Reproduction, Human Fertility, and In Vitro Fertilization (IVF)
Introduction
This section explores human reproduction, focusing on trends in fertility, the physiological aspects of male and female fertility, the challenges of infertility, and the science and application of In Vitro Fertilization (IVF). Key topics include:
Global and regional trends in total fertility rates.
The impact of age on female fertility and oocyte quality.
Trends in male fertility, including sperm counts.
The biology of female germ cells (oocytes) and follicular development.
Sperm function, including capacitation and the acrosome reaction.
The process of fertilization.
Common causes of infertility in males and females.
The history, process, and outcomes of IVF, including related technologies.
Human Fertility Trends
Total Fertility Rate (TFR):
The TFR represents the average number of children a woman would have if she experienced the age-specific birth rates of a particular year throughout her childbearing years.
Global Trends: There has been a significant decline in the world's TFR, from around 5 births per woman in the 1960s to below 2.5 births per woman by 2023.
Regional Variations:
Europe (UN data) shows a TFR that has fallen below the replacement level (2.1 births) since before 1980 and was around 1.5 births per woman in 2023.
South Korea exhibits one of the lowest TFRs, falling below 1 birth per woman around 2018 and continuing to decline.
Replacement Level: A TFR of approximately 2.1 births per woman is considered the replacement level needed to maintain population stability, excluding migration.
Female Fertility
Decline with Age:
Female fertility naturally declines with increasing age. The likelihood of pregnancy decreases significantly after the age of 30, with a sharp decline after 35-40 years. For example, the likelihood of pregnancy might be around 80% for women in their early 20s, decreasing to under 40% by age 40, and very low by mid-40s.
Aneuploidy in Oocytes: A major factor contributing to the age-related decline in female fertility is the increasing rate of aneuploidy (abnormal number of chromosomes) in oocytes (eggs).
The percentage of aneuploidy in human eggs is lowest between maternal ages 20-32 (around 20-25%).
It increases significantly for maternal ages under 20 (around 40%) and rises sharply for ages 33 and above, exceeding 40% and reaching as high as 80% in older women.
IVF Data and Oocyte Quality: Data from IVF treatments confirms that the decline in female fertility is largely attributable to oocyte/egg quality.
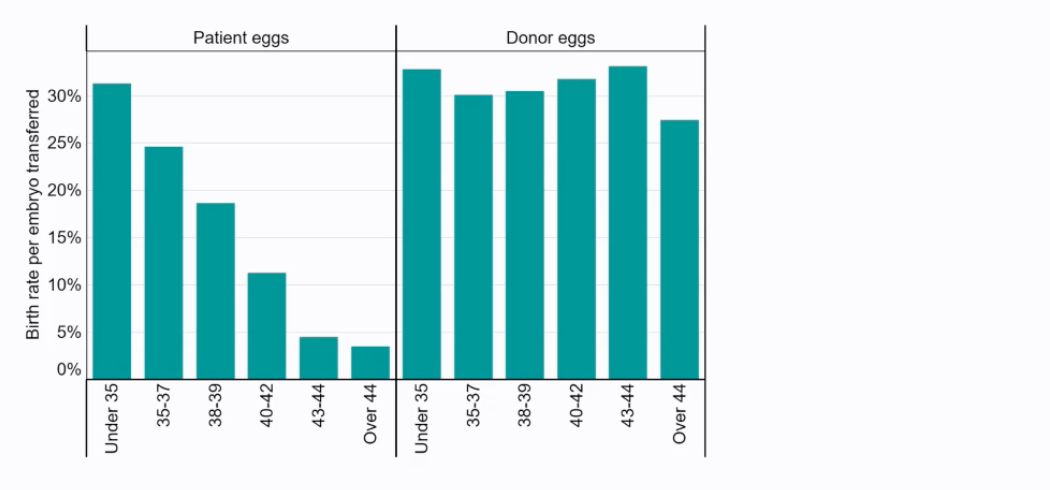
Using patient eggs, the birth rate per embryo transferred decreases with maternal age: highest for under 35 (around 30%), declining to around 10% for ages 40-42, and very low (under 5%) for ages 43 and over.
In contrast, when donor eggs (typically from younger women) are used, the birth rate per embryo transferred remains relatively high and consistent across different recipient age groups (around 27-33%), even for recipients over 44. This indicates that the uterine environment is often still capable of supporting pregnancy if a healthy embryo is available.
