Yr 1 - Organic Chemistry
Section 3 Organic Chemistry 1
Chapter 11 Introduction to Organic Chemistry:
Carbon forms rings and very long chains, which can be branched: 4 bonds, where C-C and C-H are both very strong and non-polar
The rules to naming compounds:
Root: longest unbranched hydrocarbon chain
Prefixes and Suffixes: Where prefixes represent the number of carbons on side chains, and suffixes show any additional functional groups (-ene and -ane)
numbers in from of the name: Represent the position of the side carbon chains
Compounds with more than one functional groups are wriiten in the number of position relative to the functional groups priority
Homologous series: A family of organic compounds with same functional group but different carbon chain length
They have same general formulea
The length of the chain has little effect on the chemical properties
The length of the chain has has effect on physical properties
(Structural isomerism => Positional, Functional and chain) Isomers: All have the same molecular formulea, but different structural formulea
(Stereoisomerism => (cis Z same) and (trans E diff) Isomers: All have the same molecular formulea, but the atoms are arranged in different volume of space
(Stereoismoerism => Optical) Isomers: All have the same molecular formulea, but the atoms are arranged in different volume of space.
Chapter 12 Alkanes
About alkanes and properties:
Alkanes are saturated hydrocarbons, so no C=C. They contain C-H and C-C bonds which are non-polar as they have little difference in electronegativity. So, they have weak intermolecular forces, only longer chains have higher van der waals.
KEY POINT:
As branching increases less surface contact, so weaker effect of wan der waals so lower BP and MP
Insoluble in water, because water molecules held together by hydrogen bonding which is stronger than van der waals forces between the hydrocarbons. BUT, alkanes do mix well with other non-polar molecules.
Fractional distillation of crude oil (mixture of long and short chain alkanes): Using Fractionating TOWER
The crude oil is heated, so vaporises and enters the fractionating column.
The fractionating column is hotter at the bottom and cooler at the top
So, the smaller chain hydrocarbons have weaker van der waals so have lower boiling point and are condensed at the top of the column
While the longer chain hydrocarbons have stronger van der waals so has higher boiling point and is condensed at the bottom of the column
Cracking: To form shorter hydrocarbon chains which are more flammable and less smoky
Thermal cracking: Mostly Alkenes formed
High temperatures, high pressure, no catalyst
Catalytic cracking:
High temperatures, moderate pressure, Zeolite catalyst (honeycomb structure for high surface area to volume ratio)
Pollutants:
High temperature in the engine:
N2(g)+ O2(g) —>2NO(g)
carbon dioxide formed form the complete combustion
Carbon monoxide fromed from incomplete combustion
Carbon particulates causes smog
Sulfur dioxide is formed by sulfur impurities in fossil fuel
Flue gas is a process of removing sulfur dioxide, using calcium oxide (lime)
SO2(g) + ½ O2(g) + H2O(l) —> H2SO4 (l)
CaO(s) + 2H2O(l) + SO2(g) + ½ O2 —>CaSO.2H2O(s)
Catalytic converter is a honeycomb structure (Platinium and Rhodium => catalysts), that reduce the output of carbon monoxide, nitrogen oxides and unburnt hydrocarbons.
