Clinicopathologic Spectrum of Pediatric Posttransplant Lymphoproliferative Diseases Following Solid Organ Transplant
Page 1: Introduction and Overview of PTLD
Posttransplant Lymphoproliferative Disorder (PTLD)
Significant complication in pediatric solid organ transplant (SOT) patients.
Primarily EBV-driven CD20+ B-cell proliferations.
Treatment includes reduction of immunosuppression and anti-CD20 therapy.
Low incidence of PTLD leads to scarce prospective studies.
Objectives of the Review
Analysis of clinical features, pathogenesis, and classification of pediatric PTLD.
Data Sources
Personal experiences and published works from PubMed.
Key Conclusions
PTLD encompasses a broad spectrum of disorders from nonmalignant lymphoproliferations to lymphomas.
Majority of pediatric PTLDs are EBV positive.
Need for further research into biology, risk factors for aggressive disease, and treatment strategies.
Epidemiology
History and Incidence
PTLD first reported in 1968.
5-year cumulative incidence in pediatric SOT (2005-2014): 2% to 15.8%.
Highest incidence in lung, intestine, and multiorgan transplants.
Risk Factors
Contributing Factors
EBV status, type of transplanted organ, and immunosuppressive regimen.
Kidney transplants have lower PTLD risk compared to heart and lung transplants.
Age and race also contribute, with higher incidence in young White males.
EBV serostatus of both donor and recipient is crucial.
Surveillance of EBV via PCR is routine; higher EBV DNA levels correlate with PTLD risk.
Page 2: Pathogenesis of PTLD
Primary Mechanisms
Combination of impaired immunity and EBV infection leads to PTLD.
Over 90% of pediatric PTLD cases are EBV positive.
Viral Profile
EBV is a gamma herpesvirus with tropism for B cells; can establish lifelong infection.
Distinct latency phases in infected B cells.
Latency Phases
Latency 0: EBV genome persists without protein expression.
Latency I: EBNA1 expressed.
Latency II: LMP1 & LMP2 expressed, linked to malignancies in immunocompetent hosts.
Latency III: All EBNA and LMP proteins expressed; often seen in PTLD.
Post-SOT Implications
Post-transplant immunosuppression impairs CTL response, increasing risk for B-cell lymphoproliferation.
EBV-negative PTLD usually occurs later, is frequently monomorphic, and exhibits different pathogenesis.
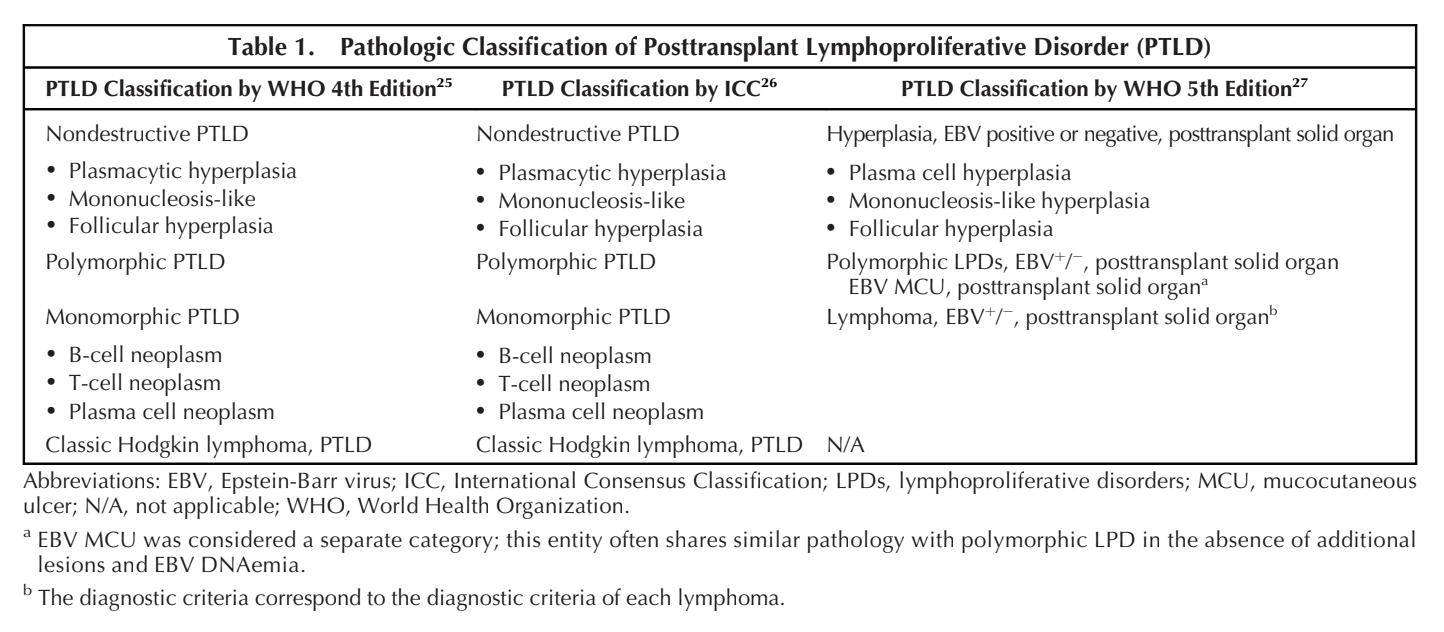
Page 3: Classification of PTLD
Current Classifications
WHO 2017 classification recognizes PTLD as an immunodeficiency-associated disorder.
Distinct categories: Nondestructive PTLD (ND-PTLD) and Polymorphic PTLD.
Nondestructive PTLD
Common in children; affects adenoids, tonsils, lymph nodes, and GI tract.
Histopathologic subtypes include plasmacytic hyperplasia, infectious mononucleosis-like, florid follicular hyperplasia.
Histopathologic Characteristics
EBV positivity in ND-PTLD without disruption of tissue architecture.
Recurrent mutations found such as NOTCH1, CREBBP, indicating clonal cytogenetic abnormalities.
Polymorphic PTLD Characteristics
Architectural destruction, extensive infiltrate with varied cell types, often with necrosis.
Difficulty in differentiating from monomorphic PTLD.