name: Carboxyl/Organic Acid
Names usually end in -oic
Acidic —> H+ donor (pH lab, less hydrogen = more acidic, more hydrogen = more basic)
Strong Odor
Ethanoic Acid
Butanoic Acid
Amino (-NH2)
Compound Name: Amines
Names usually end in: -amine
Alkaline (basic)
Smell like rotten fish

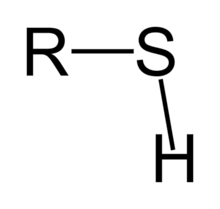
Sulfhydryl (-SH)
Compound Name: Thiols
Names usually end in: -thiol
Stabilize protein structures
Some have a stinky odor
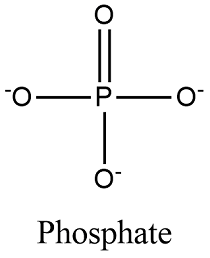
Phosphate (-PO4)
Compound Name: Phosphates
No set name ending
Transfers energy
the Anion in energy transfer (negative)
Macromolecules
Necessary substances to all living things, a macromolecule is a huge molecule that is formed when smaller molecules are linked together; macromolecles are polymers. The 4 main classes are Carbohydrates, Lipids, Proteins, and Nucleic Acids.
ex. Amino acids combine to make proteins (quizizz)
Monomer - a SINGLE sub-unit which serves as a building block for polymers
Polymer - a long chain of similar or identical monomers linked together (Monomer+Monomer = Polymer)
Dehydration Synthesis:
A process that joins monomers together by removing water, (occasionally called condensation reaction)

Hydrolysis -
Any chemical reaction where a water molecule breaks 1+ chemical bonds (seperation)
Carbohydrates
Carbohydrates are divided into 3 classes: Monosaccharides, Disaccharides, and Polysaccharides (sugars)
Water soluble (can dissolve)
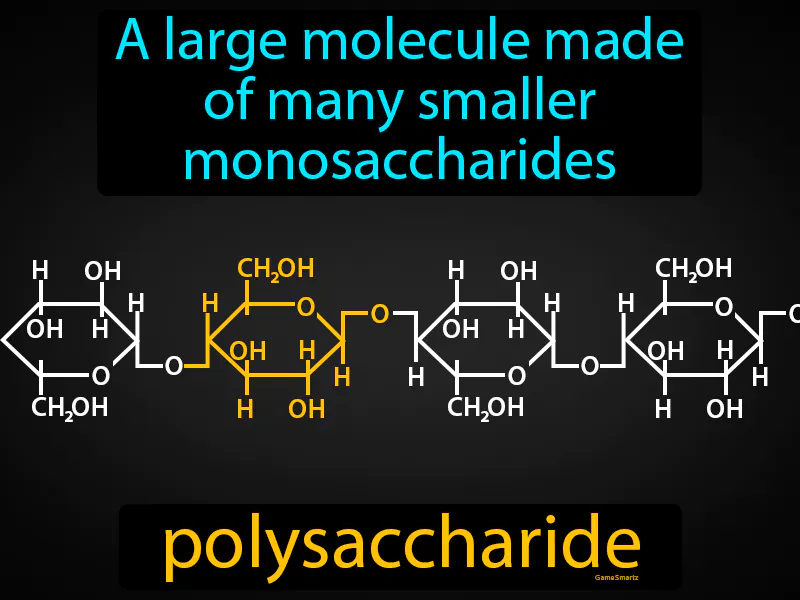
Monosaccharides:Monomer for larger carbs
Has carbonyl and hydroxyl groups
Chemical Structure: carbon, hydrogen, oxygen (CnH2nOn) (CHO)
Found in simple carbohydrates (honey, apples, cheese)
Quick energy
Building blocks (especially in plants)
Ends in -ose
Hexagon/Pentagon shaped
Isomer - molecules with the same molecular formula but differ in the arrangement of atoms
2:1 Hydrogen:Oxygen
(Glucose, Ribose)
Disaccharides:
Forms when 2 monosaccharides are joined by a -
