Chemical analysis
Purity and formulations
Chemical analysis is the different instruments and methods used to separate, identify and quantify different substances
A pure substance contains only one type of compound or element
For example, pure water with only H and O atoms
They melt and boil at specific temperatures, like 100 degrees for water
Their specific temperatures can be used to find what solutions they are - a physical test
Tests that react substances with other chemicals are known as chemical tests
Impure substances, like salt water, are a mixture of two or more different compounds
They melt and boil over a range of temperatures, dependent on the ratio between the two substances
Formulations are mixtures that have been prepared by using a specific formula
They have precise amounts of different substances, and have a particular function
For example, fuels, paints, medicines or alloys
Different components are always present in the same proportions
Each component contributes a different property
Tests for gases
Chlorine is tested for by placing damp blue litmus paper in a test tube with the sample of gas
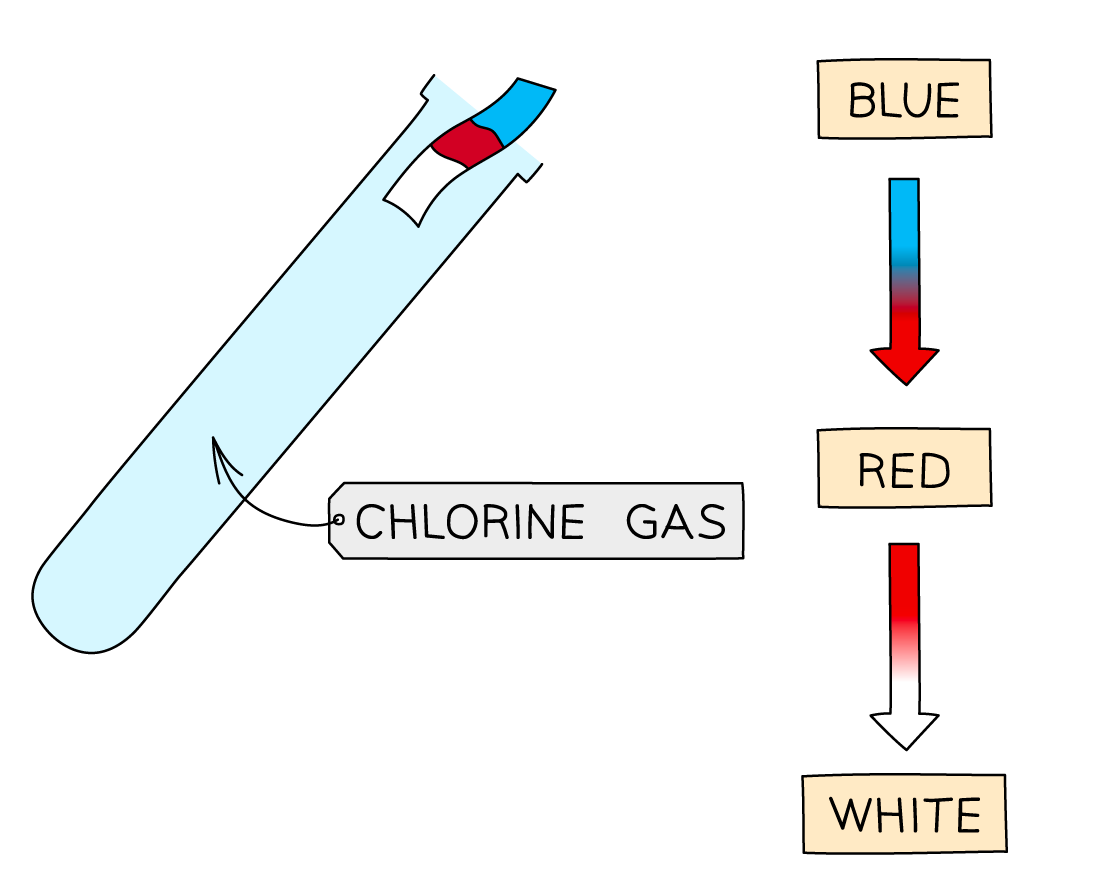
If chlorine is present, the paper turns white
Chlorine is poisonous, so a mask should be worn, and the experiment done in a fume cupboard
