solubility
Glossary
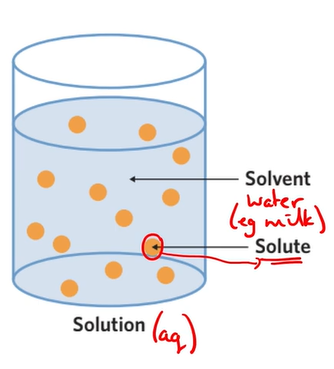
^^Solute^^
A substance that dissolves in the solvent
^^Solvent^^
A substance that has a solute dissolved in it
^^Solution^^
The final product when a solute is dissolved in a solvent
^^Solubility^^
The degree to which a solute dissolves in a solvent.
Aqueous (aq) is a solution when a solute is dissolved in water.
^^HPLC^^
Highly performance/pressure liquid chromatography
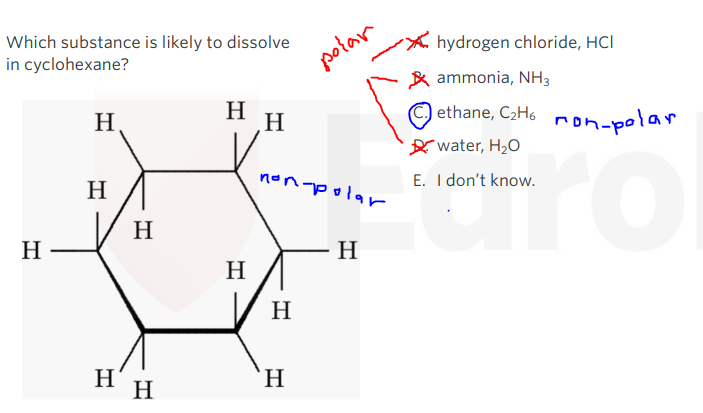
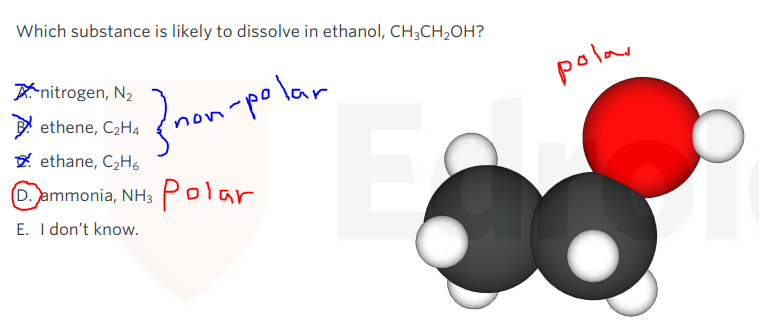
‘like dissolves in like’
- Polar solutes dissovles in polar solvents.
- Non-polar solutes dissolves in non-polar solvents.

^^WORKED EXAMPLES^^
chromatography
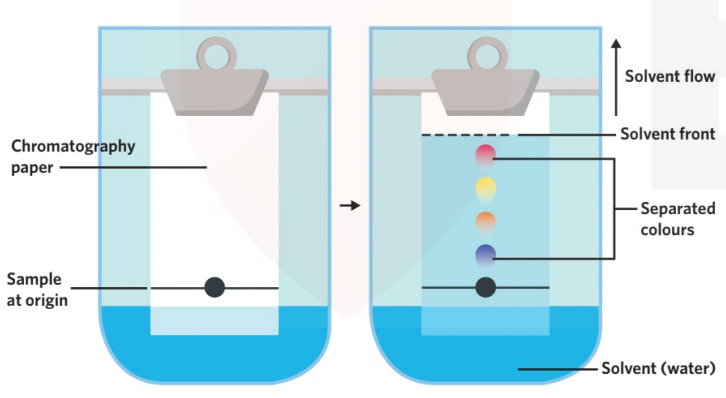
EXPERIMENT EXPLANATION:
- You put a dot on a paper and place it in some water
- Make sure the dot is higher than the water level
- Put a lid on so the water does not evaporate
- Wait for the paper to dissolve the water
- Observe the different colours in the trail the marker created
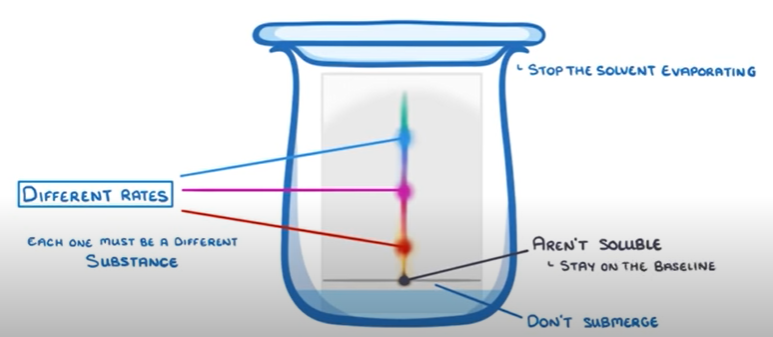
The different colours show the different substances in the marker and their solubility level.
