Chemical Equilibrium Notes
Discriminate between open or closed chemical systems.
Open System: Energy and matter can be transferred in/out.
Closed System: Energy can be transferred in/out.
Identify that physical changes are usually reversible, whereas only some chemical reactions are reversible.
Physical Changes: Are usually reversible
Chemical Reactions: Only some are reversible
Symbolise equilibrium equations using ⇋ in balanced chemical equations.
⇋: Equilirbibrium reaction (reversible reaction)
Explain observable properties and the characteristics of physical and chemical systems in a state of equilibrium.
Physical Equilibrium: A state where phase changes occur at equal rates so macroscopic properties (like temperature and phase amounts) stay constant.
Chemical Equilibrium: A dynamic state where forward and reverse reaction rates are equal, keeping reactant and product concentrations constant.
Explain that, over time, physical change and reversible chemical reactions reach a state of dynamic equilibrium in a closed system, with the relative concentrations of products and reactants defining the position of equilibrium.
Dynamic Equilibrium: In a closed system, physical changes and reversible chemical reactions eventually reach dynamic equilibrium, where the forward and reverse processes occur at equal rates and the relative amounts of reactants and products determine the position of equilibrium.
Explain the reversibility of chemical reactions by considering the activation energies of the forward and reverse reactions.
Reversibility of Reactions: A reaction is reversible because particles can gain enough energy to overcome the activation energy barriers in both the forward and reverse directions, allowing products to convert back into reactants.
Analyse data and interpret graphical representations of relative changes in the concentration of reactants and product against time, to determine the position of equilibrium.
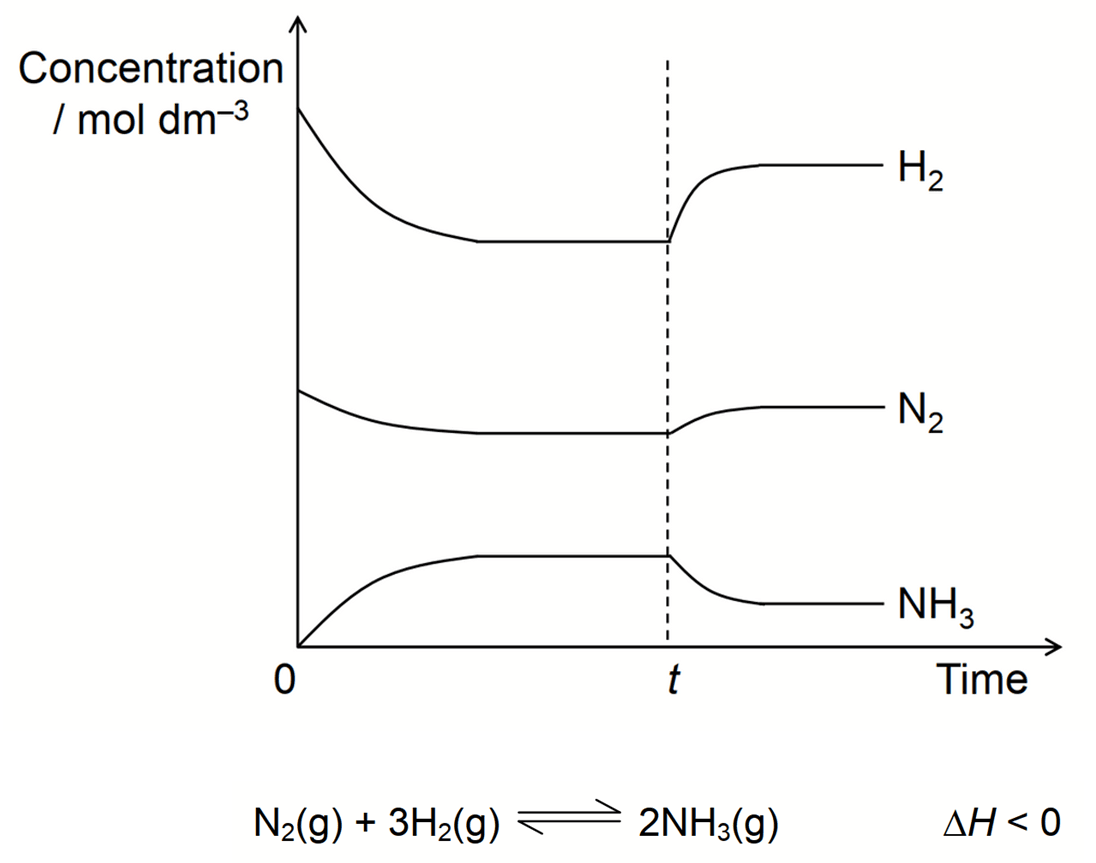
Flat Line: Means concentrationsrates of reaction are constant as there is no change in conc meaning that equilibrium is reached.