Organic Chemistry - Chem Honors
Organic vs. inorganic
organic chemistry is the study of carbon containing compounds
Ex. fuels, plastics, alcohols, fats, proteins, ethers, sugars, aldehydes
look for carbon in molecular formulas
exceptions are CO, CO2, and carbonates (XCO3) - all inorganic
Properties of inorganic
ionic bonds with crystal lattice
polar compounds
strong IMFs
fast rate of reaction
high melting and boiling point
solids at STP
low vapor pressure
Properties of organic
covalent bonds with molecules
nonpolar molecules
weak IMFs
slow rate of reaction
low melting and boiling point
gases (some liquids) at STP
high vapor pressure
Structure
carbon has 4 valence electrons so it forms 4 covalent bonds
can bond to itself and many other elements
can have single, double, or triple bonds
SP hybridization so the 4 electrons act the same in one orbital create not normal looking lewis dot

carbon can form long, repetitive chains called polymers
unusual length and elasticity of polymers due to great length and high molecular weight
What’s made of carbon?
living things (cellulose, DNA)
fuels (fossil, bio)
paints, coatings, and adhesives
industrial applications (bakelite)
plastics (name anything and it’s most likely plastic)
synthetic fibers (nylon)
Hydrocarbons
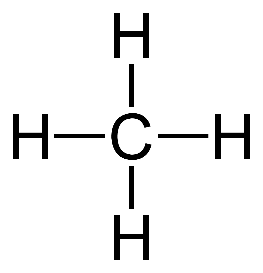
