102.12 pH
PH starts with water
PH stands for potential hydrogen and is numerical measurement that indicates the acidity or alkalinity of water-based substance.
If pH products are not within a certain pH range, they can be damaging and possibly even deadly.
The breaking apart of the H2O molecules will always result in a 1:1 ratio of positive H+ ions and negative OH- ions.
That is why pure water is considered a neutral substance.
ACIDS AND ALKALIS
Water is all around us and therefore, so are acids and alkalis.
An acid is a substance that increases the concentration of hydrogen ions when placed in water because it donates, or releases, H+ ions.
An alkali decreases the concentration of hydrogen ions when placed in water bc it accepts or combines with H+ ions.
When acids and alkalis are brought together, they neutralize each other.
Acids and Alkalis are substances that change the balance of hydrogen and hydroxide ions in water.
Acid Solution- a solution is acidic if it has more positive hydrogen ions than negative hydroxide ions.
Neutral Solution- when a solution has an equal number of positive and negative ions, it is neutral.
Alkaline Solution- A solution is alkaline if it has more negative hydroxide ions.

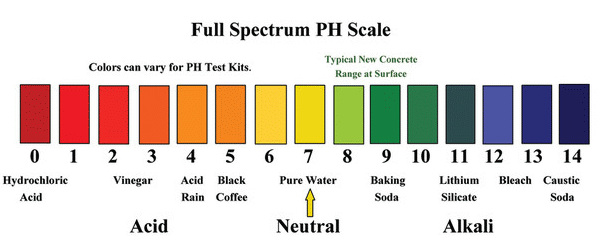
The pH Scale.
The scale ranger from 0 to 14. Seven is the midpoint, indicating a neutral substance.
PH values below 7 indicates more acidity. Higher concentration of H+ions than OH-.
PH values above 7 indicates more alkalinity. Higher concentration of OH-ions than H+ions.
The closer you get to 0, the stronger the acidity.
The closer you get to 14, the stronger the alkalinity
PH balanced refer to 4.5-5.5 healthy Hair, skin & nails
There are three main ways to measure pH: pH mater, Indicator liquid and indicator paper.
Blue litmus paper turns red in an acid.
Red litmus paper turns blue in an alkali.