2CO(g) + 2NO(g) —> N2(g) + 2CO2(g)
Hydrocarbons + nitrogen oxide —> nitrogen + carbon dioxide + water
From Alkanes → Halogenoalkanes
Reaction meachanism: Free radical substituation
Free radical: A chemcial species with an unpaired electron - highly reactive
Conditions: High temperatures and UV light
The steps: Initiation (where the bond breaks homolytically, meaning the bond breaks evenly) → Propagation 1,2 → Termination
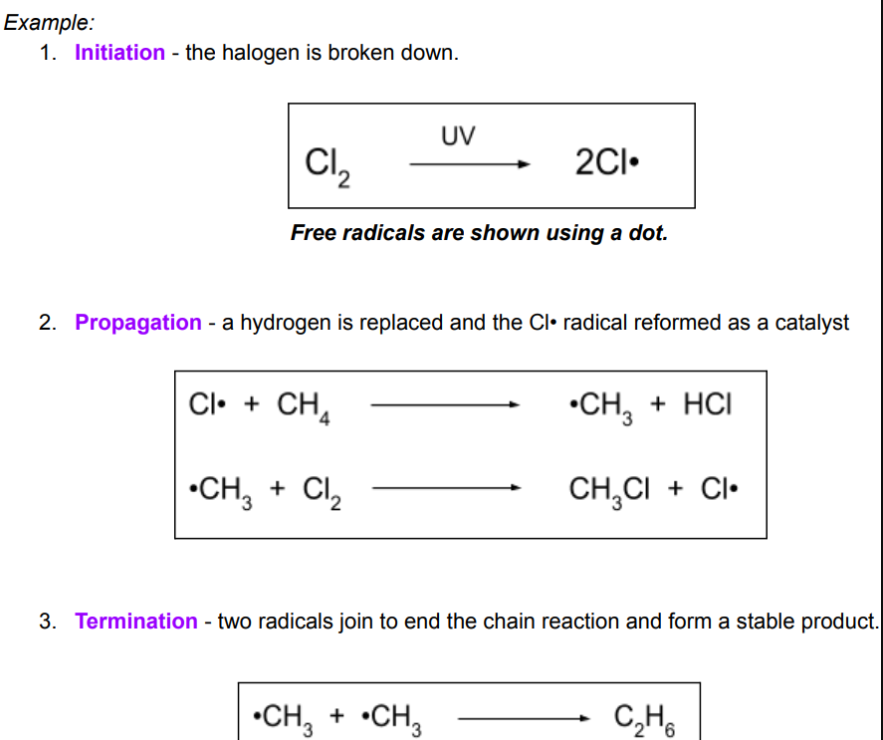
Ozone protects the earth from UV light, which can cause skin cancer. Ozone is attacked by Cl radicals which act as catalysts as they speed up the rate of reaction without being used up.
Chapter 13 Halogenoalkanes => bonding of van der waals and dipole-dipole
C-X (where X = halogen) bond is polar because there is a higher difference in electronegativity where X pulls a pair of electron in covalent bond closer to itself than C, so X is partially negative and C is partially positive.
Boiling point depends on increased chain length and halogen going down the group (more effect of van der waals so higher BP)
KEY POINT:
The reactivity of the C-X bond depends heavily on the bond enthalpy which decreases down the group increasing the rate of reaction down the group.
The reactivity of the C-X bond depends less on the polarity which is highest between C-F
Nucleophile: A negatively charged particle, attracted to positively charged
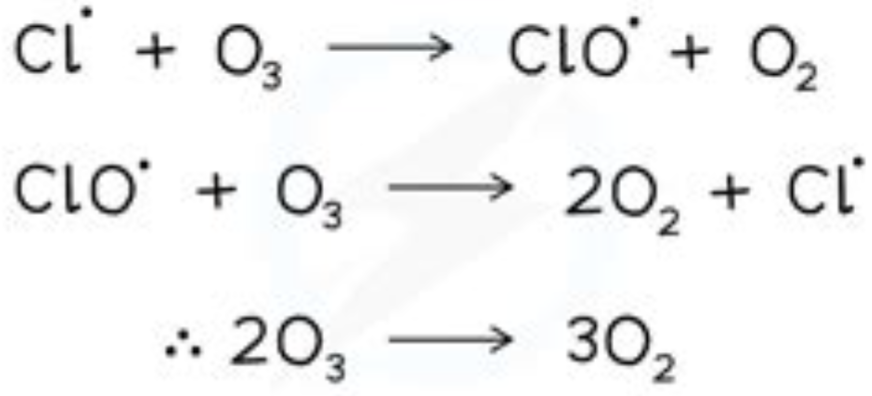
Halogenoalkanes → Alcohols
Reaction mechanism: Nucleophillic substitution