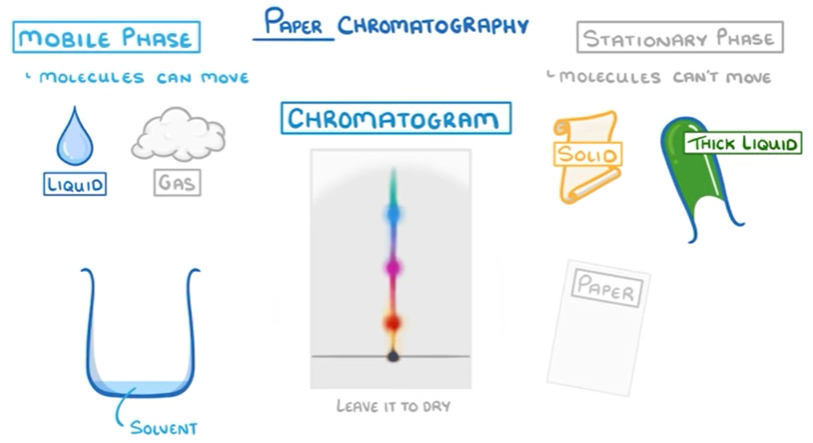
- Mobile phase: the one which moves with the sample. (water)
- Stationary phase: the one which does not move with the sample (paper)
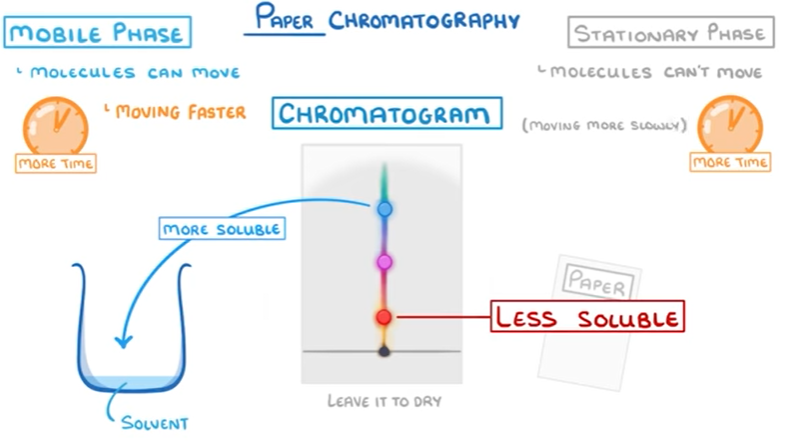
The more soluble substances spend more time in the mobile phase, which is why they move faster.
The less soluble substances spend more time in the stationary phase which is why they move slower.
Temperature & pressure makes it go faster
‘like likes like’, a polar molecule will adsorb a polar substance at a slower rate
like repels each other - a non-polar molecule will adsorb a polar substance at a faster rate
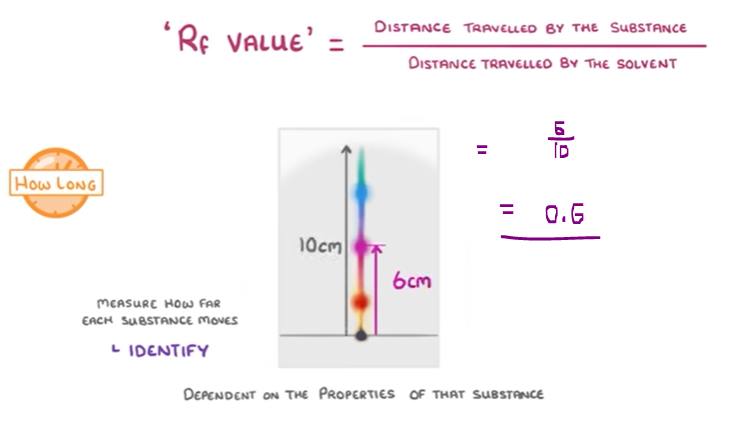
- The substances’ solubility rate is dependent on their properties.
- You can identify the substance by measuring how far it moves.
However, the length of time it was placed in the solvent can increase the measurements.
Therefore, the ‘Rf value’ formula is used to calculate the distance in a ratio.
distance travelled by the compound
Rf Value = --------------------------------------------
distance travelled by the solvent front
- Different type of paper and/or different type of solvent = different Rf value