Female Germ Cells (Oocytes) and Follicular Development:
The number of female germ cells (oogonia/oocytes) peaks during fetal development (around 7 million at ~6 months post-conception) and then declines throughout life.
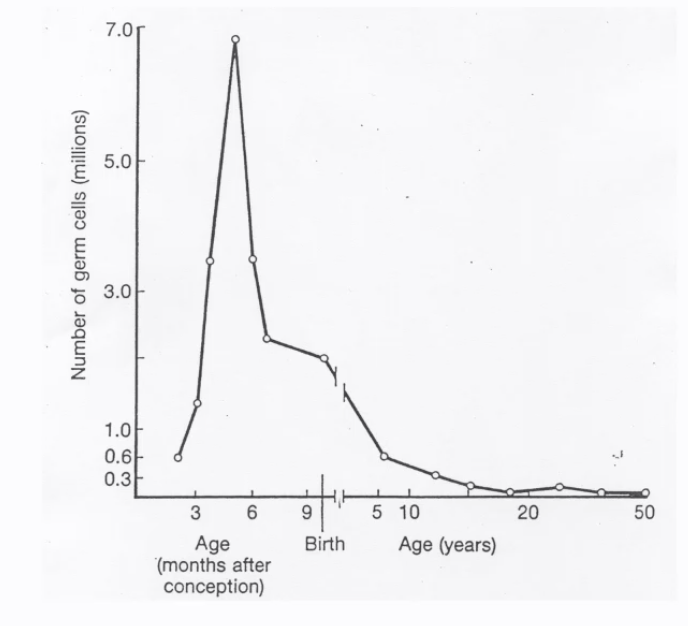
At birth, there are approximately 1-2 million oocytes.
This number falls to around 0.3-0.6 million by puberty and continues to decrease thereafter.
Folliculogenesis: Oocytes develop within ovarian follicles.
Primordial Follicle: Contains a primary oocyte arrested in prophase I of meiosis, surrounded by a single layer of follicular (granulosa) cells.
Maturing Follicle (e.g., Primary/Secondary Follicle): The oocyte grows, the zona pellucida (a glycoprotein layer) forms around it, and follicular cells proliferate to form multiple layers. Theca cells develop around the follicle.
Vesicular or Graafian Follicle (e.g., Tertiary/Antral Follicle): A fluid-filled space called the antrum develops. The oocyte is surrounded by cumulus cells. This is the stage just before ovulation.
Mature MII Oocyte (Egg): Upon completion of meiosis I (usually triggered by the LH surge before ovulation), the oocyte becomes a secondary oocyte arrested in metaphase II (MII) of meiosis. This is the mature egg ready for fertilization.
Male Fertility
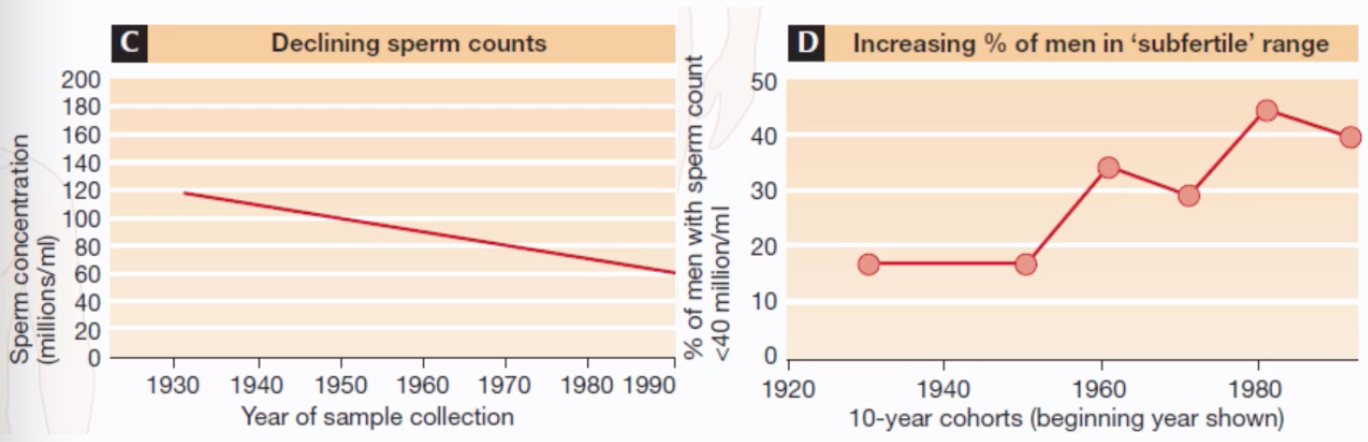
Declining Sperm Counts:
Studies have indicated a historical decline in sperm concentration in men. For example, average sperm concentration may have fallen from over 100 million/ml in the 1930s-1940s to around 60 million/ml by the 1990s.
