4.3 Electronegativity & Bond Polarity
Polar vs. Non-Polar Covalent Bonds
Electrons in a covalent bond (shared pair) may be shared either equally or unequally.
- Equal(ish)→ Non-Polar
- 0.5 difference or less
- Not equal → Polar
- 0.5 to 1.7
Some elements have a stronger ability to attract electrons in a shared pair
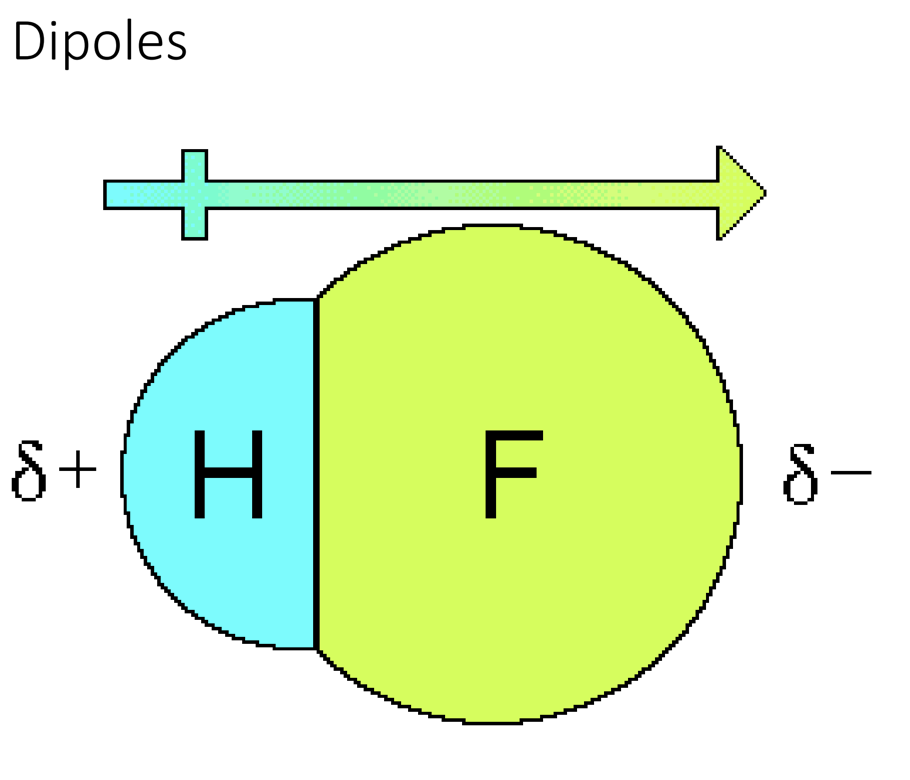
Example: hydrogen fluoride
- Contains a single covalent bond
- Fluorine has the stronger force of attraction so electrons spend more time around it.
- Contains more protons in nuclear making the radius smaller and pulling electrons in a lot
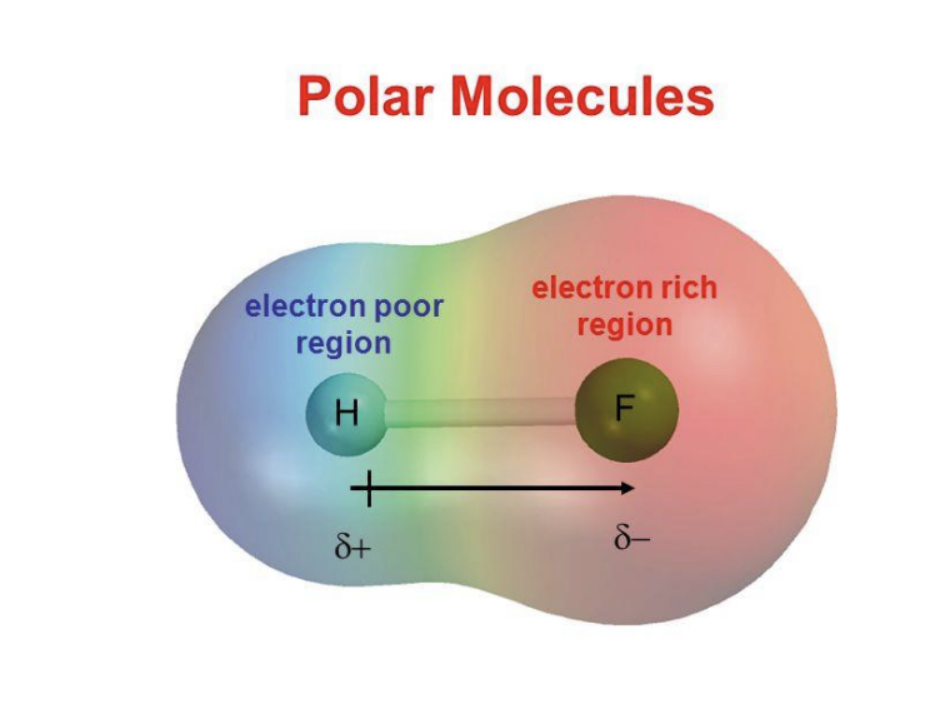
Electronegativity: an atom’s ability to attract shared electrons (in a bond) to itself.
Fluorine is the most electronegative element with 4.0.
We can use EN between elements to determine the nature of a bond.
Dipoles
Polar covalent bonds have a permanent separation of charge, we call a dipole.