glycosidic linkage (which is their covalent bond) through dehydration synthesis

Polysaccharides:
Composed of long chains of monosaccharides
Polymer of carbohydrates
Found in complex carbohydrates (breads, pastas, potatoes)
Provide longer-term energy/Energy storage
Structural
Types of Polysaccharides
Glycogen (Animal) - Storage of excess sugars (in liver and muscles), muscle contraction, breaks down glucose for quick energy
Starch (Plant) - Stored excess sugar in plants
Cellulose (Plant) - Gives plants rigidity, component of the cell well
Lipids
Fats, oils and waxes (ex. animal fats, butter, shortening)
Long-term storage of energy (double of carbs)
Insulation and cushioning
Absorption of some vitamins
Component of cell membranes
Chemical Structure: Carbon, Hydrogen, Oxygen (CHO)
Hydrophobic - insoluble in water
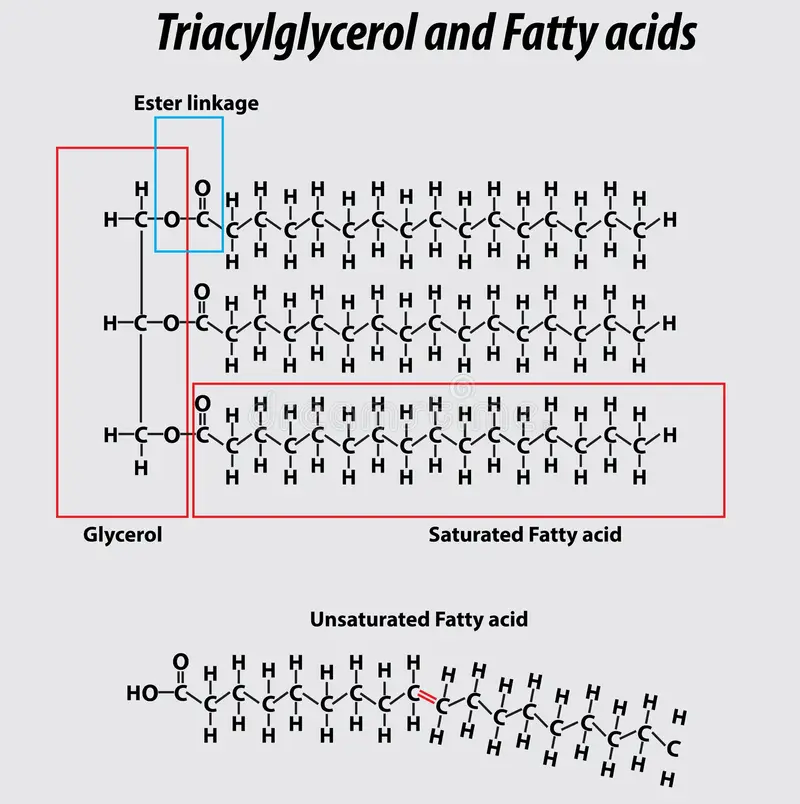
Monomer - Glycerol and fatty acid
Polymer - Triglycerides (Glycerol + 3 fatty acids tails, linked by ESTER linkages)
Fatty Acids have Carboxyl function groups
1 Glycerol + 3 Fatty Acids = Triglyceride + 3H2O
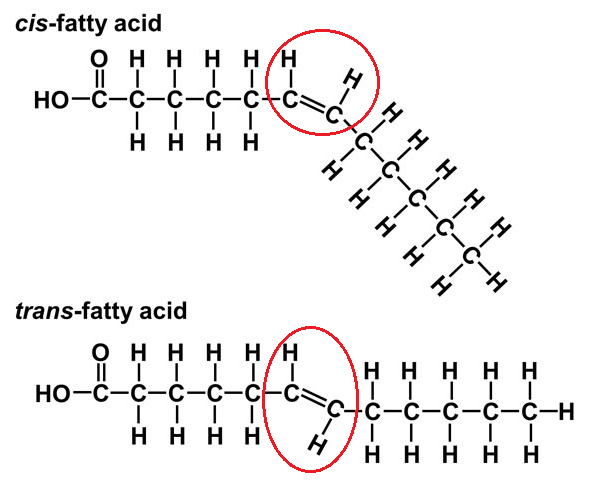
Saturated Fatty Acid -
Single-bonds,
straight,
solid at room temp,
found in animal fat
Unsaturated Fatty Acid -
some are Double-bonded,
not-saturated,
kinks in chains,
liquid at room temp,
better for health,
found in plants
Trans Fat:
Hydrogenation - Industrial process that converts unsaturated fats into trans fats by adding hydrogen (bad for body)
can lead to cardiovascular disease and clogged arteries
found in shortening and margarine
Phospholipid Bilayer:
Phospholipids make up a bilary that surrounds cells
Bilary protects cells and regulates the movement of molecules across it
Proteins
NOT used for energy
Storage
Transport
Regulation - hormones
Movement - muscles
Structure - hair, nails
Disease Protection - antibodies
Enzymes
Chemical Structure: Carbon, Hydrogen, Oxygen, Nitrogen, and sometimes Sulfur (CHON)
Ends in:
