Acids and Alkalis Notes
9 Common acids and alkalis
9.1 Common acids and alkalis
Acids:
Taste sour.
Found in various kinds of vinegar (ethanoic acid), lemons, kiwi fruit, oranges, soft drinks, and yoghurt.
Also present in household products like hair conditioner, lotion, and toilet cleaner.
Alkalis:
Give a slippery feel (like soap).
Some have a bitter taste.
Found in foods like Chinese noodles and lye water rice dumplings.
Also present in household products like kitchen cleaner, alkaline batteries, baking soda, bleach, soap, and toothpaste.
Many acids and alkalis (especially in household cleaners) are irritant or corrosive.
Handle with care.
Never taste them except in food.
Acids commonly used in the laboratory:
Hydrochloric acid HCl
Sulphuric acid H2SO4
Nitric acid HNO3
Alkalis commonly used in the laboratory:
Sodium hydroxide NaOH
Ammonia solution NH3
Calcium hydroxide Ca(OH)2 (lime water).
Most acids and alkalis used in the laboratory are irritant or even corrosive.
Safety precautions must be taken when handling them.
Safety precautions when handling acids and alkalis in the laboratory:
Read hazard warning symbols on chemical bottles carefully.
Wear a laboratory coat and disposable or protective gloves.
Wear safety spectacles.
Do NOT mix acids or alkalis with other chemicals without teacher approval.
Pour waste solutions into suitable waste bottles, not the sink.
Wash hands thoroughly after handling acids or alkalis.
Emergency treatment for accidents involving acids and alkalis:
If acids or alkalis splash onto:
Skin: Immediately wash the affected area under slow-running water for at least 10 minutes.
Eyes: Immediately wash the affected eye(s) using an eye wash bottle for at least 10 minutes.
Clothes: Carefully remove the affected clothes and avoid contact with any acids or alkalis on the clothes.
If the accident is serious, take the injured person to hospital as soon as possible.
Uses of acids:
For food preservation:
Acids inhibit the growth of microorganisms that cause food spoilage.
Vinegar (ethanoic acid) is commonly used to preserve vegetables.
For stain removal:
Hydrochloric acid is present in many toilet cleaners.
It reacts with dirt and stains, which are then easily removed when the toilet is flushed.
Uses of alkalis:
For making noodles:
Lye water (alkaline solution) is added to Chinese noodles to make them chewy.
For grease removal:
Sodium hydroxide is present in many kitchen cleaners.
It reacts with grease on kitchen stoves, allowing it to be easily wiped or washed away.
9.2 Distinguishing acids and alkalis
Natural Indicators:
Some substances in nature change color when they come into contact with acids or alkalis.
These substances can be used as acid-alkali indicators.
Examples:
Red cabbage extract
Butterfly pea flower extract
Red onion skin extract
Robert Boyle discovered the use of plant extracts as natural indicators in the 17th century
Litmus:
Commonly used acid-alkali indicator in the laboratory.
Extracted from several kinds of lichens.
Two kinds: blue litmus and red litmus.
Available in solution and paper form (litmus paper).
Behavior in different substances:
Acidic: Blue litmus turns red, red litmus remains red.
Alkaline: Blue litmus remains blue, red litmus turns blue.
Neutral: Both blue and red litmus show no color change.
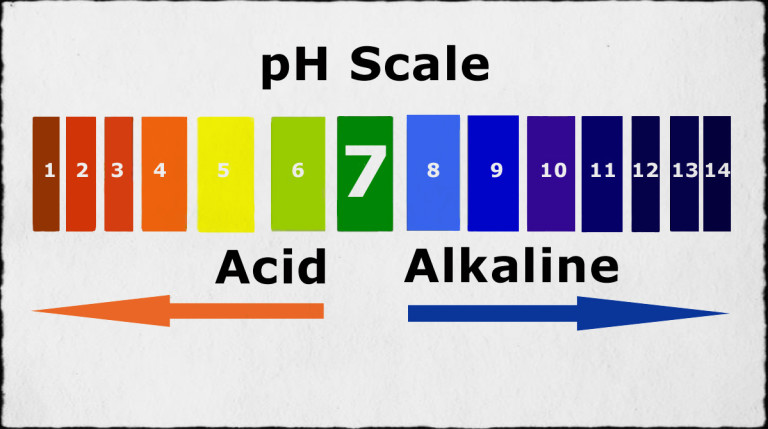
pH Scale:
Used to describe the relative acidity or alkalinity of substances.
Ranges from 0 to 14.
Acidic: pH < 7
The lower the pH, the more acidic the substance.
Neutral: pH = 7
Alkaline: pH > 7
The higher the pH, the more alkaline the substance.