Reagents: KOH
Conditions: warm Aqeous
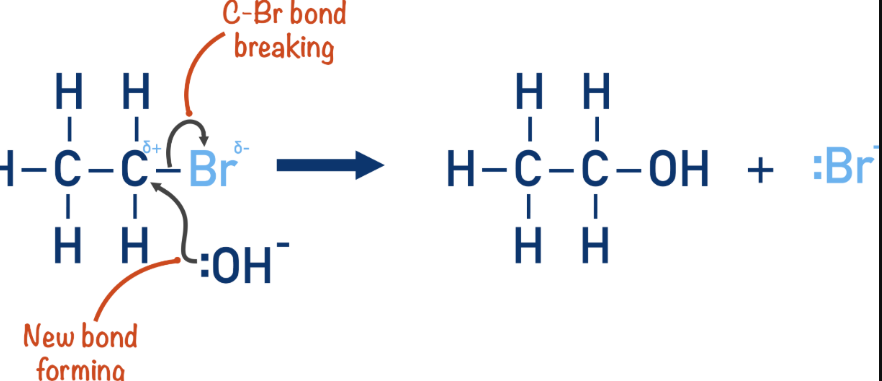
Halogenoalkane → nitrile
Reaction mechanism: Nucleophillic substitution
Reagents: KCN (additon of an extra carbon - increasing the carbon chain length)
Conditions: warm, Ethanolic
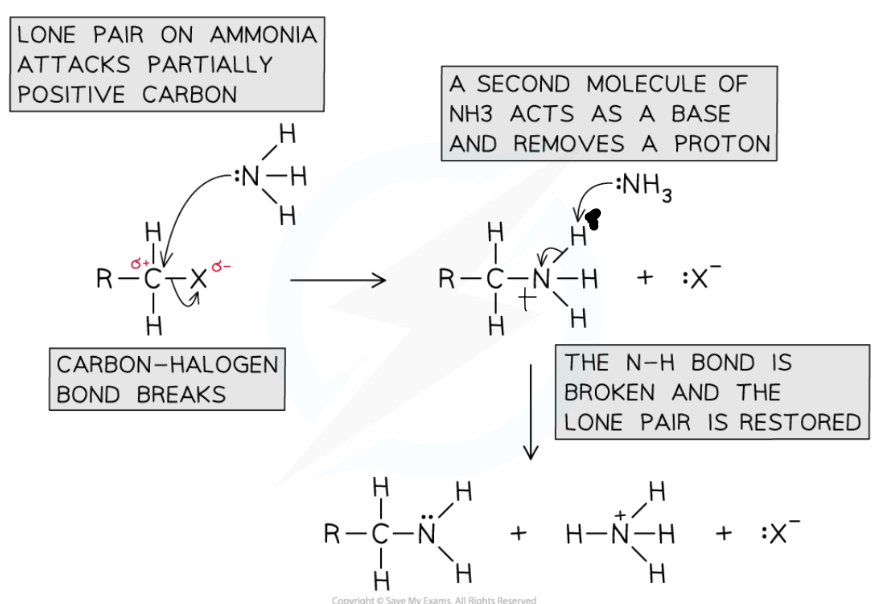
Halogenoalkane → Amine
Reaction mechanism: nucleophillic substitution
Reagent: NH3 Ammonia
Conditions: Excess ammonia, in Ethanolic solution
THE POSITIVE ON THE NITROGEN
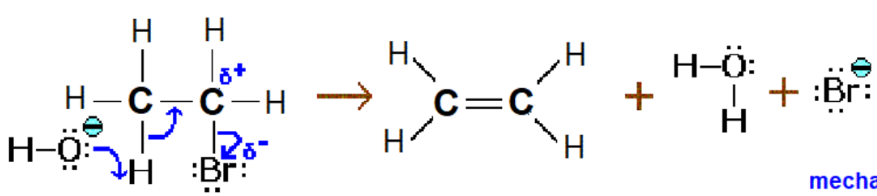
Halogenoalkane → Alkene
Reaction mechanism: Elimination
Reagent: KOH
Conditions: Hot and Ethanolic
Chapter 14: Alkenes
They are unsaturated hydrocarbons, since they have one or more C=C, so the compound does not hold maximum number of hydrogen. The double bond cannot rotate because of the pi-orbital which restricts the rotational movement.
The physical property of alkene is not differented as much as the reactivity. Alkenes are more reactive because the double bond present contains a high electron density (enabling reaction from electrophiles). So, most of the reaction is electrophillic additions.
Alkenes → Halogenoalkane
Reaction mechanism: Electrophillic addition
Reagent: Hydrogen halide
Conditions: -
The steps:
The electrophile (more + charged) is attracted towards the double bond
A positive carboncation is formed (intermediate mostly more stable)
The negatively charged ion then forms a bond with the carbon cation
How to determine the major product?
