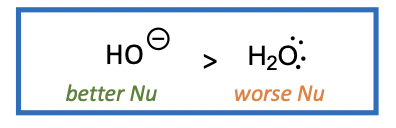Organic Chem - Alkyl Halides
What are Organic Halogen compounds?
• Organic Halogen compounds contain one or more halogens (X = halogen) combined with carbon (and other elements).
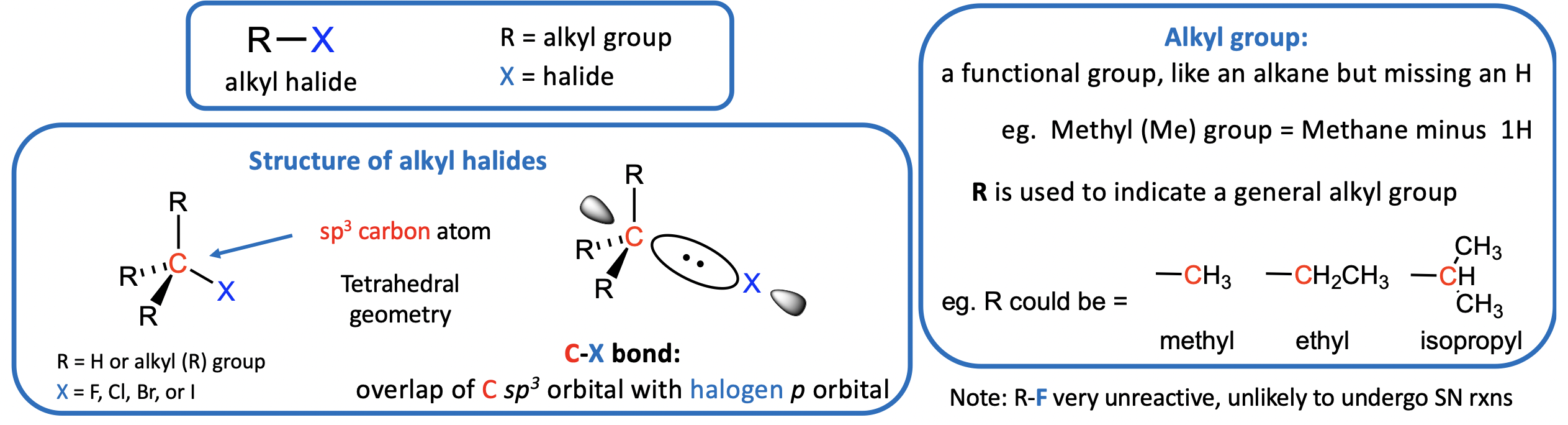
What are Alkyl Halides (RX)? (also known as halogenoalkanes)
• Are a subset of organic halogen compounds
• large class of compounds that contain an alkyl group bound, via a sp3 carbon atom, to a halogen atom (X = F, Cl, Br, or I)
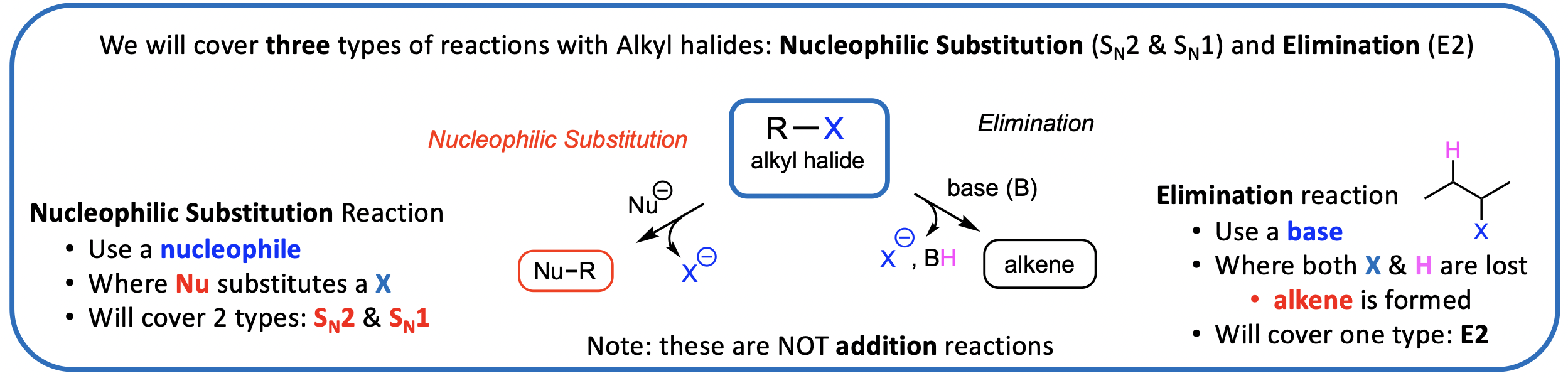
Alkyl Halides: types of reactions and why they’re useful
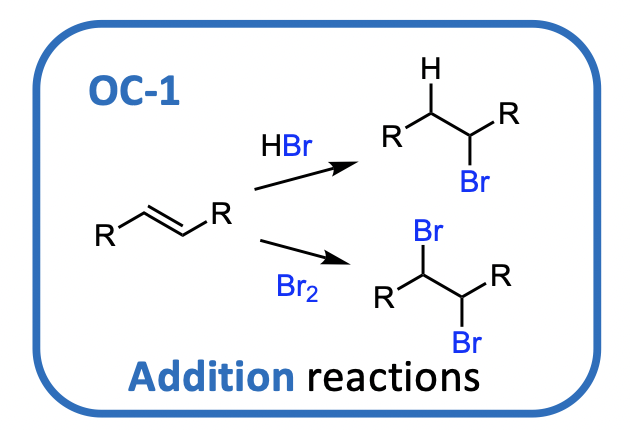
How are organic halogen compounds made? (not topic of OC-2)
• Addition of HCl or HBr to alkenes & acetylenes (ie. Hydrohalogenation)
• Addition of Cl2 or Br2 to alkenes & acetylenes (ie Halogenation)