Concurrently, there has been an increasing percentage of men falling into the 'subfertile' range, defined for instance as having a sperm count below 40 million/ml. This percentage rose from under 20% in cohorts from the 1920s-1940s to over 40% in cohorts from the 1980s.
Sperm Function:
Sperm Structure: A mature spermatozoon consists of a head (containing the nucleus with condensed DNA and the acrosome), a midpiece (packed with mitochondria for ATP and glucose for glucose to pyruvate, like skeletal muscle), and a tail or principal piece (for motility). The annulus is a ring-like structure between the midpiece and principal piece.
Journey in the Female Tract:
Over 100 million sperm may be deposited in the female tract during intercourse.
This number rapidly diminishes as sperm travel; around 1,000 may reach the fallopian tubes, and only about 10 sperm may be found around the oocyte.
Fertilization typically takes place hours to as long as 6 days after copulation.
Sperm selection during this journey can involve motility characteristics (assessed by techniques like microfluidics, density gradient centrifugation, or swim-up in IVF labs) and ability to bind to structures like hyaluronan (a component of the cumulus cells surrounding the egg).
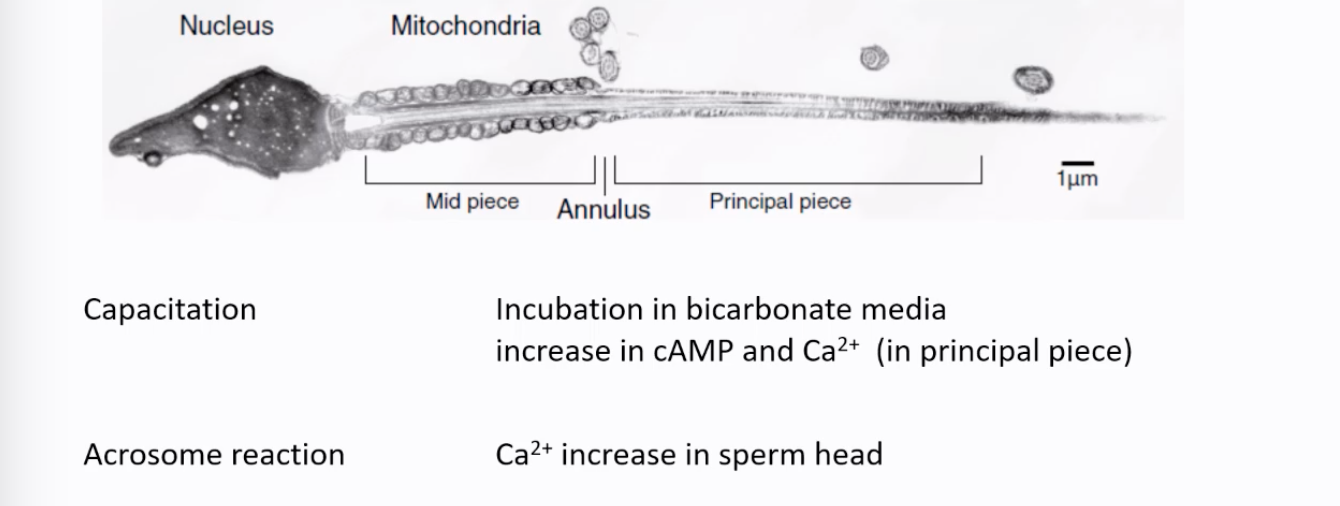
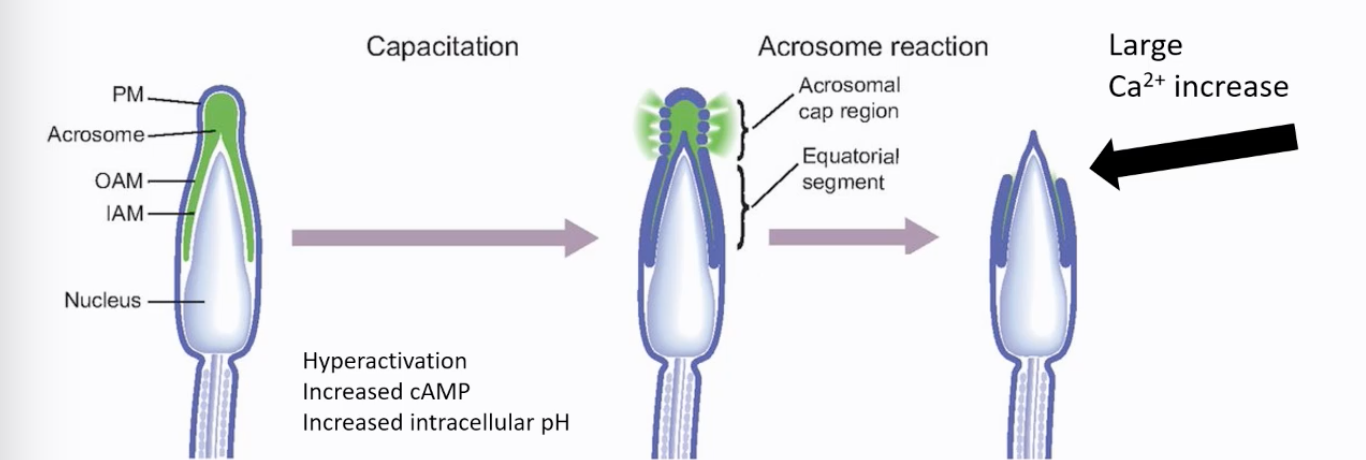
Capacitation (for IVF): A series of physiological changes sperm must undergo in the female reproductive tract (or in specific culture media in vitro) to become competent to fertilize an egg.
Involves incubation in bicarbonate-containing media, leading to an increase in intracellular cAMP and Ca²⁺ (particularly in the principal piece for hyperactivated motility) and an increase in intracellular pH.
Results in hyperactivation, a vigorous, whip-like motility pattern that helps sperm detach from the oviductal epithelium and penetrate the egg's vestments.
Acrosome Reaction: An exocytotic event where the contents of the acrosome (a cap-like structure on the sperm head containing enzymes) are released.
Triggered by Ca2+.
This reaction is essential for sperm to penetrate the zona pellucida.
The contents remain associated with the head of the sperm.
It is thought to occur before the sperm reaches the zona pellucida, possibly triggered by substances like progesterone in the vicinity of the egg. The equatorial segment of the sperm head is typically involved in the subsequent fusion with the oocyte plasma membrane (oolemma).
CatSper Channels and Calcium Influx:
Ca²⁺ influx into sperm is critical for motility and the acrosome reaction. CatSper (Cation Channels of Sperm) are pH-sensitive Ca²⁺ channels located in the principal piece of the sperm tail.