A major product is formed via a more stable carbon cation
Where tertiary > secondary > primary
Due to the Positive inductive effect, as more alkyl groups makes the compound more stable
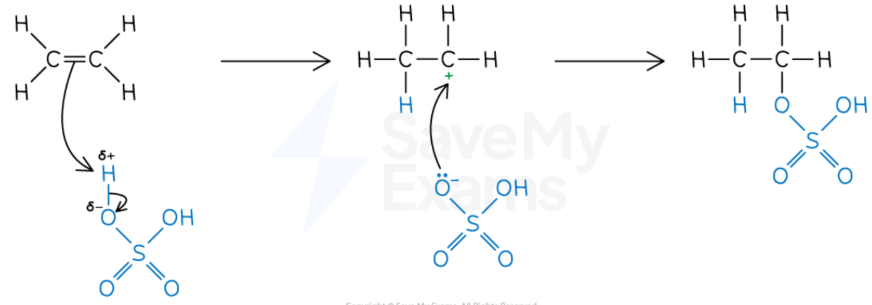
Alkene + conc sulfuric acid
Process of polymerisation
poly(monomer)
Very long chains, so high number of e-, so high van der waals forces
If there is dipole-dipole or hydrogen bonding, this causes strong intermolecular forces.
So they make solid items (strong items)
Addition polymers are inert (unreactive), because of the carbon backbone
Modifying flexibility by adding plasticisers, that are small molecules that force between the molecules making it more flexible
Not biodegradable: C-H not reactive so not attacked by biological agents
Chapter 15 Alcohols
Properties:
-OH bond in alcohols, means there is hydrogen bonding between the molecules, this allows for a high boiling point.
The OH region of the molecule is polar, but the hydrocarbon chain is non-polar. So, shorter chain alcohols are soluble in water, and longer chain alcohols are insoluble - because the non-polar hydrocarbon chain dominates.
The production of Ethanol:
Ethanol from crude oil:
Industrially making ethene from cracking crude oil
Ethene + water with catalyst (phosphoric acid) gives ethanol
Ethanol from fermentation:
Enzymes from the yeast undergo anaerobic respiration that form ethanol and carbon dioxide
Conditions: anaerobic and warm (35 degrees)
C6H12O6(aq) —> 2C2H5OH(aq)+ 2CO2(g)
Advantages and disadvantages:
Advantages of Cracking:
Fast reaction
continuous reaction but Fermentation is batch process
Purer Ethanol formed
Disadvantages of Cracking:
Non-renewable resource
not carbon neutral
