Empirical and Molecular Formula
Empirical and Molecular Formula
The molecular formula is the formula that shows the number and type of each atom in a molecule.
E.g. the molecular formula of ethanoic acid is C2H4O2
The empirical formula is the simplest whole number ratio of the elements present in one molecule or formula unit of the compound.
E.g. the empirical formula of ethanoic acid is CH2O
Organic molecules often have different empirical and molecular formulae.
Simple inorganic molecules however have often similar empirical and molecular formulae.
Ionic compounds always have similar empirical and molecular formulae.
Empirical Formula Calculations
Empirical formula is the simplest whole number ratio of the elements present in one molecule or formula unit of the compound.
It is calculated from knowledge of the ratio of masses of each element in the compound.
The empirical formula can be found by determining the mass of each element present in a sample of the compound.
It can also be deduced from data that gives the percentage compositions by mass of the elements in a compound.
Worked Example One:
Calculating empirical formula from mass
Determine the empirical formula of a compound that contains 2.72 g of carbon and 7.28 g of oxygen.
Answer:
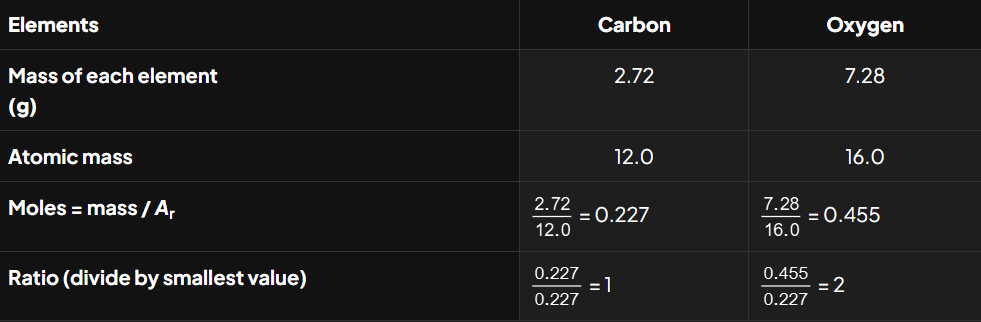
So, the empirical formula of the compound is CO2
Worked Example Two:
Calculating empirical formula from percentage
Determine the empirical formula of a hydrocarbon that contains 90.0% carbon and 10.0% hydrogen.
Answer:
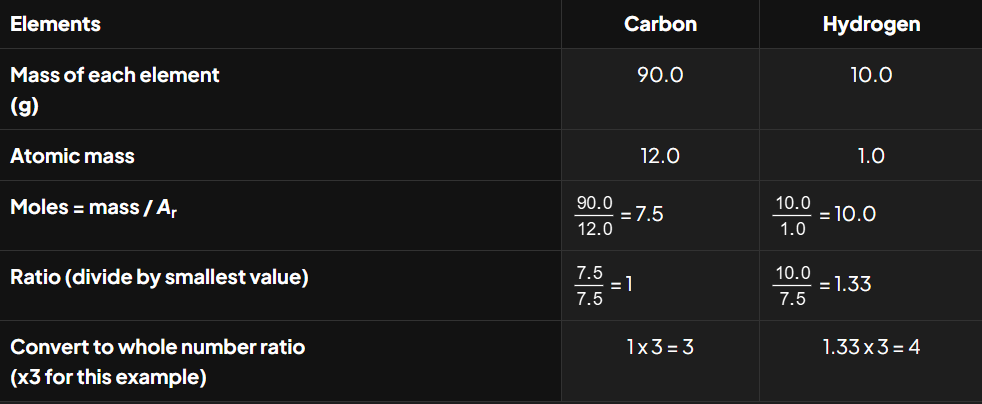
So, the empirical formula of the compound is C3H4
Molecular Formula Calculations
The molecular formula gives the exact numbers of atoms of each element present in the formula of the compound.
The molecular formula can be found by dividing the relative formula mass of the molecular formula by the relative formula mass of the empirical formula.
Multiply the number of each element present in the empirical formula by this number to find the molecular formula.
Worked Example One:
Calculating molecular formula
The empirical formula of X is C4H10S and the relative molecular mass of X is 180
What is the molecular formula of X?
(Ar data: C = 12, H = 1, S = 32)
Answer:
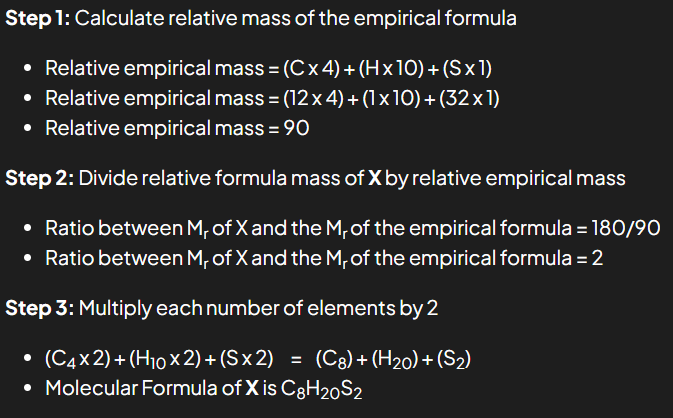
So the Molecular Formula of X is C8H20S2
Worked example Two:
Calculating empirical formula and molecular formula
Analysis of a compound X shows that it contains 24.2 % by mass of carbon, 4.1 % by mass of hydrogen and 71.7% by mass of chlorine.
Calculate the empirical formula of X.
Use this empirical formula and the relative molecular mass of X (Mr = 99.0) to calculate the molecular formula of X.
Answer:
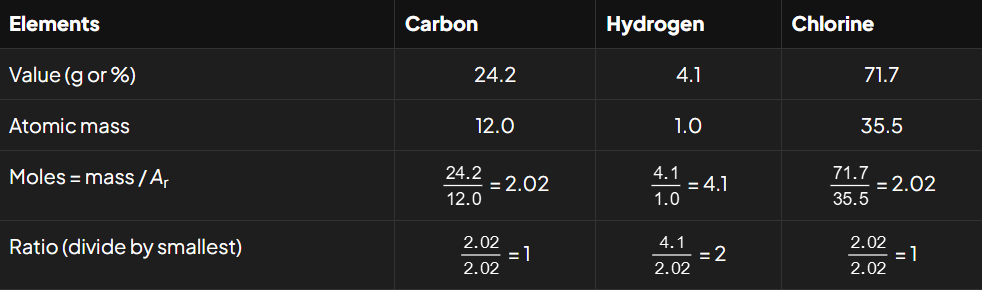
So, the empirical formula of compound X is CH2Cl
The relative formula mass of the empirical formula is:
Relative formula mass = (1 x C) + (2 x H) + (1 x Cl)
Relative formula mass = (1 x 12.0) + (2 x 1.0) + (1 x 35.5)
Relative formula mass = 49.5
Divide the relative formula mass of X by the relative formula mass of the empirical formula
Ratio between Mr of X and the Mr of the empirical formula = 99.0/45.9
Ratio between Mr of X and the Mr of the empirical formula = 2
Multiply each number of elements by 2
(C1 x 2) + (H2 x 2) + (Cl1 x 2) = (C2) + (H4) + (Cl2)
The molecular formula of X is C2H4Cl2