Molecular Geometry and Bonding Theories Notes
Molecular Shapes
Lewis structures provide bonding and lone pair information but not shape.
Used to predict molecular shapes.
Shape Determination
Bond angles and lengths define shape.
Electron pairs repel; they spread apart to minimize repulsion (VSEPR model).
Electron Domains
Electron domains indicate directions of electron pairs.
A central atom can have multiple electron domains.
VSEPR Model
Best arrangement minimizes electron domain repulsion.
Electron-Domain Geometry
Characterized by number of electron pairs, bonds, and lone pairs.
Molecular Geometry Steps
Draw the Lewis structure.
Determine electron-domain geometry.
Identify molecular geometry using bonded atom arrangement.
Common Electron Domains
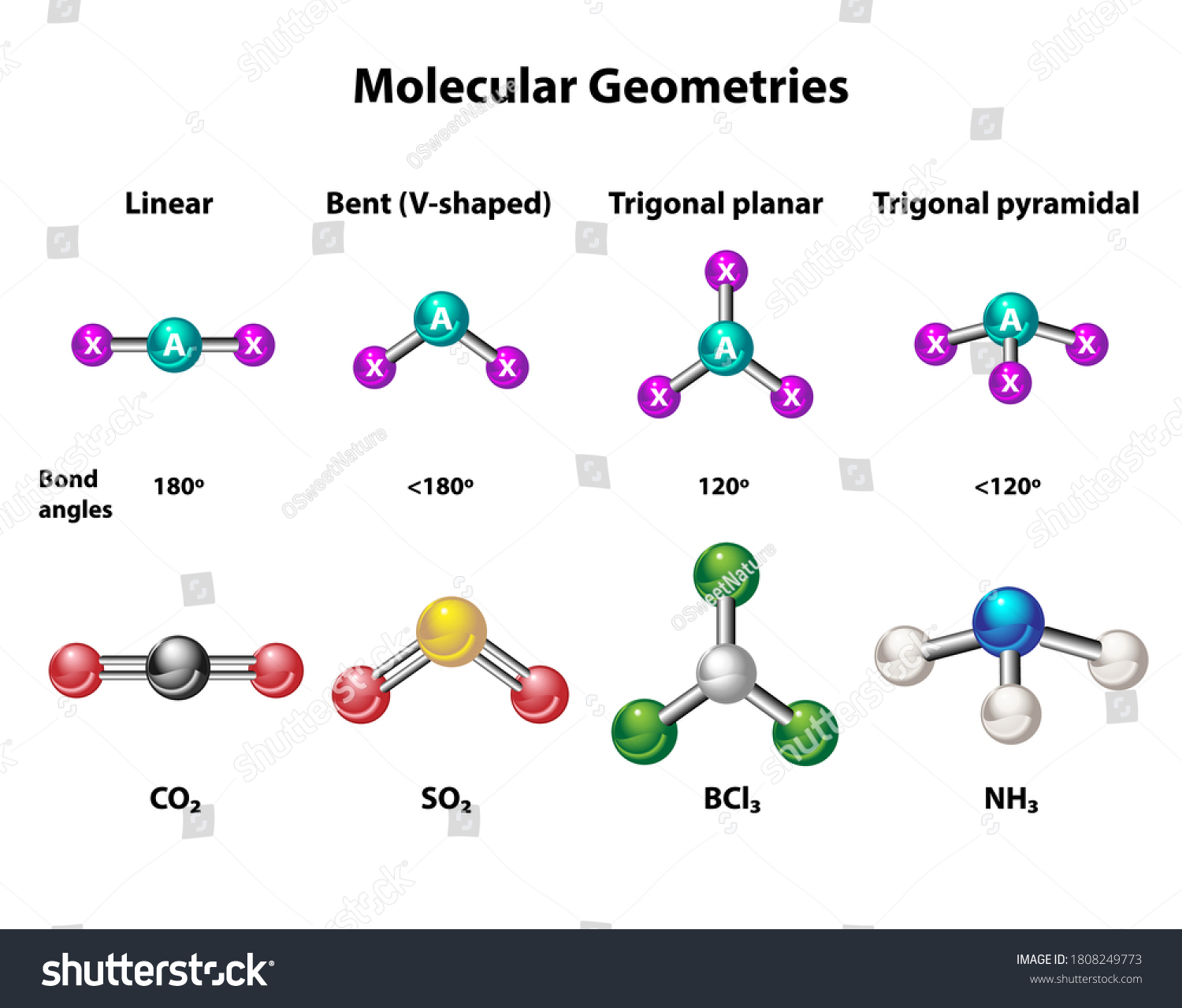
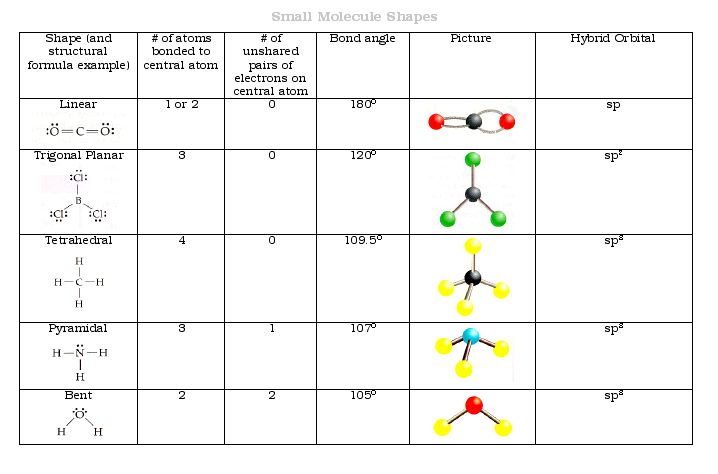
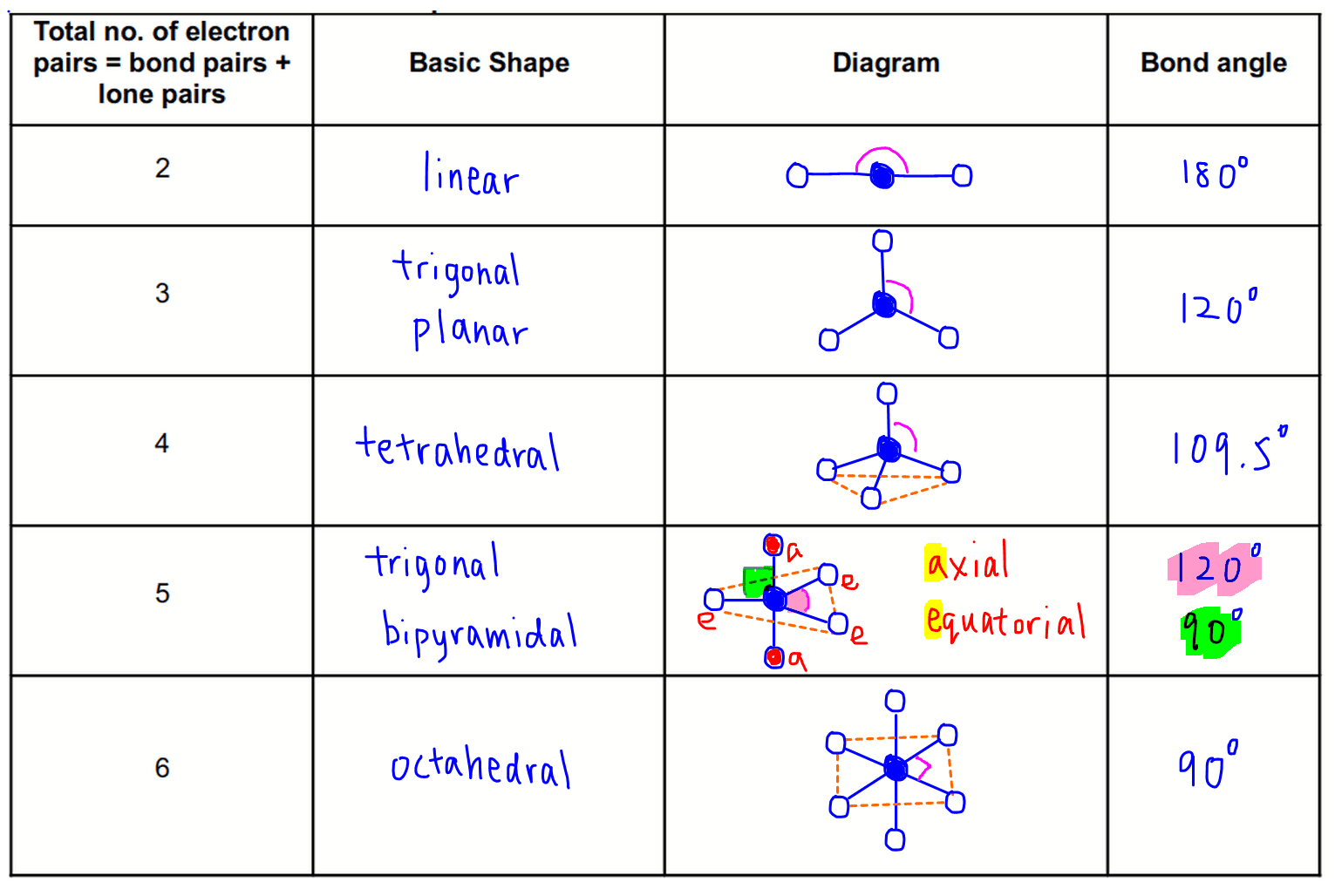
Linear: 2 atoms lead to linear geometry.