Lots of energy required
Electrophillic addition (alkene → alcohol)
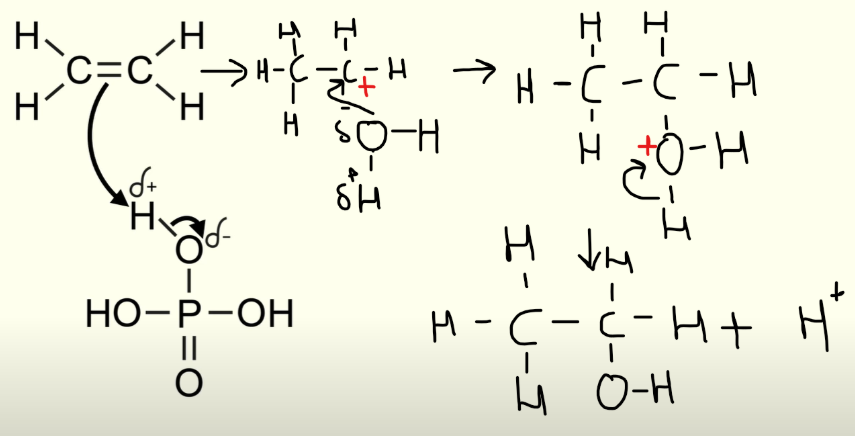
Dehydration of alcohol: concentrated sulfuric acid
Elimination
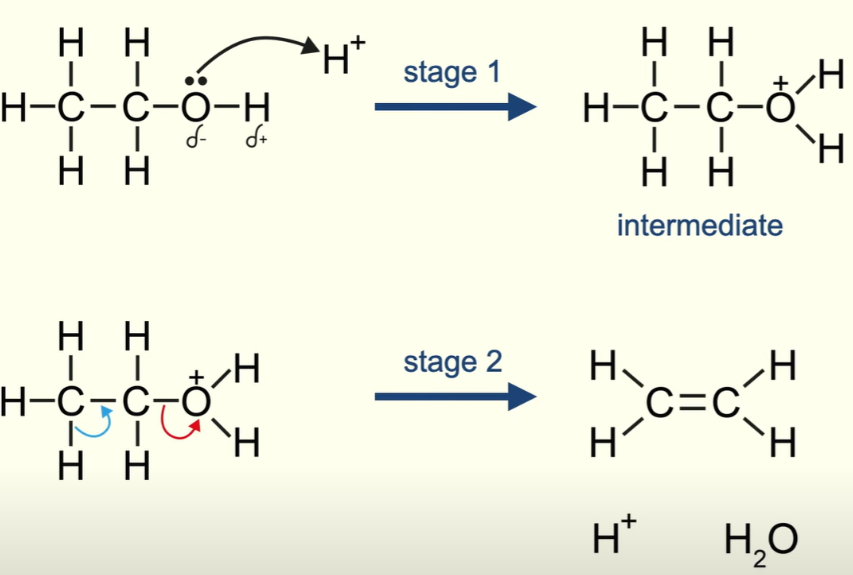
Oxidation of alcohols:
Primary alcohols → Aldehydes → Carboxylic acid
Required acidified potassium dichromate (turns orange to green)
Each requiring 1[O]
To form aldehydes (fractional distillation)
To form carboxylic acid (reflux)
Secondary alcohols → Ketone
Required acidified potassium dichromate (turns orange to green)
reuqire 1[O]
Under reflux
Tertiary alcohols → NOT OXIDISED
Because the c-c is too strong to be broken
Tollens silver test: Silver mirror for Aldehydes (Ag ions reduced)
Fehlings test: Blue to brick red (Cu2+ → Cu +) where copper reduced only in aldehyde
CH 16 Organic analysis (mostly in inorganic 1 notes)
Infrared spectroscopy:
A beam of infrared radiation containing a spread of frequencies is passed through a sample
The radiation that emerges is missing the frequencies that correspond to the types of bonds found in the sample
Since each molecule vibrates differently
And a graph is formed (transmittance) => that plost the intensity of radiation against the frequency
x-axis (wavenumber) and y-axis (transmittance)
Any molecules with more than one element absorbs infrared light at different frequencies, because of the way that the molecule vibrates.
We can get the infrared spectroscopy data, by shining light at different frequencies, where if the wavelength is absorbed by the molecules then the level of transmittance will be low (calling this a peak).
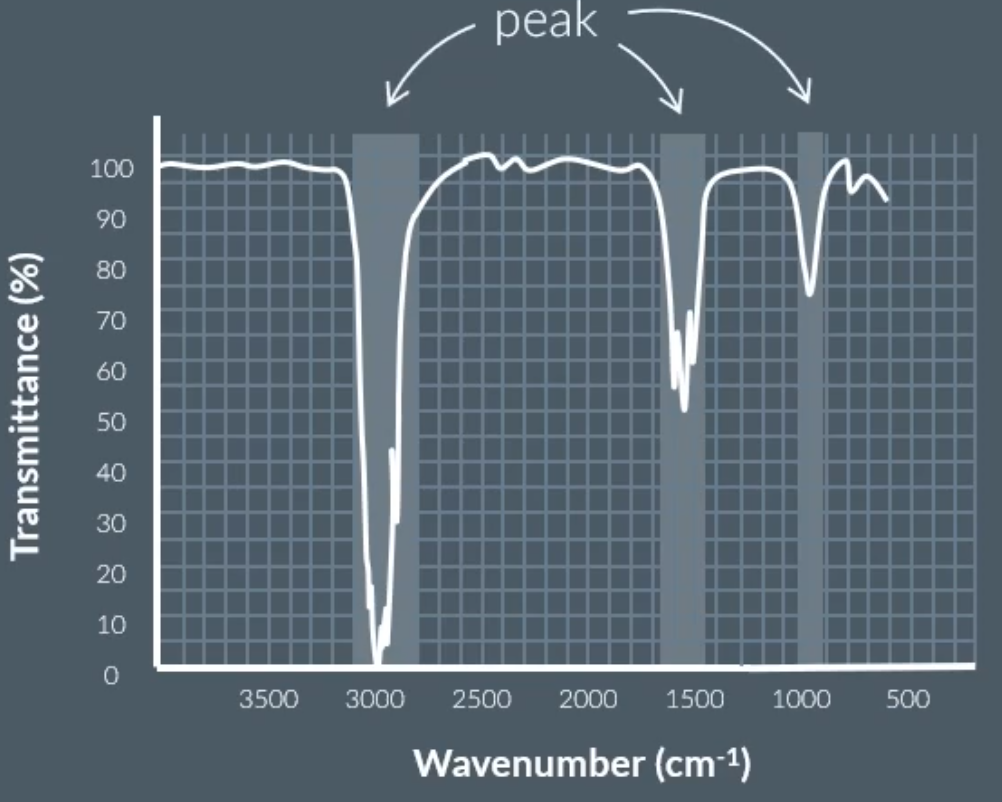
The ones with low transmittance, we can identify the bonds present in a molecule.