Structural proteins: - in
Enzymes: -ase
Monomer = Amino Acid
Amino Acid = 1 Amino Group + 1 R-Group + 1 Carboxyl Group
R-Groups are different for each amino acids; they determine the acid’s property —>
20 different acids (9 which are essential and from food, 11 which are non essential and already made by the human body)

Polypeptide = Polymer of Proteins = Protein
Straight chains of amino acids assembled through dehydration synthesis, held together by Peptide Bonds
Protein Structure
Folded — structure determines function
Protein Folding - a process that takes a polypeptide chain and turns it into a functional 3D shape
Four stages:
Primary (Polypeptide Chain) —>
Secondary (Alpha Helix, Beta Pleat) —>
Tertiary (Combines Helices and Pleats) —>
Quaternary (Multiple Tertiary Structures); not all proteins become quaternary
Denature - occurs when a protein loses shape due to its environment (ex. temperature, salt concentration, pH changes)
renature is possible
why high fevers are fatal
Nucleic Acids
Found in DNA, RNA, and ATP
Stores genetic (hereditary) info
Assists with protein synthesis (carrying instructions for building proteins)
Chemical Structure: Carbon, Hydrogen, Oxygen, Nitrogen, Phosphorus (CHONP)
Nitrogen Base
5-Carbon Sugar
Contains Phosphate functional groups
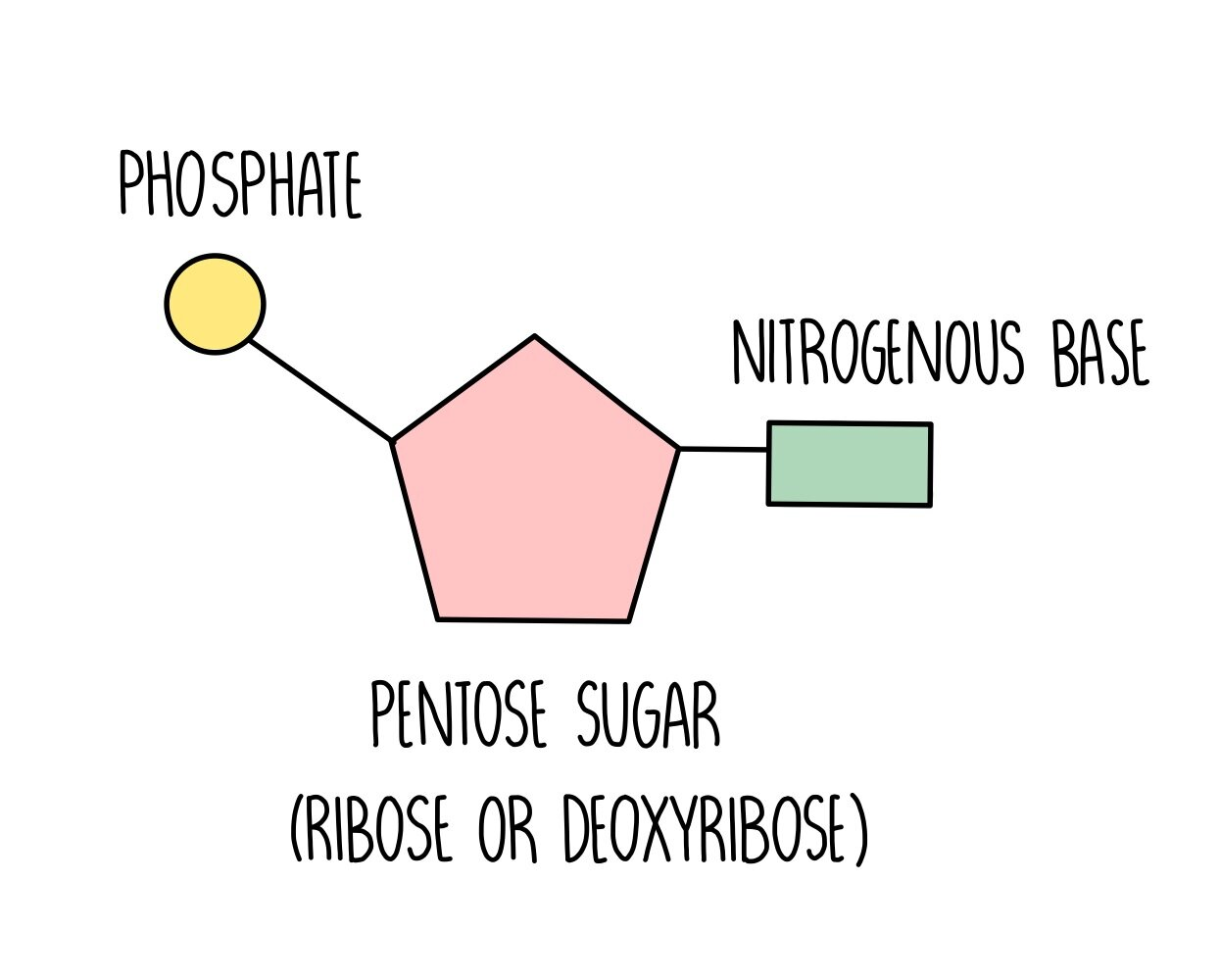
Monomer: Nucleotide
Phosphate
5- Carbon Sugar (BASE)
Nitrogen Base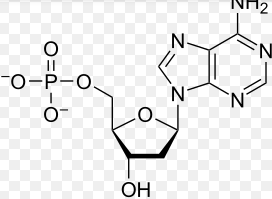
Polymer:
Polynucleotide (Formed by bonding alternate phosphate groups and sugars with phosphodiester bond
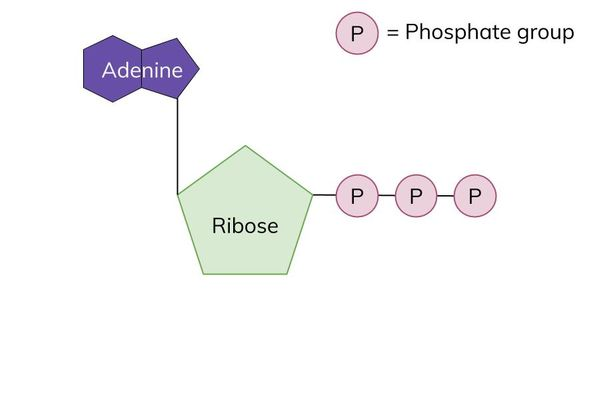
ATP - Adenosine Triphosphate
Storage of energy in high energy bonds
between phosphate groups (phosphate gives ATP charge)
Chemical Reactions & Enzymes
Chemical reactions must occur in organisms so they survive
Enzymes help speed up these reactions
Reactants - molecules that start a chemical reaction
Products- molecules that end a chemical reaction
Exergonic Reaction/Exothermic Reaction