• occurs via radical reaction pathways, leads to mixtures (not covered here)
Why are alkyl halides useful compounds? ... Alkyl halides:
Reactive compounds, due to the special nature of the C-X bond of the alkyl halide
Excellent electrophiles (R-X bond is polarizable) & generally form stable leaving groups as halide anions.
Can be reacted with nucleophiles OR with bases to form desired products
Therefore, alkyl halides undergo a wide variety of substitution and elimination reactions
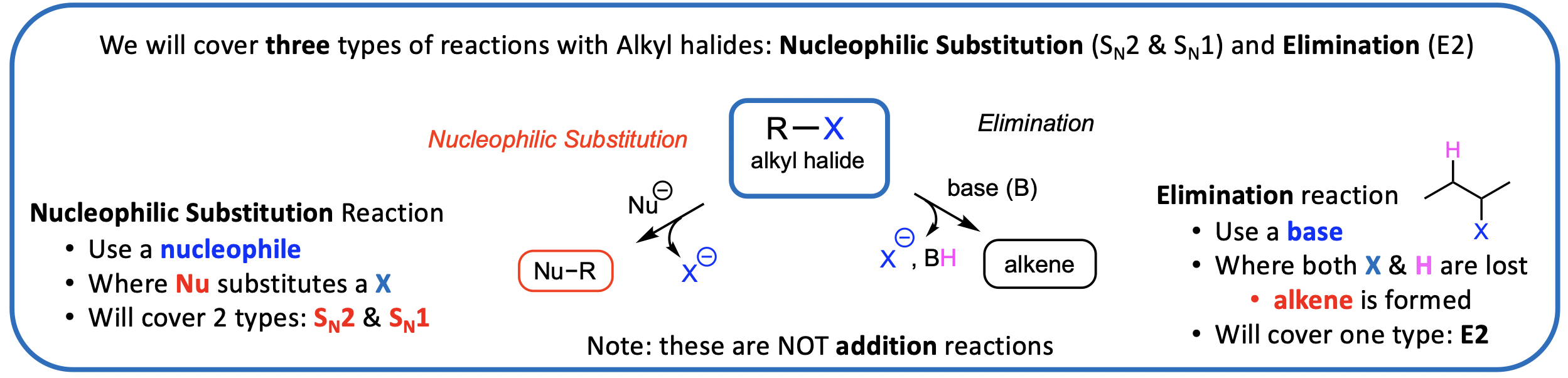
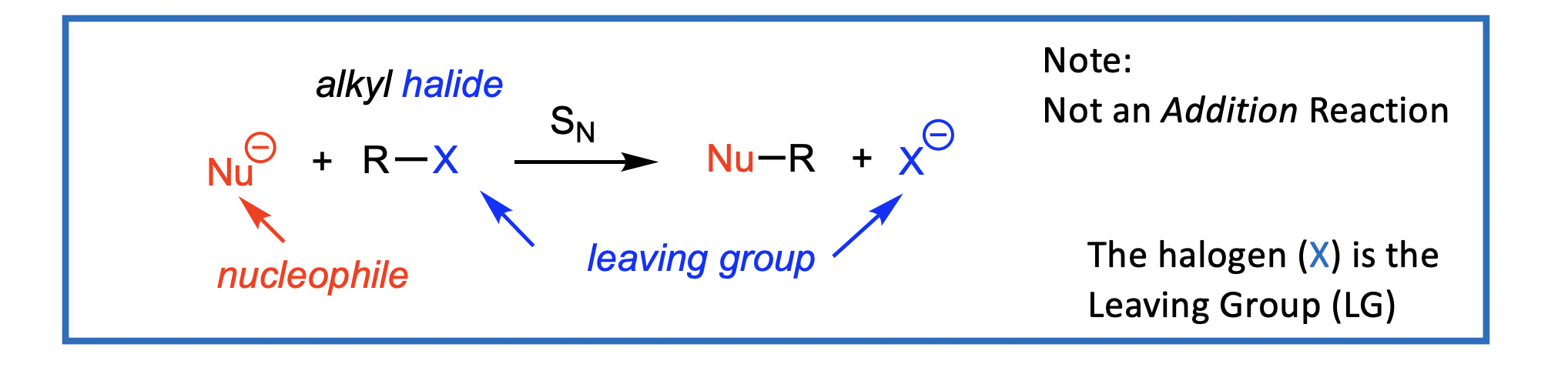
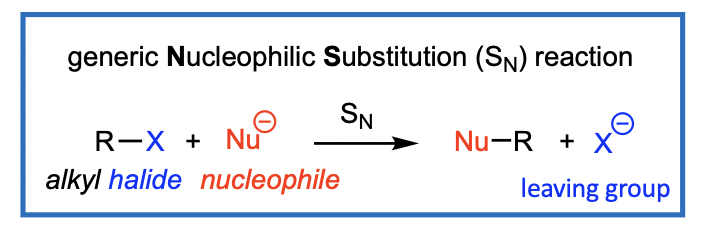
Nucleophilic Substitution (SN): Overview
A Nucleophilic Substitution (SN) of an alkyl halide (RX) leads to replacement of the halogen (X) by the nucleophile (Nu).
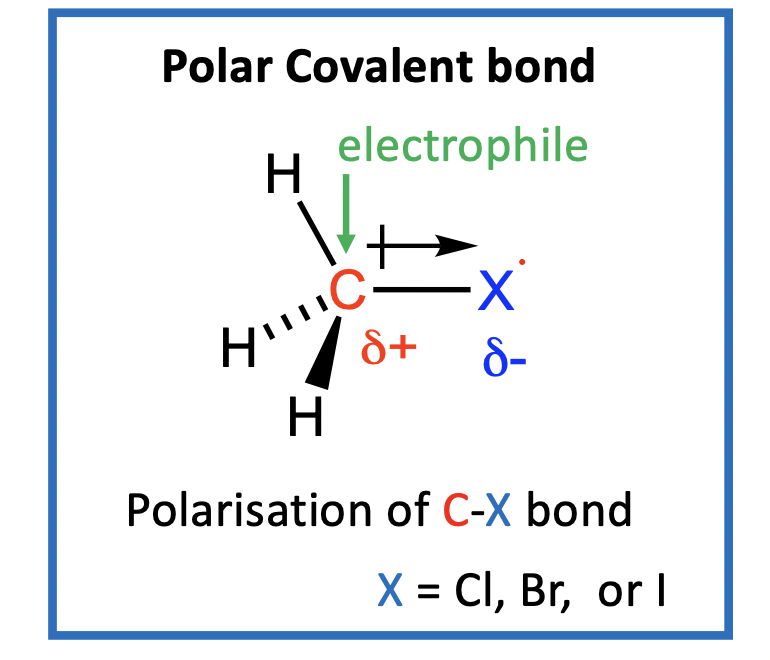
Why do alkyl halides (RX) undergo Nucleophilic Substitution (SN) reactions?
Electrophile: electron seeking because electron-poor (pos. or part-pos. eg. H+; sp3 C of C-X in alkyl halide; H3C+)
Nucleophile: nucleus seeking because electron-rich (neg. or part-neg charge) Nucleophiles react with Electrophiles (eg Nuc with sp3 C of R-X)
Note: R-F have very strong covalent R-X bonds, therefore are very unreactive, so we ignore fluoroalkanes here
Remember: SN reactivity depends on R-X bond strength, so as go from R-Cl to R-Br to R-I, bond strength gets weaker, so reactions are faster
Nucleophiles - Review
Nucleophile: nucleus seeking, electron rich = willing to give away electrons, negative or partially negative (Eg HO-, :NH3, H3C-, C=C)
1. NEGATIVE IONS are better nucleophiles than the corresponding NEUTRAL MOLECULES