BIOC*2580 - 1
Course Transition
Acknowledgment of midterm results.
Structure shift for the second half of the course:
Transition from discussing proteins to other biomolecules, mainly lipids.
Lipids Overview
Definition of Lipids:
Diverse group of molecules.
Not defined by structure but by chemical properties, primarily hydrophobicity.
Comparison to Proteins:
Proteins defined by specific structural motifs (e.g., amino acids).
Lipids defined by hydrophobic nature, requiring nonpolar solvents for dissolution (e.g., chloroform and methanol mixture).
Major Functions of Lipids
Primary functions include:
Energy storage:
Triacylglycerols (triglycerides) are main energy storage form in organisms.
Formation of biological membranes:
Lipids involved: phospholipids and sterols.
Additional roles in metabolism: signal transduction, enzyme cofactors, etc.
Types of Lipids to be Discussed
Fatty Acids:
Building blocks for complex lipids.
Triacylglycerols:
Storage form of fats and oils.
Phosphoglycerides:
Major components of cell membranes.
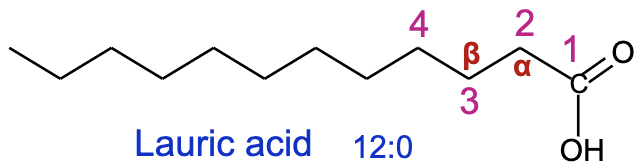
Fatty Acids
Structure:
Composed of carboxylic acid (COOH) and a hydrocarbon chain (length varies from 4 to 36 carbons).
Types based on saturation:
Saturated: no double bonds.
Unsaturated: one or more double bonds.
Monounsaturated: one double bond.
Polyunsaturated: multiple double bonds.
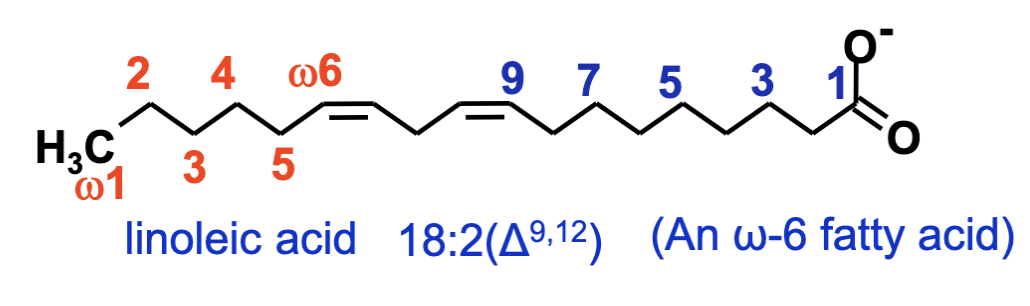
Numbering System:
Carbon atoms labeled starting from carboxylic carbon (beta, alpha, and omega carbons identified).
Properties of Fatty Acids
Reactivity:
Carboxylic acid group is reactive; hydrocarbon chain is nonreactive.
Double Bond Configurations:
Cis (bent structure) vs. Trans (linear structure) configuration:
Cis introduces kinks; Trans aligns in straight configuration affecting physical properties.
Methylene-bridged pattern for polyunsaturated fatty acids.
Common Features of Fatty Acids
Majority have even number of carbons (rare odd numbered ones).
Typically straight chains without branches.
Cis vs. Trans Configuration:
Most naturally occurring fatty acids are in cis form, affecting their melting points and structures.'
Common Fatty Acids (Saturated)
Let My Pal Stay Arounds
Laurate (C12:0) - a melting point of about 44°C, commonly found in palm kernel oil and coconut oil. bay, laurel
Myristate (C14:0) - a melting point of approximately 54°C, prevalent in dairy products and found in nut oils, contributing to their creamy texture. myrtle, nutmeg
Palmitate (C16:0) - a melting point of around 63°C, commonly found in palm oil and animal fats, and plays a significant role in the structure of cell membranes. palm
Stearate (C18:0) - a melting point of about 69°C, found in various animal fats and vegetable oils, and is important for energy storage and metabolic processes. tallow
Arachidate(C20:0) - A saturated fatty acid with a melting point of approximately 76°C, primarily found in some animal fats and certain vegetable oils, and is involved in cellular signaling and membrane stability. peanut
Naming Fatty Acids
Nomenclature:
First number indicates total carbon atoms.
Colon separates number of double bonds.
Superscripts indicate position of the double bonds from the carboxylic carbon (delta system).
Omega system identifies first double bond from the methyl carbon.
Physical Properties of Fatty Acids
Melting Point and Solubility
Saturated fatty acids have higher melting points due to tight packing.
Unsaturated fatty acids (cis) have lower melting points due to kinks leading to less packing.
Trans fatty acids behave more like saturated due to linear structure from trans double bonds.
The longer the chain the higher the melting point, the lower the solubility
Derivatives of Fatty Acids
Carboxylic acids can combine with alcohols to form esters.
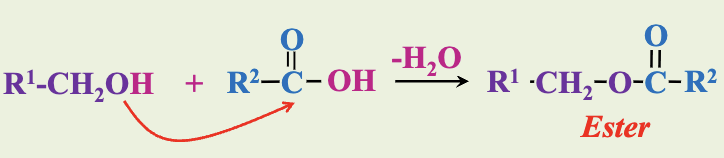
Carboxylic acids can combine with acids to form acid anhydrides.
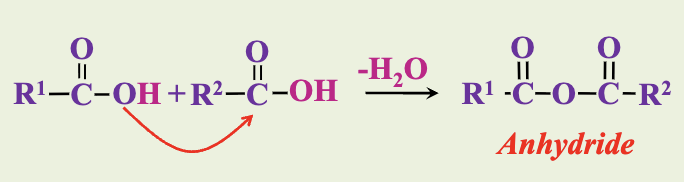
Triglycerides (Triacylglycerols) (TAGs; Fats and Oils)
Formation:
Composed of glycerol and three fatty acids (ester bonds formed through reactions of carboxyl groups with alcohols).
Properties:
Fatty acids not found free; instead, found in triglycerides.
Triglycerides are HIGHLY hydrophobic (polar caboxilic acids are tied as esters)
They come as mixed triglycerides (different fatty acids) vs. simple triglycerides (same fatty acids).
Melting point is dependent on length and degree of saturation of fatty acid constituents
Source of Energy:
Commonly stored in the body as triacylglycerols, providing energy.
Physical State of Triglycerides
At room temperature:
Fats (solid)
Oils (liquid)
Solid fats usually higher in saturated fats, while oils have higher unsaturated fats.
Examples from Nature:
Olive oil, butter, beef fat vary in composition affecting states and melting points.
Conclusion
Triglycerides form the bulk of dietary fats and energy storage molecules.
Importance of understanding complex lipids for broader biochemical studies in metabolism and cell structure.