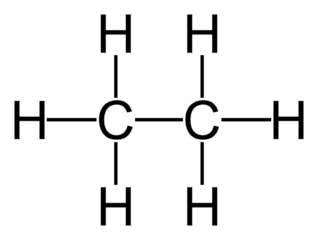
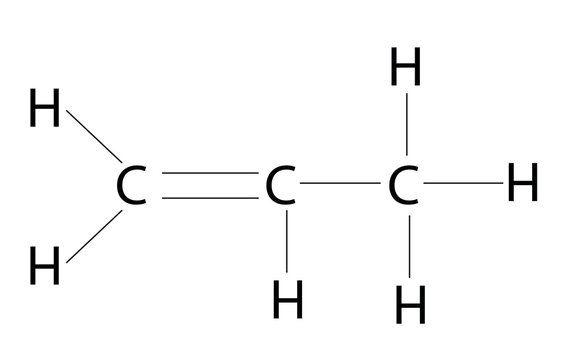
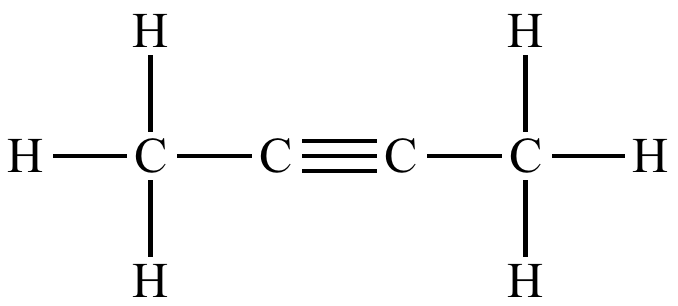
molecules that only contain carbon and hydrogen in their formulas
can have single, double, or triple bonds (Table Q)
alkanes have single bonds
alkenes have at least one double bond
alkynes have at least one triple bond
saturated hydrocarbon means it contains max number of hydrogen atoms possible
alkanes are saturated
basic naming
prefix (number of electrons)
suffix (type of bonds)
use Table P and Q
Simple hydrocarbons
butane - C4H10
ethene - C2H4
propyne - C3H4
pentene - C5H10
hexyne - C6H10
propane - C3H8
Methane | Ethane | Propene | Butyne | |
Chemical formula | CH4 | C2H6 | C3H6 | C4H6 |
Structural formula |
|
|
|
|
Condensed structural formula | CH4 | CH3CH3 | CH2CHCH3 | CH3CCCH3 |
General formula | CnH2(n) + 2 | CnH2(n) + 2 | CnH2(n) | CnH2(n) - 2 |
Isomers
same molecular formula but different structural formula
Ex. straight chain butane - C4H10
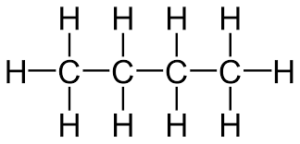
has bp. 0.5 degrees Celsius
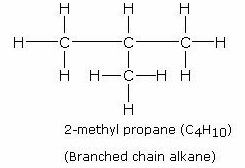
branched chain methyl propane - C4H10
has bp. 10 degrees Celsius
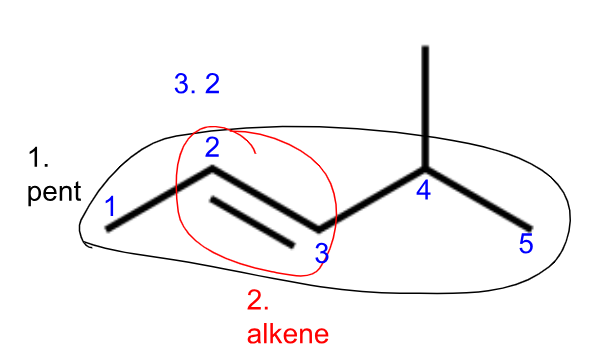
Naming hydrocarbons (IUPAC)
Last name
1. find # of carbons in parent chain/longest continuous group of carbons