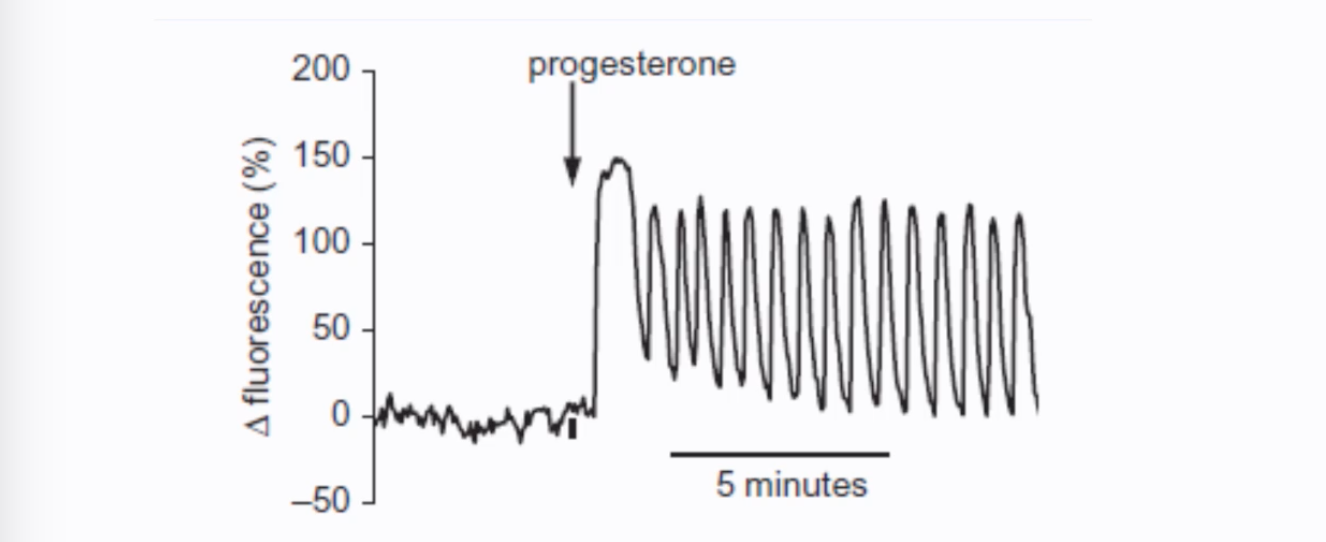
They are activated by increased intracellular pH and in some species progesterone, leading to Ca²⁺ entry during the Capacitation process.
This is important as the sperm does not store Ca2+ (no SR)
Progesterone can stimulate Ca²⁺ oscillations in human spermatozoa, as shown by patch clamp experiments of individual sperm cells and fluorescence measurements.
Fertilization
Fertilization is the fusion of sperm and egg to form a zygote.
Sperm-Egg Interaction: Sperm that have undergone capacitation and the acrosome reaction can penetrate the cumulus cells and bind to the zona pellucida. Enzymes released during the acrosome reaction help digest a path through the zona pellucida.
Sperm-Egg Fusion: After penetrating the zona pellucida, the sperm reaches the perivitelline space (PVS) and fuses with the oocyte plasma membrane (oolemma). This fusion is typically mediated by the equatorial segment of the acrosome-reacted sperm.
Oocyte Activation: Sperm fusion triggers a series of events in the oocyte known as activation. A key event is a series of intracellular Ca²⁺ oscillations (spikes) in the oocyte cytoplasm.
In mammals, the sperm delivers a sperm-specific Phospholipase C zeta (PLCζ) into the oocyte upon fusion. PLCζ is a ~70 kDa protein found in all mammals that hydrolyzes phosphatidylinositol 4,5-bisphosphate (PIP₂) to generate inositol trisphosphate (InsP₃) and diacylglycerol (DAG). InsP₃ then binds to receptors on the endoplasmic reticulum, causing the release of Ca²⁺ and initiating the oscillations. PLCζ is localized in the sperm head, including the equatorial segment, consistent with its delivery at fusion.
These Ca²⁺ oscillations are crucial for completing meiosis II (extrusion of the second polar body), cortical granule exocytosis (which leads to the zona hardening, preventing polyspermy), and initiation of embryonic development.
The oscillations occure once every 10-30mins.
