C1
Atoms - Smallest particle of a chemical element that can exist
Everything in this world are made out of atoms
Element - A substance that is made of only one sort of atom
For example → Na (Sodium) and Li (Lithium)
Compound - Contain more than one type of atom and they are formed from elements by chemical reactions which involves the formation of one or more new substance by an energy change. Contain two or more elements chemically combined in fixed proportions and can be represented by formulae using the symbols of atoms from which they formed
Naming Compounds:
Metal and Non Metal (Ionic):
Metal’s name doesn’t change
Non Metal’s name ends with “ide”
EG → Na
2S = sodium sulfide/ K2O = potassium oxide
Metal and 2+ Covalent Non Metals:
The metal name does not change
The non-metal's name ends in “ate” if oxygen is present
EG → Na2CO3 = sodium carbonate/ KNO3 = potassium nitrate
State symbols:
s → Solid
g → Gas
I → Liquid
Aq → Aqueous (Dissolved in water)

Mixtures - Two or more elements or compounds not chemically combined together but physically combined. The chemical properties in the mixture remain unchanged
Examples are, shampoos, alloys, air, seawater
Solvent - liquid in which a solute dissolves in.
Solute - the substance that dissolves in a liquid to form a solution
Solution - the mixture formed when a solute has dissolved in a solvent
Soluble - substance that will dissolve
Insoluble - substance that will not dissolve
Ways of separating mixtures physically:
Filtration
Crystallisation
Simple distillation
Fractional distillation
Chromatography
Filtration:
Separates substances that are insoluble in a solvent from those that are soluble
Begin with an insoluble solid and a liquid, for example sand and salt where salt dissolves in water but sand doesn’t.
Pour the mixture into the filter funnel. When you pour the mixture into a filter funnel lined with filter paper, the sand will be caught by the filter paper and the water with salt dissolved will drip through.
The salt solution is now separated from the sand
Crystallisation:
Separates a soluble substance from a solvent by evaporation
Begin with a soluble solid dissolved in a solvent. We begin with a salt solution, containing salt (solute) dissolved in water (solvent).
Warm the mixture in an evaporating basin. The mixture is poured into an evaporating basin and gently heated. This will evaporate away some of the solvent, which makes the solution more concentrated , until it becomes a saturated solution.
Crystals start to develop. The point at which crystals form is called the point of crystallisation.
Remove the heat. When insoluble crystals form, the heat is removed, and the saturated solution is allowed to cool. The crystals are separated from the remaining solvent using filtration and left to dry, within the folds of filter paper.
Simple Distillation:
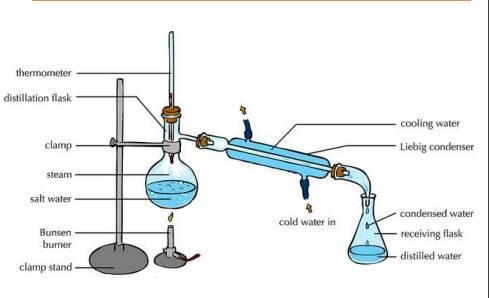
Separates a liquid from a mixture by evaporation follow by condensation
Set up the apparatus. The distillation apparatus involves a round-bottomed flask, a heat source, a thermometer, a condenser and a collecting beaker.
Use a round-bottomed flask because it is less prone to cracking under heat.
The solution is heated and the liquid begins to evaporate. The vapour from the solution rises and then passes down the condenser, in which it is cooled and condensed using cold water.
liquid collects in the beaker. The cooling in the condenser turns the vapour into liquid again, and this liquid is collected in the beaker.
Fractional Distillation - Separates a mixture into a number of different parts, called fractions. Substances with high boiling points condense at the bottom and substances with low boiling points condense at the top
Chromatography:
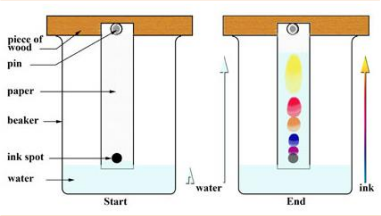
Separates small amounts of dissolved substances by running a solvent along absorbent paper
Draw a baseline on the chromatography paper using a pencil. The baseline is insoluble, so it won’t move with the solvent.
Place a small spot of the sample mixture on the baseline.
Pour the solvent into a beaker. Place a glass rod over the beaker, and tape or clip the paper to it, ensuring that the paper’s base just touches the solvent without being submerged.
Allow the solvent to slowly travel upwards through the paper, taking with it a few soluble pigments from the sample mixture.
Remove the paper from the beaker before the solvent reaches the top.
Analyse the chromatogram to identify the different components of the sample mixture.
Finally, mark the distance travelled by the solvent.
History of the atom:
Early 1800s → John Dalton said that atoms were tiny spheres that couldn’t be divided
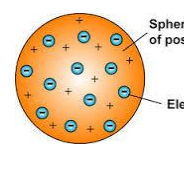
1890s → J.J Thompson discovered electrons and scientists believed that atoms were spheres of positive charge with negative charge spread throughout the plum pudding model
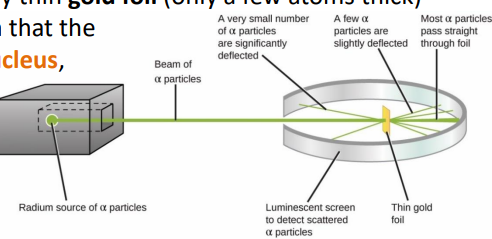
1908-1913 → Ernest Rutherford designed an experiment carried out by Geiger and Marsden. They fired alpha particles at a piece of very thin gold foil (only a few atoms thick) which scattered, leading to the conclusion that the mass of an atom was concentrated in a nucleus, which was charged. It proposed that electrons orbited around the nucleus.
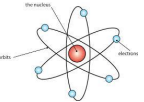
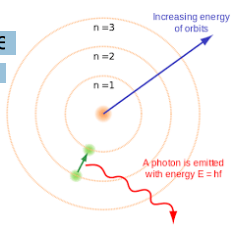
1914 → Niels Bohr noticed that the light given out when atoms were heated only had specific amounts of energy and he adapted the nuclear model by suggesting that electrons orbit the nucleus at specific distances in certain fixed energy levels (or shells).
1914-1932 → Positive charge of nucleus could be subdivided into particles of positive charge called protons
1932 → James Chadwick bombarded beryllium atoms with alpha particles. An unknown radiation was produced. Chadwick interpreted this radiation as being composed of particles with a neutral electrical charge and the approximate mass of a proton. This particle became known as the neutron.
Sub Atomic Particles:
Protons -
Positive charged
1 Mass
Located in the nucleus
Electrons -
Negative charged
Very small mass
Located on the outer shells of the nucleus
Neutrons -
Neutral charged
1 Mass
Located in the nucleus
Number of Neutrons = Mass Number - Atomic Number
Mass Number - The total number of protons and neutrons
Atomic Number - The number of protons (Number of protons = Number of electrons

If an atom loses electrons, it forms a positive charge due to the fact that there are now more protons than electrons
If an atom gains electrons, it forms a negative charge due to the fact that there are now more electrons than protons
Isotopes - Atoms of the same element can have different numbers of neutrons