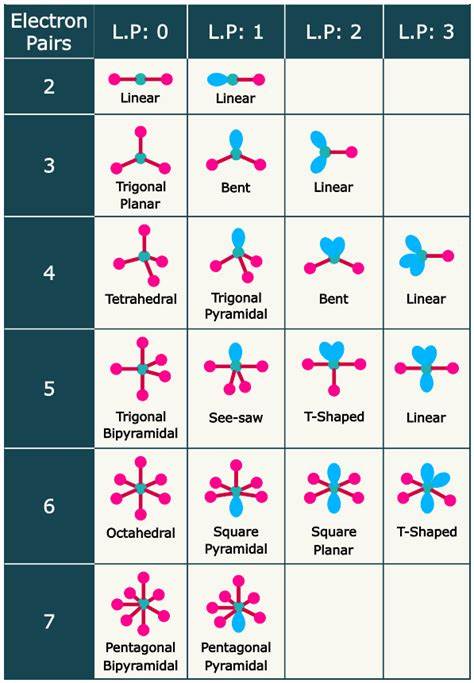
Trigonal Planar: 3 bonding domains (trigonal planar); 1 lone pair (bent).
Tetrahedral: 4 bonding domains (tetrahedral); 1 lone pair (trigonal pyramidal); 2 lone pairs (bent).
Influence of Lone Pairs and Multiple Bonds
Nonbonding pairs compress bond angles due to size.
Multiple bonds exert greater repulsion than single bonds.
Octet Rule
Some elements can expand beyond the octet, allowing 5 (trigonal bipyramidal) or 6 (octahedral) bonds.
Valence Bond Theory
Covalent bonds form through orbital overlaps.
Increased overlap leads to balance between repulsion and attraction.
Hybrid Orbitals
Formed from mixing atomic orbitals, resulting in degenerate orbitals.
Example: Be uses sp hybridization; C uses sp³ hybridization.
Types of Bonds
Sigma (σ) bonds: head-to-head overlap.
Pi (π) bonds: sideways overlap.
Molecular Orbital Theory
Considers wave properties and energy levels of electrons.
Formation of bonding and antibonding orbitals from atomic overlaps.
Bond order calculation indicates bond strength.
Molecular Orbital Characteristics:
-Max 2 electrons per orbital
-Electrons in the same orbital have opposite spin
Electrons in Bonding
Localized electrons are shared between two atoms; delocalized electrons are shared among multiple atoms.