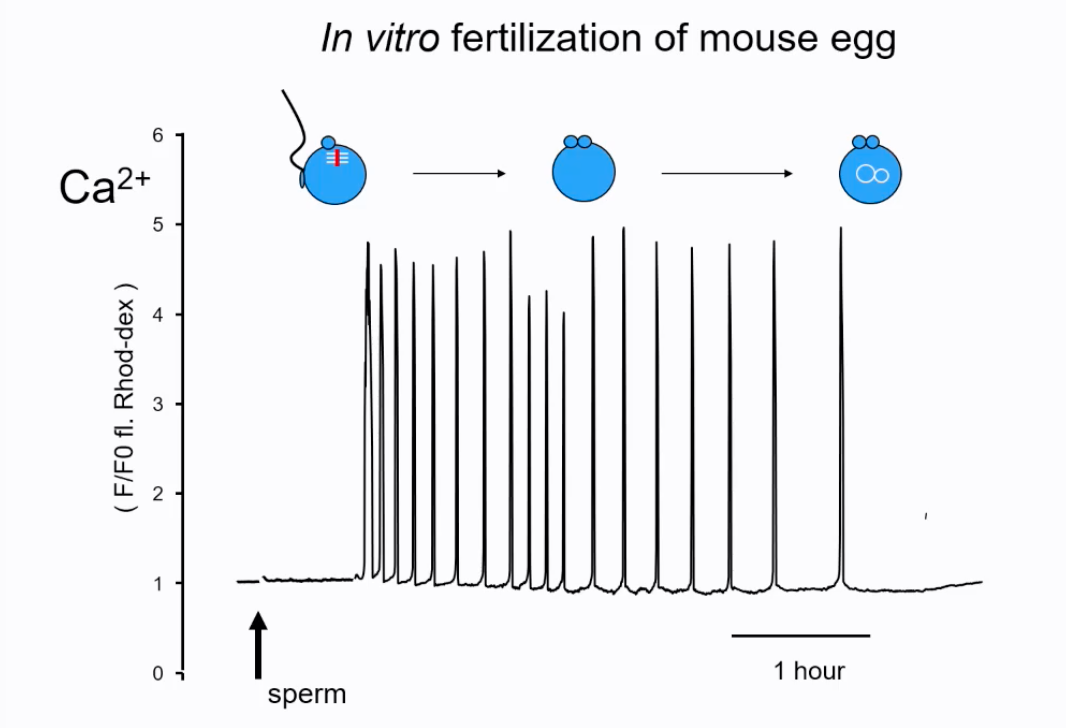
Pronuclear Formation: After oocyte activation and completion of meiosis II, the maternal chromosomes decondense to form the female pronucleus. The sperm nucleus decondenses to form the male pronucleus. The fertilized oocyte with two pronuclei (2PN) is now called a zygote.
Infertility
Definition: Generally defined as the failure to conceive after 1 or 2 years of regular unprotected intercourse.
Prevalence: Affects over 10% of couples.
Causes: Can be due to female factors, male factors, or a combination.
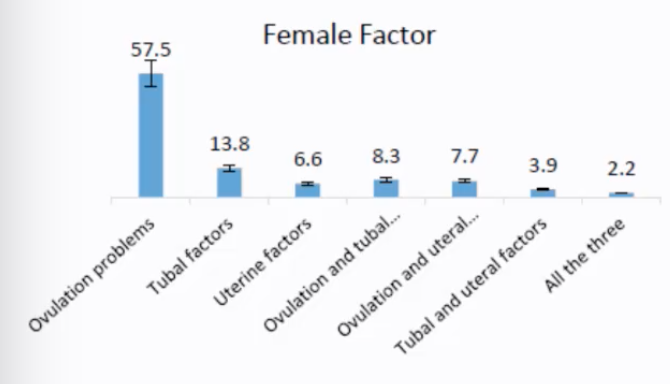
Female Factors:
Ovulation problems (most common, ~57.5%)
Tubal factors (e.g., blocked fallopian tubes, ~13.8%)
Uterine factors (e.g., fibroids, adhesions, ~6.6%)
Combinations like ovulation and tubal factors (~8.3%), ovulation and uterine factors (~7.7%), tubal and uterine factors (~3.9%), or all three (~2.2%).
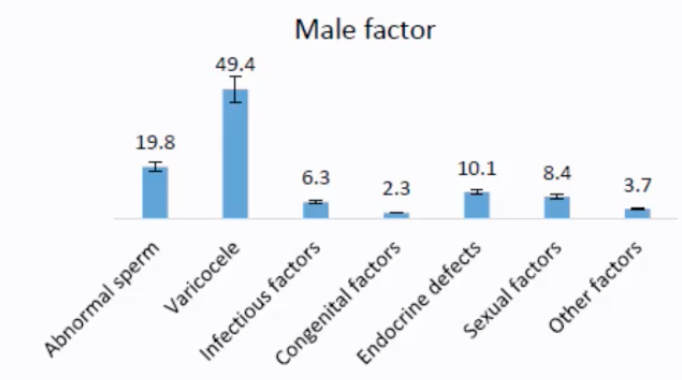
Male Factors:
Abnormal sperm (e.g., low count, poor motility, abnormal morphology, ~19.8%)
Varicocele (enlarged veins in the scrotum, potentially affecting sperm production, ~49.4% - note: this high percentage for varicocele as a specific listed cause seems unusually prominent compared to "abnormal sperm" in general; typically, abnormal sperm parameters are the most common finding, with varicocele being one potential underlying cause)
Infectious factors (~6.3%)
Congenital factors (~2.3%)
Endocrine defects (~10.1%)
Sexual factors (e.g., erectile dysfunction, ejaculatory problems, ~8.4%)
Other factors (~3.7%).
