Periodicity - Melting Point Trend
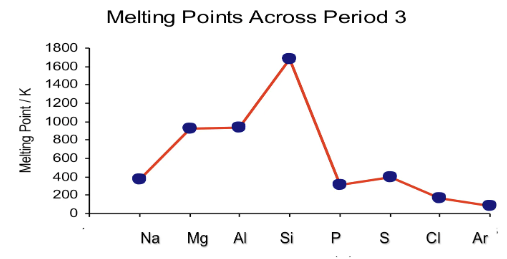
Na → Al
Metallic Bonding.
Melting point increases as number of electrons increases. But charge increases as well (increasingly positive). This causes stronger electrostatic force of attraction (stronger metallic bonds) which require more energy to overcome
Si
Si is a giant covalent structure.
It requires a lot of energy to overcome its strong covalent bonds
P → Ar
all simple molecular structures
P4, S8, Cl2
Sulphur has the most electrons available, thus has stronger intermolecular forces, strongest VDW