Organic Chemistry Chapter 1: Structure and Bonding
Structure and Bonding
Organic Chemistry
Historical use
compounds in living organisms
Modern use
the study of carbon-containing compounds
1.1 Atomic Structure
Protons
+1 charge
mass = 1.6726 × 10^-27 kg
Neutrons
0 charge (neutral)
mass = 1.6750 × 10^-27 kg
Electrons
-1 charge
mass = 9.1096 × 10^-31 kg (smallest)
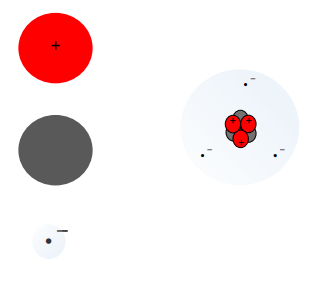
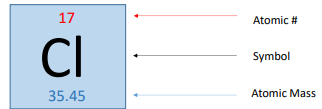
Atomic number (Z)
equal to number of protons an atom contains
Mass number (A)
total number of protons and neutrons

Isotopes
same atomic number (Z) but a different number of neutrons, so a different atomic mass
example: chlorine-35 and chlorine-37
Atomic Mass
weighted average mass of an element’s natural occurring isotopes
1.2 Orbitals
Wave equation
mathematical expression describing specific behavior of an electron in an atom
Wave function ψ = orbital
defined region of space around nucleus where electron may be
Types:
s
p
d
f
s orbital
p orbital
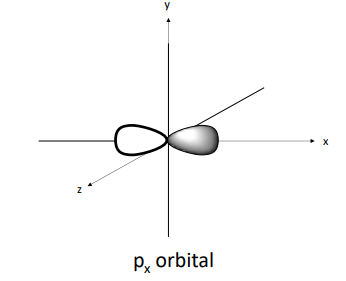
Electron shells
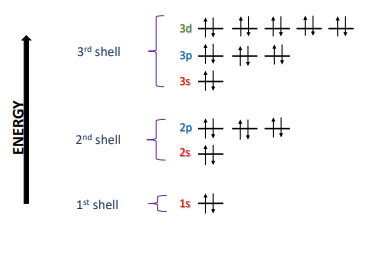
1.3 Electron Configuration
Rules:
1. Orbitals are filled from lowest energy to highest energy
2. Only two electrons can occupy an orbital, and they must be opposite spin
3. If more than one orbital of equal energy is available, one electron is placed in each orbital before electrons are paired.
Carbon → C → 6 (atomic number, so need 6 electrons) → so 6 protons and 6 electrons) → 6 e- → (1s²2s²2p²) (where ² means electrons)
(→ ←) 1s
(→ ←) 2s
(→ ) (→ ) ( ) 2p… the signs are the same so they cannot be paired together, and we need to fill up all empty space first
2s²2p² will be valence electrons
1s² is core
Carbon has 4 valence electrons
1.4 Chemical Bonding Theory
Kekule and Couper (1858)
Carbon is tetravalent
Carbon bonds to other carbons to form extended chains
Van’t Hoff and Le Bel (1874)
four bonds to carbon are not randomly oriented
atoms bonded to carbon arranged in a tetrahedron
Nature of Chemical Bonds
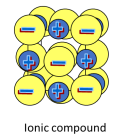
Ionic bonds
Force of electrostatic attraction between oppositely charged ions
More common in inorganic chemistry than in organic chemistry
Carbon is less likely to form a cation than metals
Carbon is less likely to form an anion than nonmetals
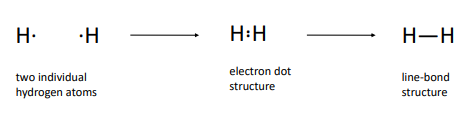
Covalent Bonding
An electron pair that is shared between two atoms
Molecules are neutral groupings of atoms held together by covalent bonds
1916 by Lewis
Lewis structures (electron-dot structures)
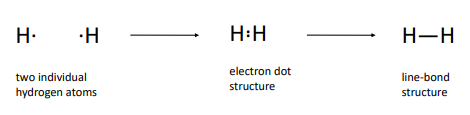
Octet rule
Atoms gain, lose, or share electrons to give a stable electron configuration characterized by 8 valence electrons.
Exception: Hydrogen only has 2 electrons (via sharing its 1s electron with another atom)
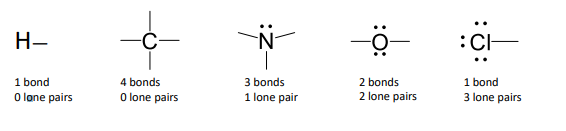
H - Cl
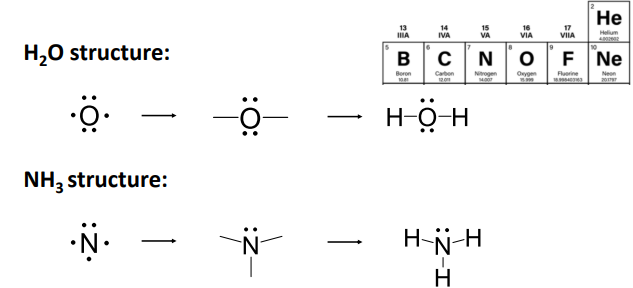
Example: Draw the line-bond structures of H2O and NH3 .
Practice: Draw the electron-dot and line-bond structures for methanol, CH3OH.
Example: Draw the line-bond structures of the following organic molecules.
C_3H_8
CH3CH2CH2CH2CH3