Topic 5 Physical chemistry
Energy transfer in reactions
An exothermic reaction is one which gives out energy to the surroundings, usually in the form of heat and usually shown by a rise in temperature of the surroundings.
An Endothermic reaction is one white takes in energy from the surroundings, usually in the form of heat and usually shown by a fall in temperature of the surroundings.
The change in energy is called the enthalpy change. The overall change in energy in a reaction is called the enthalpy change. It has the symbol ΔH.
Reaction Profiles
Graphical Representation:
X-axis: Progress of the reaction.
Y-axis: Energy levels.
Exothermic Profile: Starts high (reactants), drops to a lower energy (products) with an activation energy peak.
Endothermic Profile: Starts low, rises to a higher energy level (products) with an activation energy rise.
Essential for predicting reaction behavior, optimizing conditions, and studying thermodynamics.
Practical Applications
Chemical Manufacturing: Optimizing reactions.
Environmental Science: Understanding ecosystem energy transfer.
Biotechnology: Harnessing biological energy processes.
Calculating enthalpy changes
The heat energy transferred (Q) can be calculated using the formula:
Q=m×c×ΔTQ = m \times c \times \Delta TQ=m×c×ΔT
Q = heat energy (J)
mmm = mass of the liquid (kg)
ccc = specific heat capacity (J/kg°C)
ΔT\Delta TΔT = change in temperature (°C)
molar Enthalpy Change:
The molar enthalpy change can be calculated from the heat transferred when a substance undergoes a phase change.
Formula:
Use the following steps:
Calculate the heat change (Q) using the heat energy formula.
Divide the heat transferred by the number of moles of the substance.
Bond energies
Energy Requirement:
Energy must be supplied to break bonds (endothermic process).
Energy is released when new bonds are formed (exothermic process).
Bond Breaking and Forming:
Endothermic Reactions: Require energy to break bonds.
Exothermic Reactions: Release energy when forming bonds; the energy released is greater than the energy needed to break the bonds.
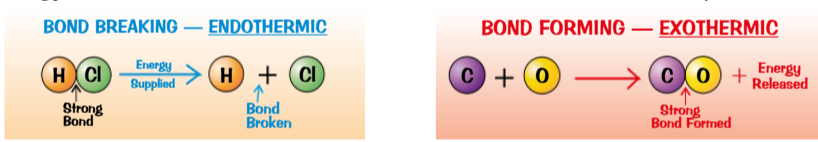
Understand the bond energy associated with breaking and forming bonds (e.g., between C-C and C-H).

Rates of reaction
Variation in Rates:
Reactions can occur at different rates depending on various factors, such as the nature of the reactants and conditions.
Examples:
Slow: Rusting of iron.
Fast: Combustion of fuels.
The rate of a reaction can be observed by recording the change in the amount of product formed or reactant used over time.
Graphs of reaction rates
Analyzing Graphs:
The steepness of the curve on a graph indicates the rate of reaction:
A steeper line shows a faster reaction.
A flatter line indicates a slower reaction.
Graph Comparison:
Different graphs can represent the same initial conditions but show variations in reaction rates.
Example Graphs:
Graph 1: Represents the original reaction.
Graphs 2 and 3: Represent faster reactions, showing how changes can lead to different rates.
The rate of a chemical reaction, according to collision theory, depends on how often reacting particles collide and how much energy they have. More frequent and energetic collisions make reactions happen faster. You can increase the number of collisions by raising the concentration of reactants or the temperature, which helps particles reach the necessary activation energy for successful reactions.
Factors Affecting the rate of reaction
Temperature:
Increasing temperature raises the energy of particles, leading to more frequent and energetic collisions, which speeds up the reaction.
Concentration (or Pressure):
Higher concentration of reactants or increased pressure in gases results in more collisions, thus increasing the reaction rate.
Surface Area:
A greater surface area allows more particles to collide, enhancing the reaction rate. Smaller solid particles have a larger surface area compared to larger ones.
catalysts:
The presence of a catalyst lowers the activation energy needed for a reaction, allowing it to proceed faster without being consumed in the process.
Catalysts
A catalyst increases the rate of a reaction without being consumed, keeping the reaction equation unchanged. It selectively facilitates reactions by lowering the activation energy, providing an easier pathway. This allows more particles to have the minimum energy needed for successful collisions and reactions.
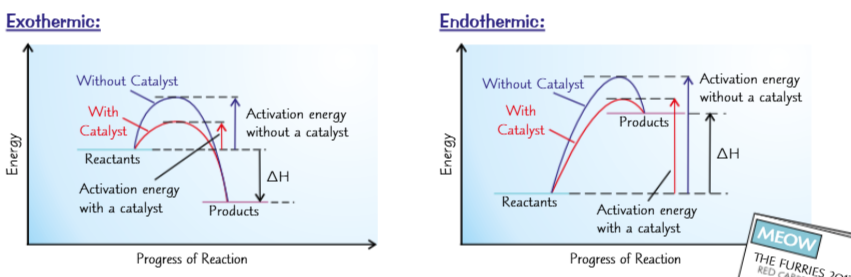
You can sow the effects of catalysts using reaction profiles
Activation energy is the difference between the energy of reactants and the highest point on a reaction profile.
Reaction profiles compare activation energy with and without a catalyst.
The peak for a reaction with a catalyst is lower than without.
Profiles show how a catalyst affects activation energy in exothermic and endothermic reactions.
Measuring rate of reactions
The speed of a reaction can be observed either by how quickly the reactants are used up or how quickly the products are formed. Its m=normaly a lot easier to measure products forming.

Precipitation:
Mix two solutions in a flask.
Observe the mixture for a change (like cloudiness) that indicates a reaction.
The faster the change, the quicker the reaction.
Change in Mass (Gas Given Off):
Measure mass in a flask as the reaction occurs.
If gas is released, the mass will decrease.
The quicker the mass drops, the faster the reaction.
The Volume of Gas:
Use a gas syringe to measure the gas produced during a reaction.
Track the volume of gas at regular intervals.
More gas produced in a shorter time means a faster reaction.
Reversible reactions
Reversible Reactions: These reactions can proceed in both forward and backward directions.
Dynamic Equilibrium:
When a reversible reaction reaches a state where reactants and products are formed at the same rate, it's in dynamic equilibrium.
The position of equilibrium can shift based on changes in concentration, temperature, or pressure.
Temperature and Pressure Changes:
Adjusting temperature affects the endothermic and exothermic directions of the reaction.
Changing pressure impacts reactions involving gases, shifting equilibrium towards fewer gas molecules.