In Vitro Fertilization (IVF)
IVF is an assisted reproductive technology (ART) where fertilization occurs outside the body.
History: The first successful IVF birth was Louise Brown in 1978, a result of the work by Robert Edwards, Jean Purdy, and Patrick Steptoe. Robert Edwards received the Nobel Prize for this work.
Prevalence: Approximately 4 million IVF treatments are performed globally per year, resulting in about 1 million live births annually. IVF accounts for about 2% of births in the UK and up to 5% in some other countries.
IVF Process:
Superovulation (Controlled Ovarian Stimulation):
The woman is given hormonal medications (containing LH and FSH analogs) to stimulate the ovaries to produce multiple mature oocytes (typically aiming for 2-10 oocytes per cycle) instead of the single oocyte typical of a natural cycle.
Oocyte Retrieval:
Mature oocytes, still surrounded by cumulus cells, are collected from the ovaries, usually via transvaginal ultrasound-guided aspiration.
Sperm Preparation:
A semen sample is processed to select the healthiest, most motile sperm. Techniques include:
Swim-up: Allowing motile sperm to swim out of the seminal plasma into culture media.
Density Gradient Centrifugation: Separating sperm based on their density.
A high number of sperm (e.g., >10 million initially) are processed.
Fertilization:
Intrauterine insemination (IUI) is a widely used fertility treatment for couples with unexplained subfertility, cervical factor subfertility, and mild male subfertility. IUI involves depositing processed sperm in the uterine cavity, bypassing natural barriers. While less invasive and expensive than IVF, IUI's effectiveness is debated. Evidence suggests that IUI with ovarian hyperstimulation (OH) increases live birth rates compared to IUI alone. However, OH raises concerns about multiple pregnancies. The optimal time for IUI is 12-36 hours after hCG injection. Success rates are highest in the first three treatment cycles. Despite its popularity, large, high-quality randomized controlled trials comparing IUI with or without OH to timed intercourse are lacking.
Standard IVF: The retrieved oocytes and selected sperm are incubated together in a culture dish for fertilization to occur (typically takes up to 16 hours for signs of fertilization like pronuclei formation).
Intracytoplasmic Sperm Injection (ICSI): If male factor infertility is severe, or if previous IVF attempts failed, ICSI may be used. A single sperm is selected and injected directly into the cytoplasm of each mature oocyte. ICSI bypasses many natural barriers to fertilization including the cumulus cells, zona pellucida, and oolemma fusion. ICSI is used in approximately 70% of all IVF treatments.
Sperm cell is damaged in order to release the important PLCζ, this is what then triggers Ca2+ oscillations that initiates activation.
Sperm retrieval combined with intracytoplasmic sperm injection (ICSI) has revolutionized the treatment of severe male infertility, particularly azoospermia (absense of motile sperm). Various surgical techniques have been developed to retrieve sperm from the epididymis or testis, including PESA, MESA, TESA, TESE, and micro-TESE. These methods are used for both obstructive and non-obstructive azoospermia, with success rates varying depending on the underlying cause. Testicular sperm retrieval has shown promising results even in challenging cases like Klinefelter syndrome. While some studies suggest that certain techniques may be more effective than others, there is insufficient evidence to recommend any specific method.
Embryo Culture:
Fertilized oocytes (zygotes) are cultured in the laboratory for several days, allowing them to develop into embryos.
Developmental Stages:
Day 0: Mature oocyte.
Day 1: Fertilized oocyte (2 pronuclei - 2PN).
Day 2: 2-4 cell embryo.
Day 3: 8 cell embryo.
Day 4: Morula (a compact ball of cells).
Day 5: Blastocyst (an embryo with an inner cell mass, which will form the fetus, and an outer layer called the trophectoderm, which will form the placenta, and a fluid-filled cavity called the blastocoel).