# of C = prefix from Table P
2. find single, double, or triple bonds
bond type = suffix from Table Q
3. note location of any multiple bonds
label numbers in direction that puts lowest number on the carbon with multiple bonds
use di- or tri- if there’s more than one multiple bond
this # is listed before the last name (2 in 2-Pentene)
Ex.
Naming hydrocarbons (IUPAC)
first name
1. name side chains branching off of parent chain
count # of C in side chain
name - prefix from Table P + “yl” (1 carbon = methyl)
side chains are listed in alphabetical order (ethyl before methyl)
use di- or tri- as needed
2. note location of side chains branching off of parent chain
label numbers in direction so branches get lowest number
if C already numbered due to having multiple bonds, use that numbering (if you have double or triple bonds within parent chain)
# is listed before first name (4 in 4-methyl)
Ex.
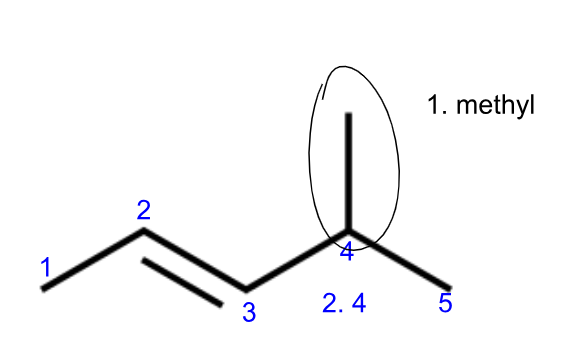
whole name becomes 4-methyl-2-pentene
Branched hydrocarbons
Ex. 2, 2, 4 trimethyl pentane
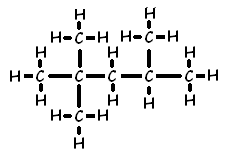
Ex. 2, 3-dimethyl hexane
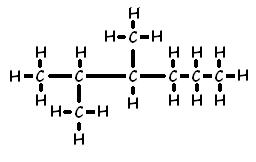
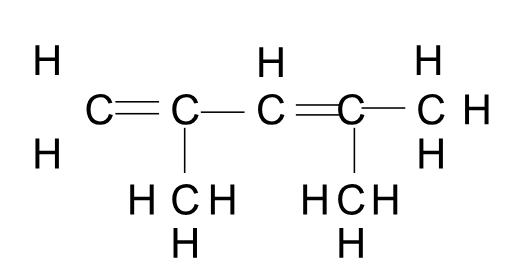
Ex. 2, 4-dimethyl-1, 3-pentadiene
Cyclic hydrocarbons
all have single bonds
Ex. cyclopropane - triangle
Ex. cyclobutane - square
Ex. cyclopentane - pentagon
Ex. cyclohexane - hexagon