Universal Indicator:
A solution made up of several indicators that shows a range of colors depending on the acidity or alkalinity of the substance being tested.
Also available in paper form (pH paper).
pH Meter:
Electronic instrument used to measure the pH values of solutions.
Must be calibrated before use.
Provides a more accurate pH value than universal indicator.
9.3 Neutralization
Definition: The reaction between an acid and an alkali.
Change in pH value:
Acids have pH values lower than 7.
Alkalis have pH values higher than 7.
When an acid and an alkali are mixed, the pH value of the resulting solution changes, approaching 7 (neutral).
Products of Neutralization:
Salt and water are formed.
Thermal energy (heat) is released during neutralization.
Word Equation:
acid + alkali → salt + water
The type of salt formed depends on the types of acid and alkali that react.
Skill Builder 9.1 Writing word equations for neutralization
The general format of the word equation for neutralization is as follows:
acid + alkali \rightarrow salt + waterConservation of Mass:
The total mass of the reactants (acid and alkali) is the same as that of the products (salt and water).
Applications of Neutralization:
Treating stomach ache with antacid:
Antacids (alkaline) neutralize excess stomach acid (hydrochloric acid).
Brushing teeth with toothpaste:
Toothpaste (slightly alkaline) neutralizes acids produced by bacteria in the mouth, preventing tooth decay.
Treating chemical waste from factories:
Waste is neutralized with suitable alkalis or acids before discharge to minimize pollution.
Treating insect stings and bites:
Bee stings, mosquito bites, and ant bites are acidic and can be neutralized with soap solution (alkaline).
Wasp stings are alkaline and can be neutralized with vinegar (acid).
Regulating soil acidity and alkalinity:
Lime (alkaline) is added to acidic soil to raise the pH.
Sulphur is added to alkaline soil, which bacteria convert into sulphuric acid to lower the pH.
9.4 Corrosive nature of acids
Effects of acids on metals:
Dilute acids corrode some metals (e.g., zinc, magnesium, iron).
Hydrogen gas is produced when dilute acids react with metals.
Word equation: metal + acid → salt + hydrogen
Effects of acids on building materials:
Dilute acids corrode building materials containing calcium carbonate (e.g., marble and limestone).
Carbon dioxide is produced when dilute acids react with calcium carbonate.
Word equation: carbonate + acid → salt + water + carbon dioxide
Acidic cleaners should be avoided for marble
9.5 Acid rain
Definition: Rainwater with a pH value lower than 5.6.
Causes:
Burning of fossil fuels (e.g. coal, oil) releases acidic gases into the air (sulphur dioxide, nitrogen oxides).
These acidic gases react with oxygen and water in the air to form sulphuric acid and nitric acid.
The increased acidity leads to acid rain.
Animation: Acid rain formation process
Effects:
Damages plant leaves directly, hindering photosynthesis.
Releases toxic minerals into the soil, harming plant growth.
Harms or kills living things in water.
Corrodes buildings and statues made of calcium carbonate.
Corrodes metal objects (e.g., car bodies, metal bridges).
Controlling Acid Rain(這part不太重要):
US:
Providing financial benefits to the industry for reducing emissions of sulphur dioxide and nitrogen oxides
Rewarding petrol refiners if they produce petrol cleaner than required
China:
Closing inefficient factories which release large amounts of air pollutants
Enhancing public awareness on reducing pollution from vehicles
Restricting the mining of coal with high sulphur content
Germany:
Largely adopting renewable energy sources to replace coal as the main source of energy
Over 40% of electricity is powered by renewable energy sources
Sweden:
Restricting the burning of crude oil with high sulphur content
Liming in rivers and lakes to improve water quality and decrease the death rate of fish
9.6 Potential hazards related to the use of acids and alkalis
Concentrated vs. Dilute Acids and Alkalis:
Concentrated acids and alkalis are more corrosive than dilute ones.
Mixing Cleansing Products:
Mixing different cleansing products can be dangerous.
Reactions may occur, releasing toxic substances or thermal energy (potentially leading to explosions).
NEVER mix any cleansing products.
General Safety Rules When Using Cleansing Products:
Keep the room well-ventilated.
Wear gloves, a mask, and protective clothing.
Wash hands thoroughly after using the products.
Prepare 1:99 diluted bleach containing 5.25% of active ingredient.
Emergency treatment for accidents:
*If the cleansing product has splashed onto the body, wash the affected area with a large
amount of slow-running water.
*If a harmful gas is inhaled, open the windows and move the injured person to an area with lots of fresh air.
*If the injury is serious, take the injured person to hospital as soon as possible.