Oxygen is tested for by placing a glowing splint in a test tube of the gas
If it relights, oxygen is present
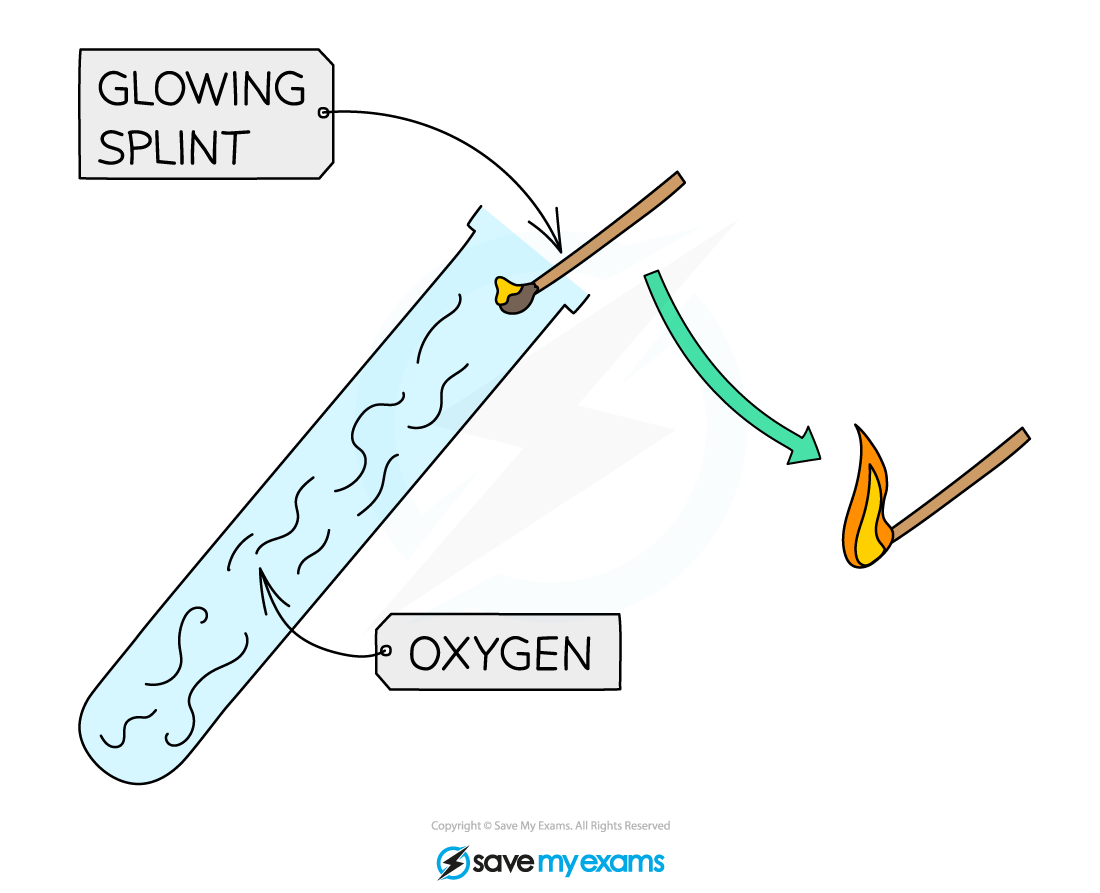
Hydrogen is tested for by placing a burning splint above a test tube of the sample
If present, there will be a squeaky pop sound!

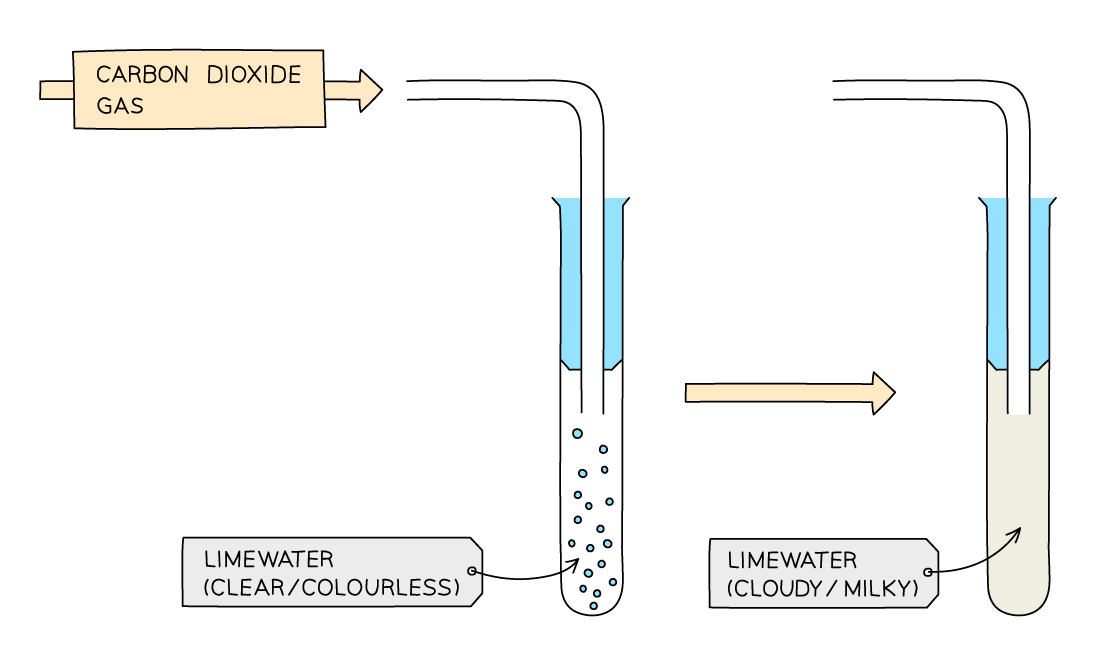
Carbon dioxide is tested for by bubbling the gas in a limewater solution
If present, the limewater goes cloudy
This happens as the carbon dioxide and calcium carbonate react to produce water and CaCO3, a solid with particles that are visible
Paper chromatography
Chromatography is used to separate substances in a mixture
Paper chromatography can be used to separate dyes in an ink

The solvent doesn’t touch the line or the ink, but comes up the paper, and the lid is added to prevent evaporation
The solvent is seeped up by the paper, and it separates it’s colour
They move at different rates
Leave the paper to dry to create a chromatogram
The mobile phase is where the molecules move in, as a liquid or gas, and is the solvent in the chromatography
The stationary phase is where molecules can’t move, the paper in the chromatography
The different molecules in the ink constantly change phases
They dissolve, move a bit, bind to the paper, etc.
