AP Chemistry Unit 5 Thermochemistry
Energy: capacity to do work or to produce heat.
Law of conservation of energy: energy can be converted from one form to another but can be neither created nor destroyed.
Potential energy: energy due to position (higher it is more potential energy), composition, and attractive/repulsive forces.
Kinetic energy: energy due to the motion, mass (m), and its velocity v: KE = 1/2mv²
Frictional heating (friction): when object transfers kinetic energy to the surface as heat, which causes the surface to increase in temperature, while slowing the object down.
Difference between heat and temperature: temperature is a property that reflects the random motion of the particles in a particular substance (like when you increase the temperature for a gas). Heat involves the transfer of energy between two objects due to a temperature difference.
Work: force acting over a distance; Two ways to transfer energy: through work and through heat.
The pathway is the vehicle that work and heat is dependent on when transferring the total energy; these amounts will differ due to the conditions of the pathway. But, the energy change is independent of the pathway, and the total energy transferred will be constant.
State function: property of the system that depends only on its present state. It does not depend on how it gets got to its present state.
A change in the state function (property) from one state to another is independent on the pathway taken between the two states.
When two objects at different temperatures are in contact, they will eventually reach the same intermediate temperature. For example, when an ice pack touches skin, the energy is transferred as the initially faster particles decrease in motion in the skin and the initially slower molecules in the ice pack increase in motion. Soon, both will reach thermal equilibrium: average kinetic energy = same temp.
When two gases are placed in adjoining chambers with different temperatures, the particles with higher initial velocity slow down, while the particle with the lower initial velocity speeds up. Soon, both gases will have equal kinetic energy, temperature, and average velocity.
System: the part of the universe on which we wish to focus attention; for example in a calorimeter, the system is the molecules being measured.
Surroundings: everything else in the universe; in a calorimeter it would be the container or thermometer, etc.
When a reaction is exothermic; energy flows out of the system
When a reaction is endothermic, energy flows into the system, and absorbs energy from the surroundings.
The energy gained by the surroundings is equal to the energy lost by the system.
In any EXOTHERMIC reaction, some of the POTENTIAL energy stored in the chemical bonds converts to THERMAL energy (random kinetic energy) via heat. Therefore, this LOWERS the POTENTIAL ENERGY from the REACTION SYSTEM, and the reactants have higher potential energy than the products.
This is the verse for an endothermic reaction. The energy that flows into the system as heat is used to INCREASE the POTENTIAL energy of the system. So, the PRODUCTS have HIGHER potential energy than the reactants.
Themodynamics: study of energy and its interconversions (changes)
First Law of Thermodynamics: Also known as the law of conservation of energy, “The energy of the universe is constant.”
The internal energy, or E, of a system can be found by the sum of the kinetic and potential energies of the “particles:” in the system. Can either be changed by flow of work, heat, or both.
The equation that models the internal energy of a system is:
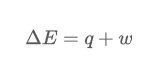
To model thermodynamic quantities, one uses a number and sign for both heat (q) and work (w). The number shows the magnitude of the change, and the sign shows the flow.
When the sign is positive, it shows an endothermic process, and the system’s energy is increasing. Negative is exothermic, and the system's energy is decreasing.
Work in thermodynamics and chemical processes is usually done BY a gas (expansion) or work done TO a gas (compression).
When gas is expanding, the volume is positive because it is increasing, and the work is doing work on the surroundings and flows out of the system, so w will be negative
When gas is compressed, the volume is negative because it is decreasing, and the work is flowing into the system.
When w is positive, work is done ON the system. But, when w is negative, work is done BY the system.
When calculating the work in a system where gas is expanding and compressing the volume against external pressure, the equation used is
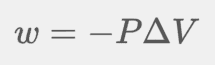
Enthalpy can be found and defined by H = E + PV. E is the internal energy of the system, P is the pressure of the system, and V is the volume of the system. It is also a state function.
At constant pressure, where only work is allowed, the change in enthalpy of the system is equal to the energy flow as heat. This is modeled by the change of H = Hproducts - Hreactants.
If the change of heat is positive, it is endothermic, and if the enthalpy is negative, it is exothermic.
Calorimetry is based on observing the temperature change when a body absorbs or discharges energy as heat.
Heat capacity of a substance is how much energy it takes to raise it's temperature by one degree. It is modeled by this equation:
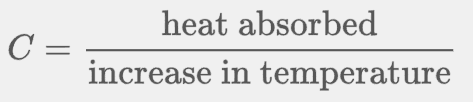
Specific heat capacity is when the heat capacity is given per gram of substance
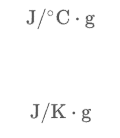
It is molar heat capacity when the heat capacity is given per mole of the susbtance.
Constant-pressure calorimetry is used to determine the changes in enthalpy for reactions in a solution/system.
Standard enthalpy of formation is the change in enthalpy that accompanies the formation of 1 mole of a compound from its elements in their standard states. When writing reactions in their standard state, products are always given per 1 mole of product in it’s standard state.
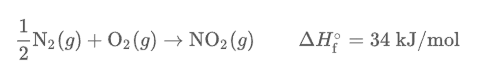
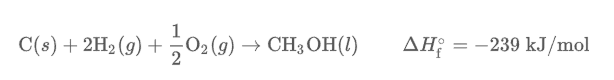
The standard conditions for an element to exist in a standard state is 1 atmosphere and 25 degrees Celcius.
When using standard enthalpies of formation, enthalpy can be found by the sum of standard enthalpies of the products minus the sum of the standard enthalpies of the reactants.
Enthalpy can also be found by the sum of the bonds broken minus the sum of the bonds formed. When bonds are broken, energy must be added to the system, which leads to an endothermic process. This means the energy is positive, whereas bonds being formed is negative. This is because energy is being released to form a bond, which is an exothermic process.
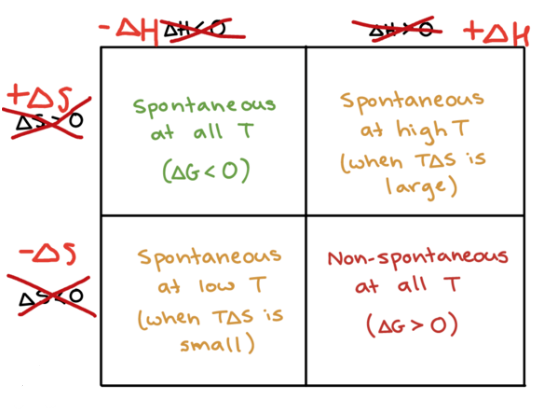
A process is spontaneous if it occurs without outside intervention, such as rusting of metal. Spontaneity is influenced by entropy which can tell us the direction in which the process will occur, but does not determine how fast or slow the process is.
The driving force for a spontaneous process is an increase in the entropy of the universe.
Entropy is the measure of molecular randomness or disorder. In states of matter, the measure of entropy increases from solid to gas, with gas having the highest entropy.
The Second Law of Thermodynamics is that the entropy of the universe is increasing. This contrasts with the first law where energy of the universe is being conserved, but entropy is not.
We know that the universe is comprised of a system and surroundings. So, we can represent the change in the entropy of the universe as:

To know if a given process will be spontaneous, the change in the entropy of the universe must be POSITIVE because nature moves towards entropy.
Also, the change of the entropy of the universe is NOT a state function. Remember, a state function is where the initial and final changes matter, and not how it gets there.
There are two important characteristics of entropy changes in the surroundings: the sign and magnitude.
The sign is dependent on the direction of heat flow.
Entropy changes in the surroundings are determined by heat flow. When heat is transferred out from a system in an exothermic process, it increases the entropy of the surroundings because the energy flow increases the random motions in the surroundings.
The magnitude of the entropy change of the surroundings is dependent on the temperature.
When the components of the change of the entropy of the universe are in opposition in favorability (one is negative, other is positive), the temperature controls the situation.
Due to the idea of heat flow, temperature controls the entropy of the universe and surroundings. If the temperature of the surroundings is very high, an X amount of Joules would not make much of a difference to the random order of the atoms in the surroundings. However, if the surroundings is at a very low temperature, and the atomic motion is slow, the energy would cause a large percent change in the motions.
THEREFORE, at LOWER temperatures, the impact of heat to or from the surroundings will be greater.
With these two characteristics combined, the change in the entropy of the surroundings results in the following reaction that takes place under conditions of constant temperature in KELVIND and pressure:
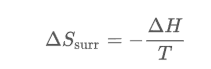

Another way to find the change of entropy in the universe is

Which can be derived from the equation
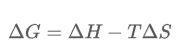
When divide both sides by T, you can get this:
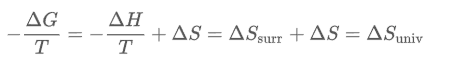
As you can see, a negative change in free energy results in a positive change of entropy of the universe, which results in a spontaneous reaction.
Entorpy is also affected by the number of molecules in a compound. If there is less molecules in a system, there is a fewer chances of collision and therefore less disorder. The more complex the molecule, the higher the standard entropy value.
When a reaction involves gaseous molecules, the change in positional entropy is dominated by the relative numbers of molecules of gaseous reactants and products.
The third law of thermodynamics is the entropy of a perfect crystal at 0 K is zero. This is because every particle is in place, so it not possible for the molecules to be in motion. This makes entropy the only tehrmodynamic characteristic of a system to have an assigned absolute entropy values. Enthalpy and free energy can not be determined.

In chemical reactions, the temperature change is for the surroundings. So endothermic reactions are negative delta T, and exothermic reactions are positive delta T.
