2.2
key questions
How does the structure of water contribute to its unique properties?
How does water’s polarity influence its properties as a solvent?
Why is it important for cells to buffer solutions against rapid changes in PH?
the water molecule
- ^^polarity: A molecule in which the charges are unevenly distributed is said to be polar^^
* ^^magnet with 2 poles^^ - ^^Hydrogen bond: the attraction between 2 hydrogen atoms with polarity^^
water: polar molecule » forming multiple hydrogen bonds » causing unique properties of water
- ^^Cohesion:^^ The attraction between molecules of the ^^same^^ substance
* Ex: drops of water forming beads on a smooth surface - ==Adhesion:== the attraction between molecules of ==different== substances
- Heat capacity: the amount of energy needed to raise the substance's temperature, caused by the movement in the molecules
Solutions and Suspensions
- ^^Solutions: ions gradually becoming disprised in the water^^
- evenly distributed components
- %%Solute: the substance that is dissolved%%
- %%Solvent: the substance in which the solute dissolves%%
- %%Saturated: a given amount of water has dissolved all of the solutes it can%%
^^Suspensions: water and non-dissolved materials^^
* \
* ex: blood; mostly water and undissolved particles
Acids, Bases, and pH
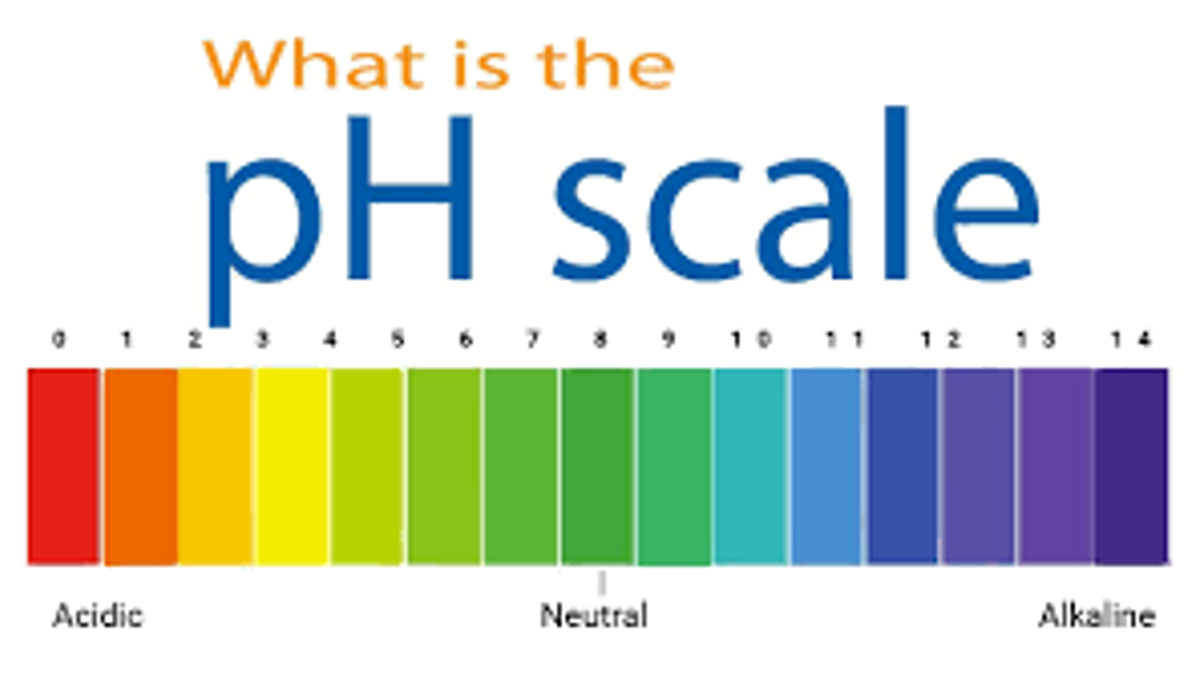
electrically neutral: hydrogen ions (H+)= negative hydroxide ions (OH-) \n
pH scale: indicates the concentration of Hydrogen positive ions in solution
- ==below 7 = acidic==
* ==more hydrogen-positive ions than OH-negative ions==
* ==lower the number, the more acidic it is== - @@7 = neutral@@
* @@Hydrogen-positive and OH-negative are equal@@
*
*- Each step is a factor of 10
- %%Acid: any compound that releases H+ ions (below 7)%%
- %%Base: a compound producing OH- ions (above 7)%%
- %%Buffers: weak acids or bases that can react with strong acids or bases to prevent sharp, sudden changes in pH%%