Emergent Properties of Water
Water is a Polar Molecule
- Hydrogen is bonded through covalent bonds to oxygen atoms
- Oxygen has a greater electronegativity than hydrogen
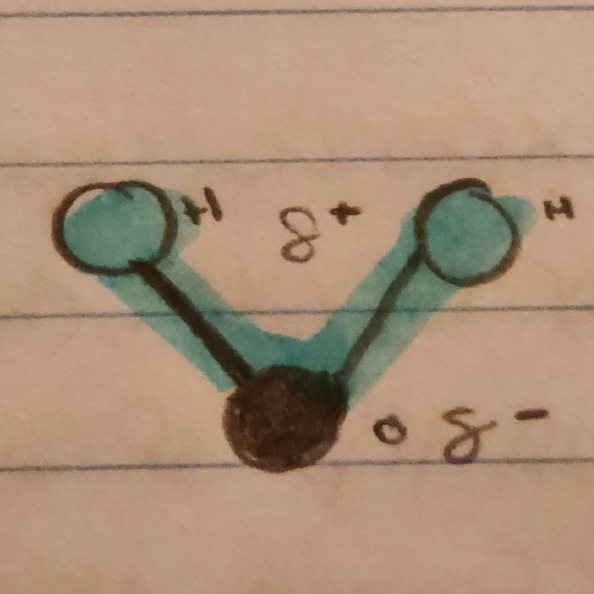
- Water molecules will create hydrogen bonds and organize at a high level of structural organization
Cohesion (like molecules sticking together) Vs. Adhesion (unlike molecules sticking together)
- Allows for xylem sap (water and minerals) to move vertically, up against gravity in plants; phloem flows down (cohesion)
- Allows for water to stay in the veins of plants even without tension (adhesion)
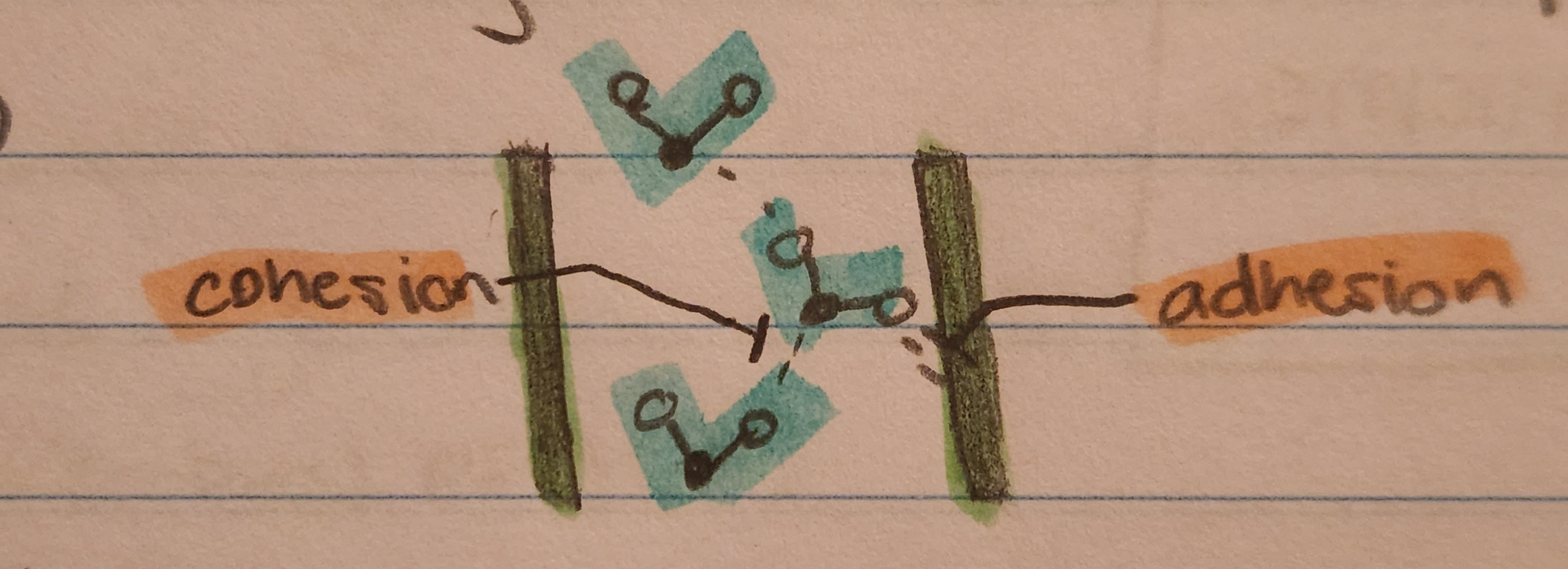
High Surface Tension
- Meaning that it takes greater force to shear or break the surface
- This allows some organisms to live on the surface of the water
Moderation of Temperature
- Water resists quick changes in temp. due to hydrogen bonding and high specific heat
- High Specific Heat: the amount of kinetic energy it takes to change the temperature of 1 gram of a substance by 1 degree
- Oceans act as heat reservoirs, moderating climates
- This helps organisms maintain a homeostatic (internal steady state of conditions) body temperature
- Allows for evaporative cooling; sweat
- High Heat of Vaporization: the amount of heat it takes for liquid to vaporize
Temperature vs. Heat
- Temp. is the average amount of kinetic energy independent of volume
- Heat is the total amount of kinetic energy dependent of volume
Water Expands upon Freezing
- Water is most dense at 4 degrees Celcius
- Ice on top of water creates insulates the water below
- When frozen molecules form a lattice-like structure
Versatile Solvent
- Solvent: dissolving agent
- Attracted to hydrophilic (water-liking) molecules, ionic compounds, and polar molecules
- Not attracted to hydrophobic (water-fearing) molecules and non-polar molecules
- Helps to transport substances in the solution phase