Aromatics
cyclic hydrocarbons with double bonds
electrons shared evenly around the ring
structure means very different properties compared to other compounds
fragrant - cloves, vanilla, almond essence
each carbon equally sharing electrons and experiences “resonance”
Functional groups
specific groups of atoms or bonds within molecules that are responsible for the characteristic chemical properties and reactions of those molecules
create polar molecules which dissolved in water
alcohols are soluble in water
Table R
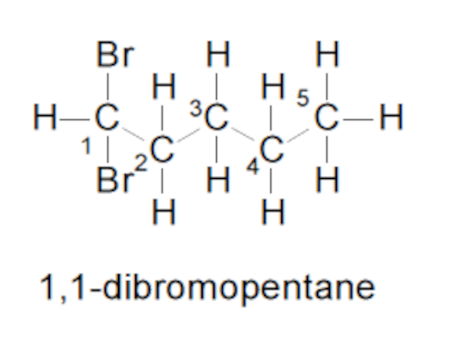
Halides
Ex. 1,1 dibromopentane
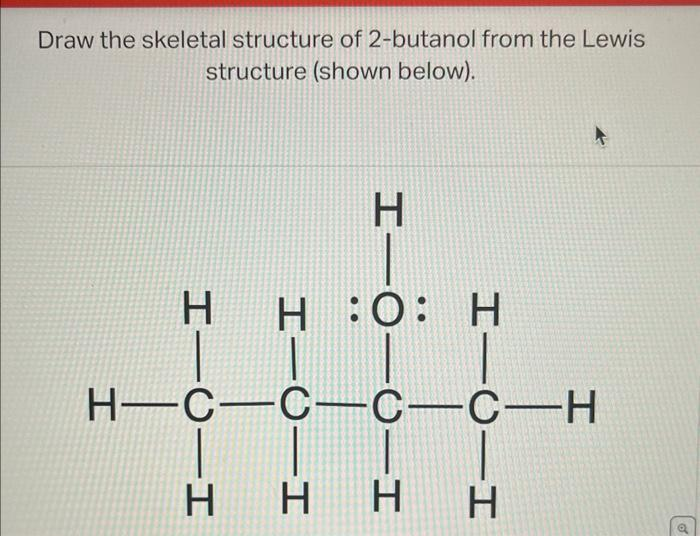
Alcohols
Ex. 2-butaonol (secondary alcohol bc OH- is on 2nd carbon)
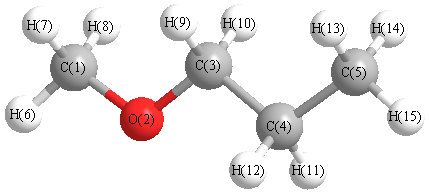
Ethers
Ex. Methyl propyl ether (shorter group first)
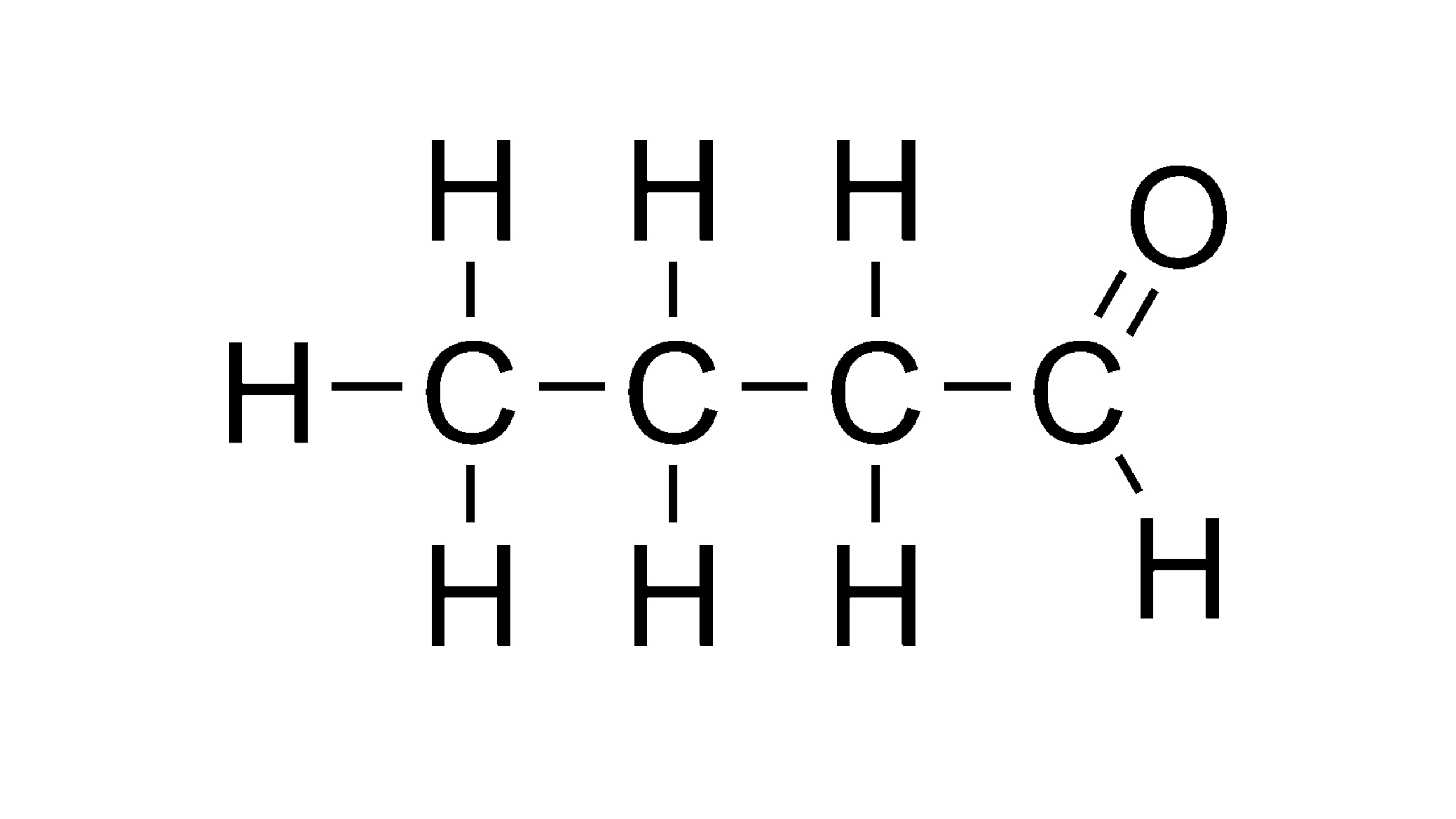
Aldehydes
Ex. Butanal (double bond O and H always at first or last carbon)
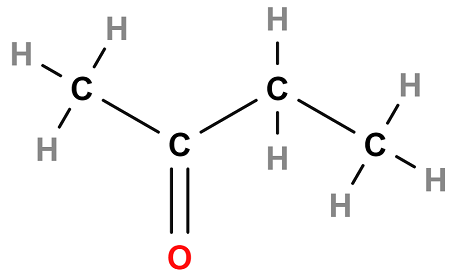
Ketones
Ex. 2-butanone
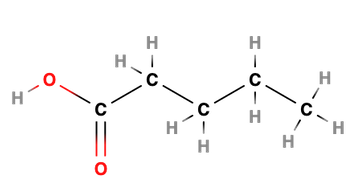
Organic acids
Ex. Pentaonic acid (always at first or last carbon)
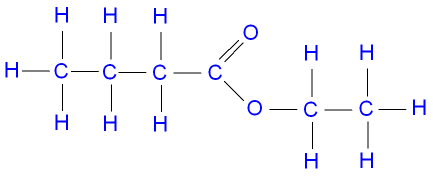
Esters
Ex. Ethyl butanoate (one on oxygen goes first)
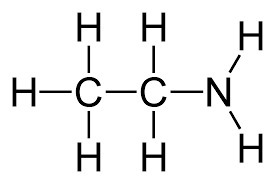
Amines
Ex. Ethanamine
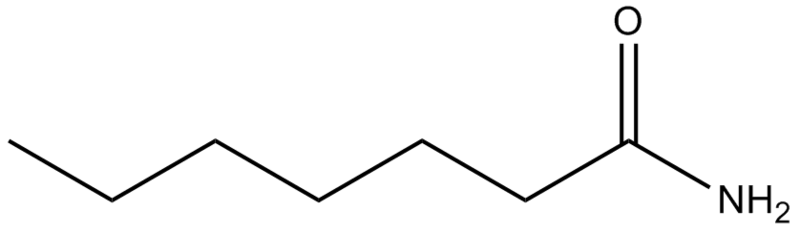
Amides
Ex. Heptanamide
Reactions
addition