Embryo Transfer:
One or two embryos are selected and transferred into the woman's uterus, usually at the blastocyst stage (Day 5) or sometimes earlier (Day 3).
One is transfered in an older woman (twins are a risk factor) and two to three in younger women.
Embryo selection is crucial for maximizing pregnancy rates while minimizing the risk of multiple pregnancies.
Embryos are selecting if they divide the most or they divide the most evenly.
IVF/ICSI can lead to higher rates of twins (~5-25%) and triplets (~3% if more than two embryos are transferred, though this practice is less common now) compared to natural conception.
IVF: Adjunct Technologies:
Time-Lapse Imaging (e.g., EmbryoScope): Embryos are cultured in special incubators equipped with cameras that take frequent images, allowing for continuous monitoring of development without removing them from the controlled environment. This can help in selecting the embryos with the best developmental potential for transfer.
Embryo Cryopreservation (Vitrification): High-quality embryos not used in the fresh transfer cycle can be frozen (cryopreserved) for future use. Vitrification is an ultra-rapid cooling technique that minimizes ice crystal formation, improving embryo survival rates post-thaw.
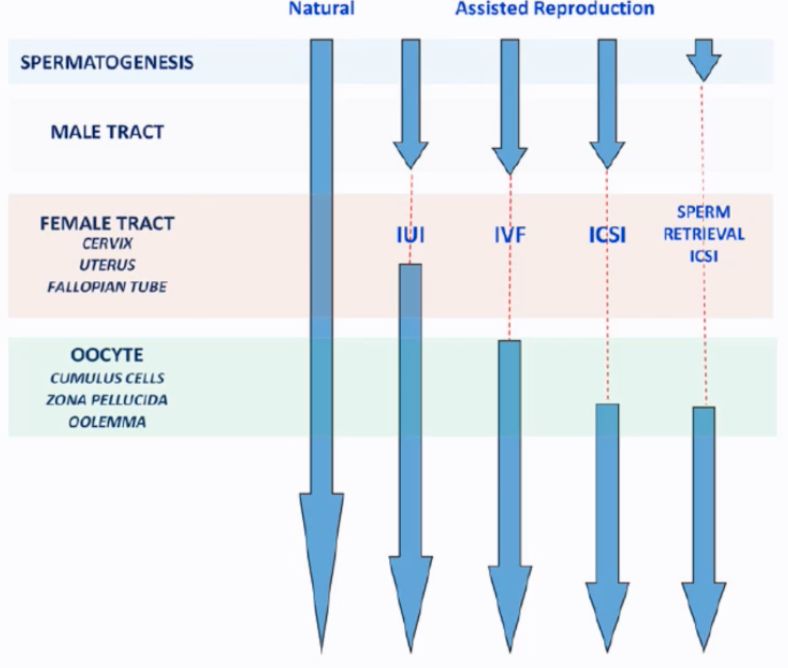
Barriers Bypassed by Assisted Reproduction:
Intrauterine Insemination (IUI): Bypasses the cervix.
Standard IVF: Bypasses the cervix, uterus (for sperm travel), and fallopian tube environment. Sperm still need to penetrate cumulus and zona pellucida and fuse with the oolemma.
ICSI: Bypasses all oocyte vestments (cumulus, zona pellucida) and the oolemma fusion step.
Sperm Retrieval + ICSI: Used when there is no sperm in the ejaculate (azoospermia), sperm are retrieved directly from the epididymis or testes and used for ICSI. This bypasses spermatogenesis completion (in terms of release) and the entire male tract transport.
Experiments to change Ca2+
You can reduce Ca2+ signals by remove extracellular Ca2+ (in medium). This reduces Ca2+ influx and will stop Ca2+ oscillations and stops motility in sperm. It can reduce the frequency of Ca2+ oscillations in eggs. Some of these effects can also be achieved by applying Ca2+ channel blockers (e.g. heavy metal ions such as Gadolinium).
One can increase intracellular Ca2+ by applying Ca2+ ionophore such as A23187. This causes Ca2+ influx in sperm and mostly Ca2+ release from stores in eggs. In sperm the Ca2+ increase is persistent, and this inhibits motility but triggers the acrosome reaction. In eggs this causes a large single Ca2+ increase that activates development of the egg.