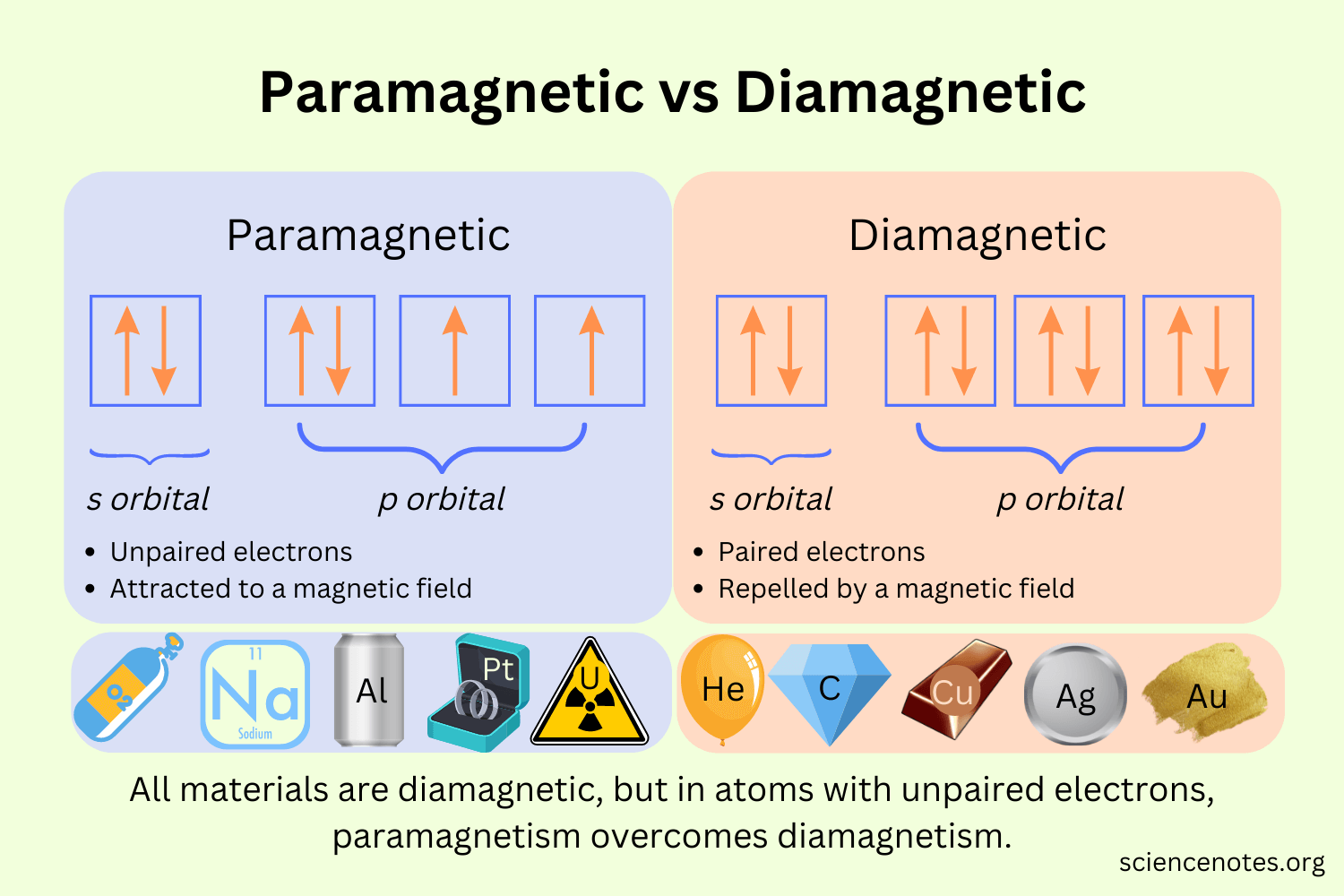
Paramagnetism and Diamagnetism
Paramagnetic: unpaired electrons
Diamagnetic: all paired.
MO Diagrams for Heteronuclear Molecules
Different energy levels affect bonding characteristics.
Overlap: Electrons of two atoms sharing the same space. This can occur in two types:
Sigma (σ) Bonds: Formed by head-to-head overlap of orbitals, allowing free rotation.
Pi (π) Bonds: Formed by sideways overlap of p orbitals, creating a bond that restricts rotation due to the presence of a nodal plane.
The sharing of space between 2 electrons of opposite spins creates a covalent bond
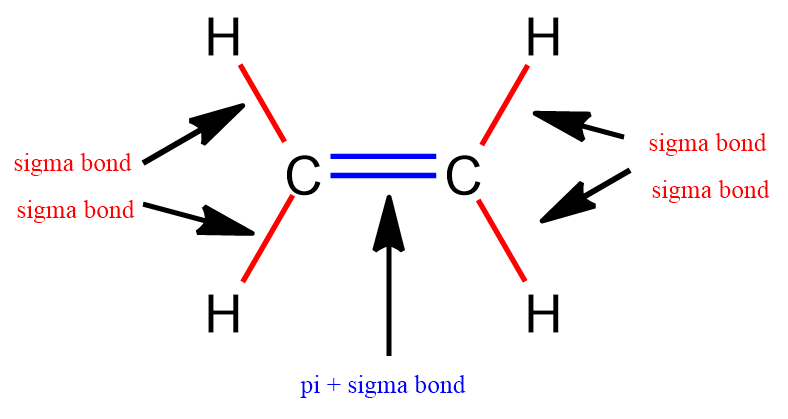
Double Bonds: Consist of one sigma bond and one pi bond, which enhances bond strength and affects molecular shape.
Triple Bonds: Comprise one sigma bond and two pi bonds, resulting in even greater bond strength and further restriction of rotational movement in the molecule.