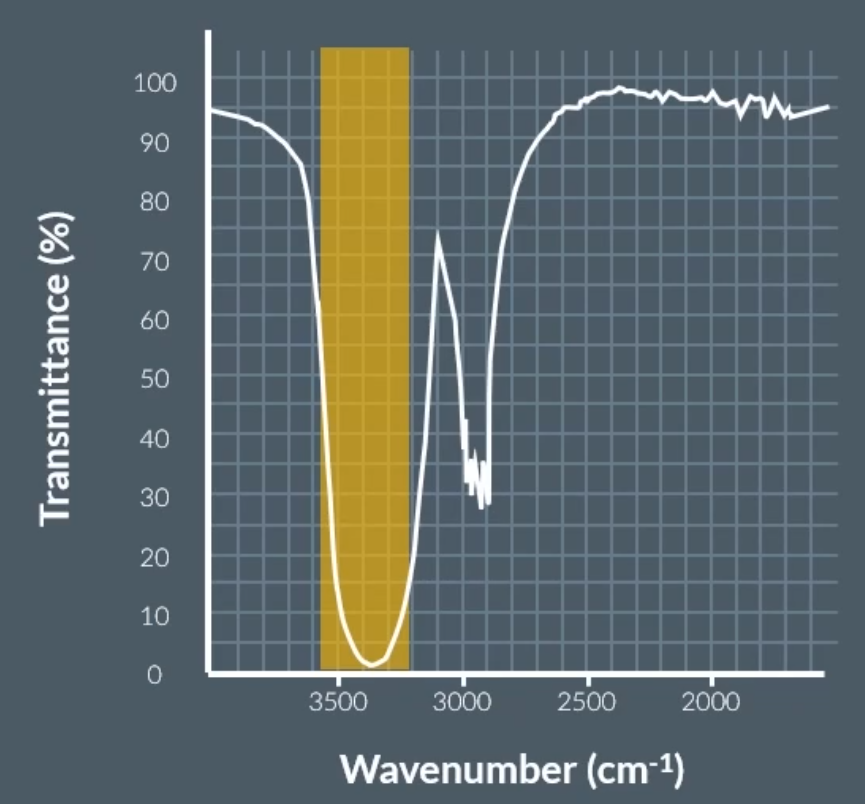
O-H bond in alcohol: (whale head)
O-H bond in carboxylic acid: (mountain range)
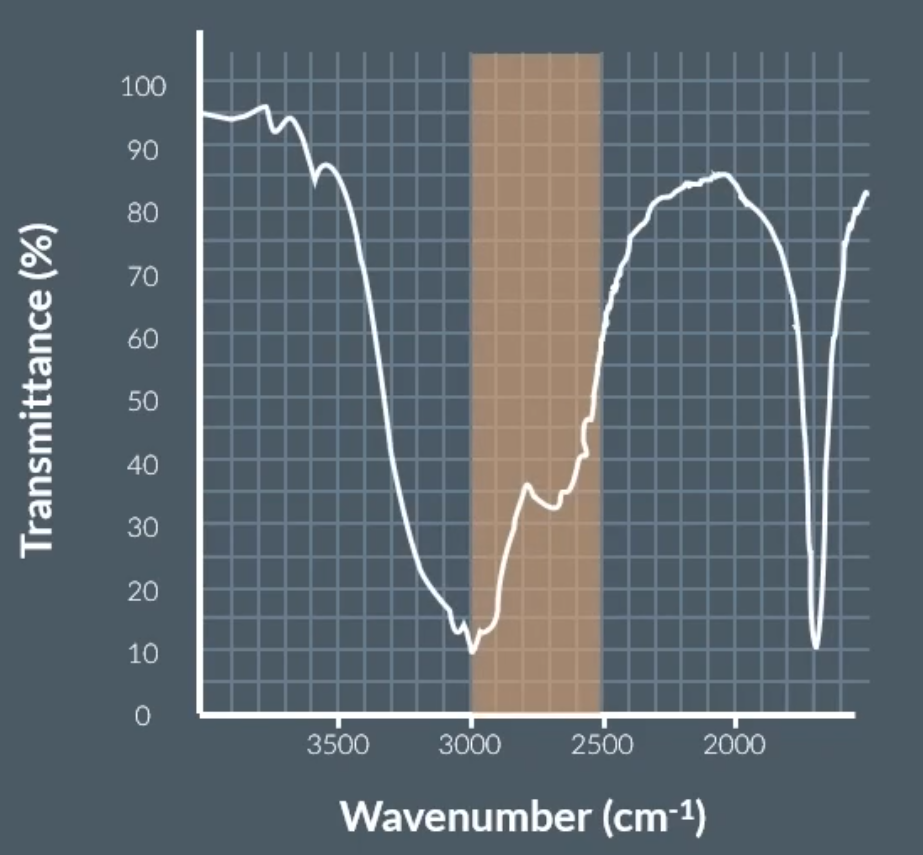
