Determination of electrode potentials
Connecting half-cells together
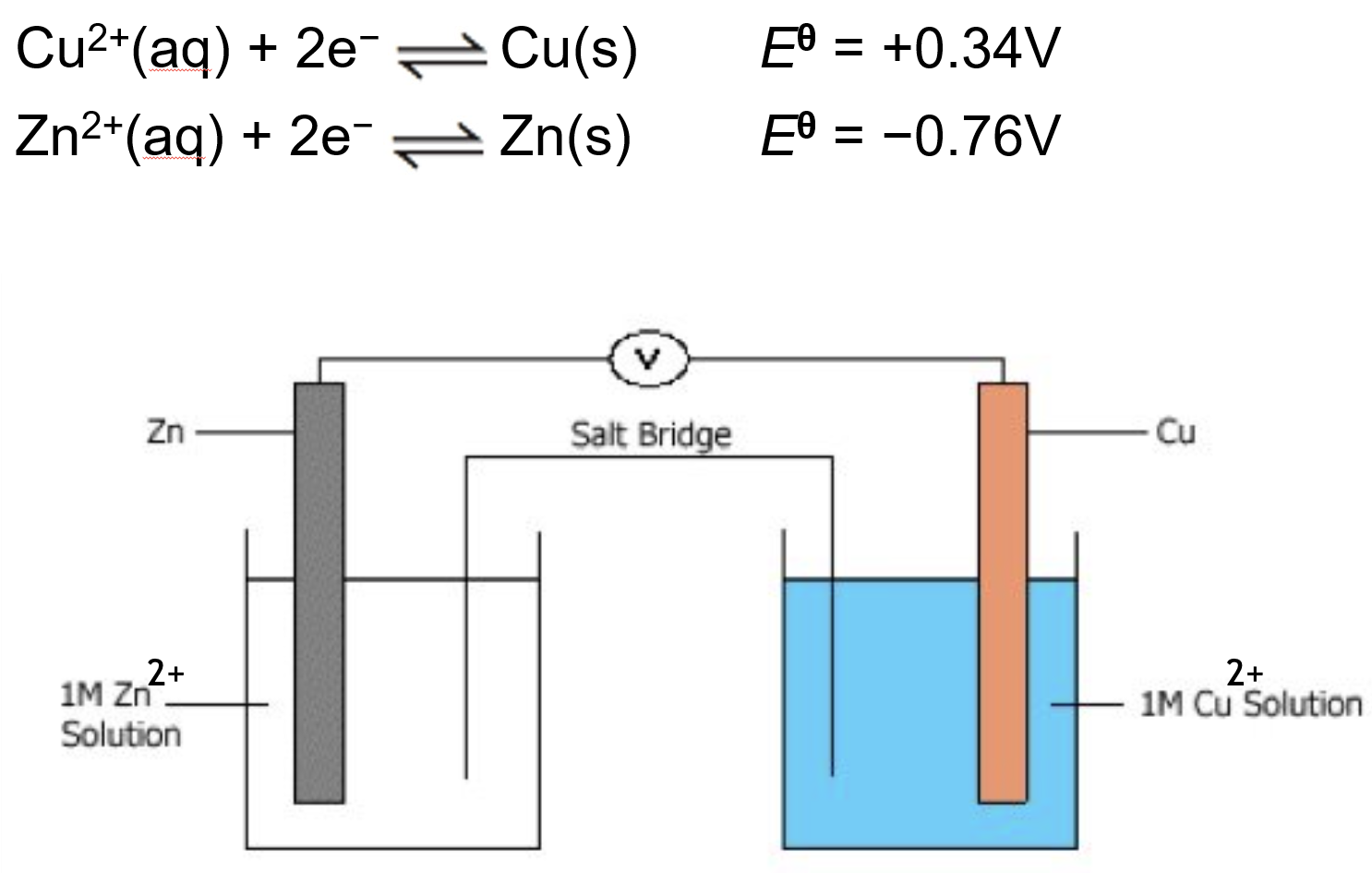
The two half-cells are setup, under standard conditions
The two electrodes are connected together usually with a voltmeter
The two solutions are connected with a salt bridge
A salt bridge allows ions to flow
Salt bridges are usually made of a strip of filter paper soaked in concentrated solution of a salt that won’t react with either half-cell solution
For example KNO3(aq) is commonly used
Different types of half-cells
Metal and solution of the metal ions
The metal is used as the electrode
And the metal ion solution at 1.00 moldm-3
Non-metal and solution of non-metal ions
A gaseous non-metal and its aqueous solution
This is set up per the standard hydrogen electrode
Ions of the same element but in different oxidation states
All ions are present in the same solution at 1.00 moldm-3
A platinum electrode is used, because it is inert
Calculating cell potentials
Compare the two half equations, the more positive Eo half-cell will go forwards (reduced) the least positive will go backwards (oxidised)
Cu2+(aq) + 2e- ⇌ Cu(s) E0 = +0.34V (forwards, reduced)
Zn2+(aq) + 2e- ⇌ Zn(s) E0 = -0.76V (backwards, oxidised)
Eocell = Eoreduced - Eooxidised
+0.34 - -0.76 = +1.10V
Representing cells
Al(s) | Al3+(aq) || Cu2+(aq) | Cu(s) E0cell = +2.00V
A solid vertical line indicates a change in phase e.g. the change from a solid to a solution
A double vertical line shows a salt bridge
The species with the highest oxidation state is show next to the salt bridge
The right hand half-cell dictates the sign of the E0cell, as the copper half cell has a more positive E0 then the value in this case is positive
Remember electrons will flow from the least positive E0 half-cell (oxidised) to the most positive (reduced) half-cell.
Anodes and cathodes
The polarity of the anode and cathode is different between electrolysis (when electricity drives the chemistry) to electrochemical cells (when chemistry produces electricity)
Electrochemical cells | Electrolysis | ||
Anode | Oxidation | Negative | Positive |
Cathode | Reduction | Positive | Negative |