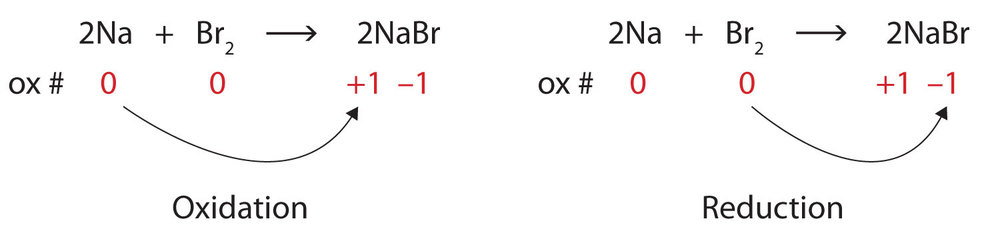Chapter 10 Oxidation and Reduction
Oxidation: the loss of one or more electrons by an atom
Reduction: the gain of one or more electrons by an atom
Oxidation-Reduction (or redox) reactions: Chemical reactions that involve the transfer of electrons
Oxidation Number: A number assigned to an atom that helps keep track of the number of electrons on the atom.
Rules for assigning oxidation numbers
Atoms in their elemental state are assigned an oxidation number of 0.
Atoms in monatomic (i.e., one-atom) ions are assigned an oxidation number equal to their charge. Oxidation numbers are usually written with the sign first, then the magnitude, to differentiate them from charges.
In compounds, fluorine is assigned a −1 oxidation number; oxygen is usually assigned a −2 oxidation number [except in peroxide compounds (where it is −1) and in binary compounds with fluorine (where it is positive)]; and hydrogen is usually assigned a +1 oxidation number [except when it exists as the hydride ion (H−), in which case rule 2 prevails].
In compounds, all other atoms are assigned an oxidation number so that the sum of the oxidation numbers on all the atoms in the species equals the charge on the species (which is zero if the species is neutral).