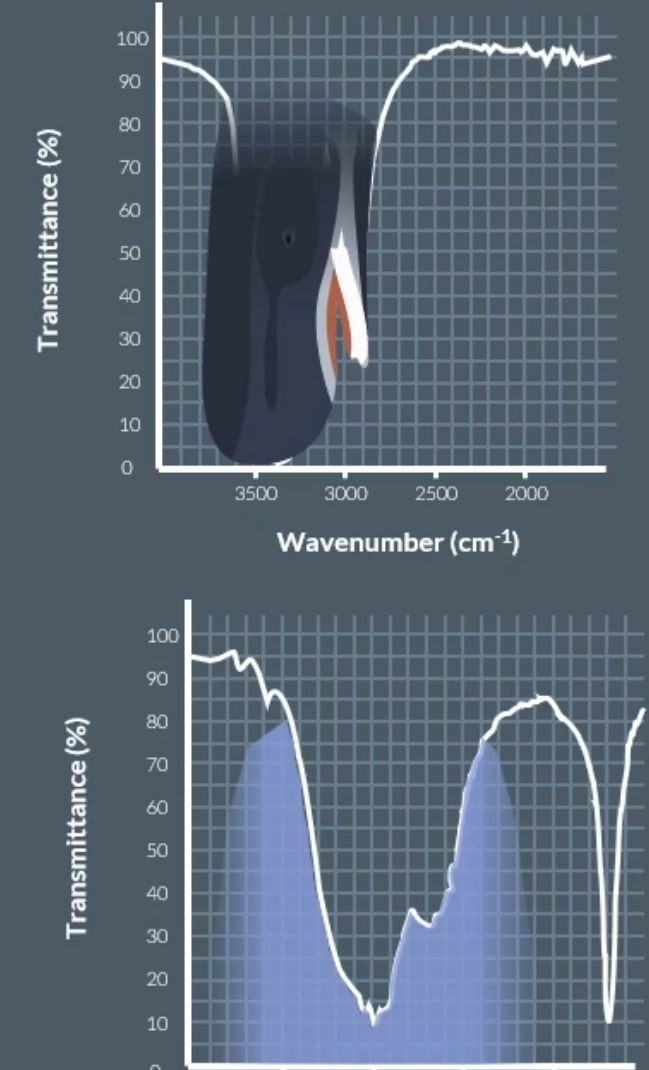
Chemists can use the fingerprinting region which is unique to each molecule => below 1500 cm