always unsaturated hydrocarbon (double or triple bonds)
break a multiple bond and add in two new atoms
Ex. C
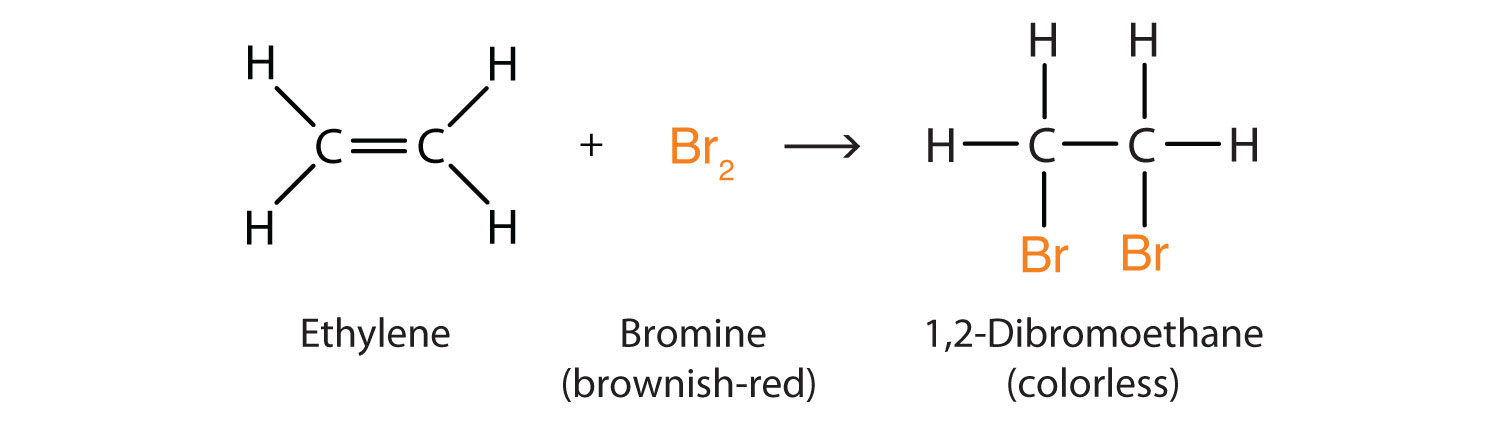
substitution
always saturated hydrocarbon (single bonds)
single replacement of one hydrogen with another atom
Ex. C2H4 +Br2 → C2H4Br2
combustion
oxidation of a hydrocarbon
needs O2
Ex. CH4 +2O2 → CO2 +2H2O
fermentation
sugar reacts with an enzyme as a catalyst (zymase) to make alcohol and CO2
Ex. C6H12O6 → CO2 + 2C2H5OH
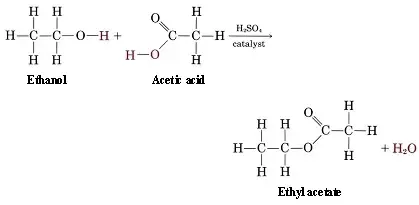
esterification
organic acid and alcohol makes an ester and water
Ex. CH3COOH + C2H5OH → C4H8O2 + H2O
saponification
hydrolysis of fats by bases create an alcohol and soap
soap has polar and nonpolar end
Ex. C21H99O6 + 3NaOH → C3H9O3 + 3C16H31O2Na
polymerization
monomers join to make polymers (repeating units)
usually joining unsaturated molecules
Polymers
natural and synthetic
types of polymers are linear, branched, cross-linked, network
linear held by London Dispersion forces
branched more flexible and less dense because of random branches
cross linked help by covalent bonds
thermosetting - once it’s set/cooled, shape won’t change
network has lots of crosslinks making it tight and not flexible
Condensation polymerization
two monomers join to make a big molecules and water
usually joining saturated molecules