When the change in energy is negative
Energy is released when bonds are broken
Products have less energy
Spontaneous
Endergonic Reaction/Endothermic Reaction
Needs energy to react
Energy is absorbed when bonds are created
Products have higher energy
Change in energy is positive
Slow to start; requires assistance — needs constant energy

Enzymes
Specialized, Reusable Proteins that speed up chemical reactions in cells
Catalyst - speeds up reactions
Structure determines function
Unchanged by reactions
Activation Energy - the amount of energy required to start a reaction
lowered activation energy = working enzyme
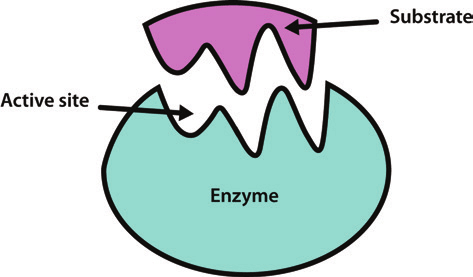
How Enzymes Work -
Lock & Key Model - Each enzyme only fits the shape of its corresponding reactant molecule(s)
(therefore each reaction requires a different enzyme)
Lock = Substrate (what is acted on by an enzyme, the reactant)
Enzymes are typically named after their substrate
Key + Enzyme’s Active Site (region of an enzyme in which a substrate fits in
Steps:
Substrate bonds to enzyme’s active site
Water is needed for hydrolysis; removed for dehydration synthesis
Enzyme catalyzes reaction by lowering activation energy to form/break bonds
New product(s) form and are released
Enzyme is reused again
Factors that Affect Enzymes
Environmental Conditions (temperature, pH)
Adding Substrate —> Increases Reaction Speed
(only up to Saturation Point)
Cofactors -a non-protein compound or ion that is necessary for an enzyme’s function
Coenzyme - an organic molecule that assists the enzyme’s reaction by binding onto its active sites
Enzyme Inhibitors -
Prevents Substrate from Binding
Competitive Inhibitors - molecule that prevents substrate from binding by binding to the active site
Noncompetitive Inhibitor - molecule that prevents substrate from binding by binding to an enzyme’s non-active site and changing its shape
pH
drastically different pH will cancel each other out