The more soluble they are, the faster they move
If they are less soluble, they are more attracted to the paper, so move slower
How far each chemical travels is dependent on the properties of that sample
A sample will have the same results every time
We can compare the distance moves per chemical with a data log to find and identify the chemical
However, the distanced travelled is dependent on the time, so we use an Rf value
Rf value = distance travelled by substance (individual)/distance travelled by solvent (overall)
If a pure substance is used, there will be a single dot only shown on the chromatogram
Mixtures have multiple
If the paper or solvent changed, results would differ and therefore the Rf value as mobile/stationary stages have changed
This has to be taken into account when comparing results
Testing for anions
Anions are negatively charged ions
Carbonates
A carbonate and an acid react to form a salt, carbon dioxide and water
We can test for carbon dioxide, which proves a carbonate was a reactant
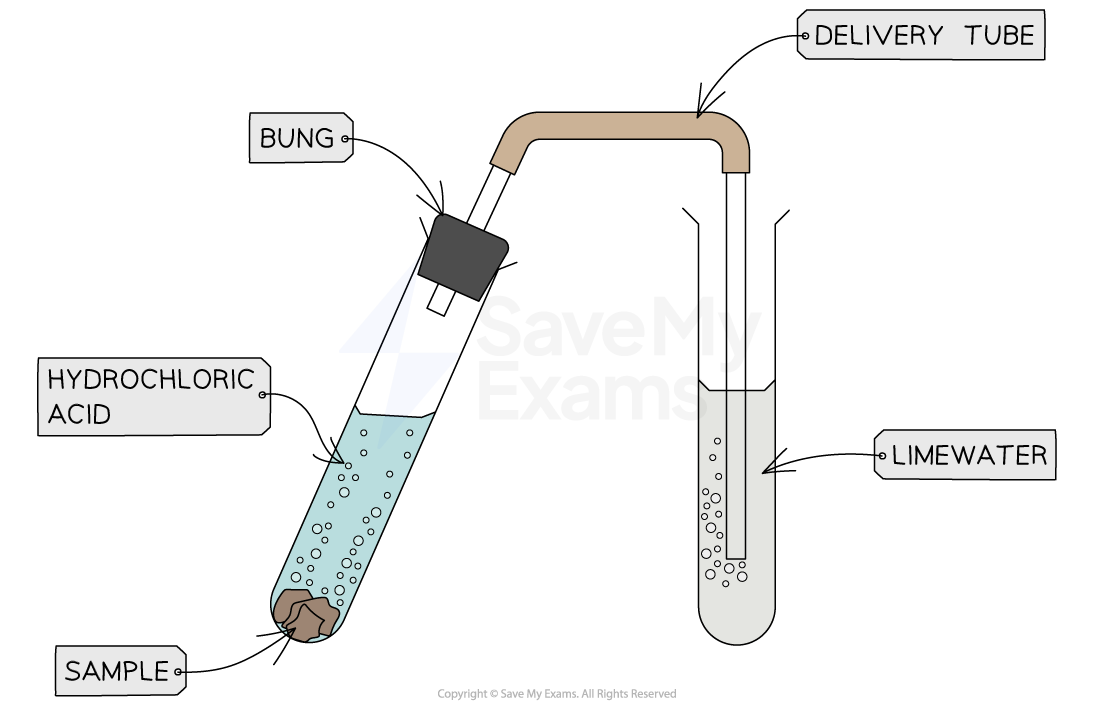
Dilute HCl and the carbonate sample are reacted together, and the products are bubbled into limewater
If the limewater turns cloudy (CO2 present), a carbonate was reacted
Sulphates
Sulphate ions and barium ions react to form barium sulphate
Barium sulphate is a white precipitate
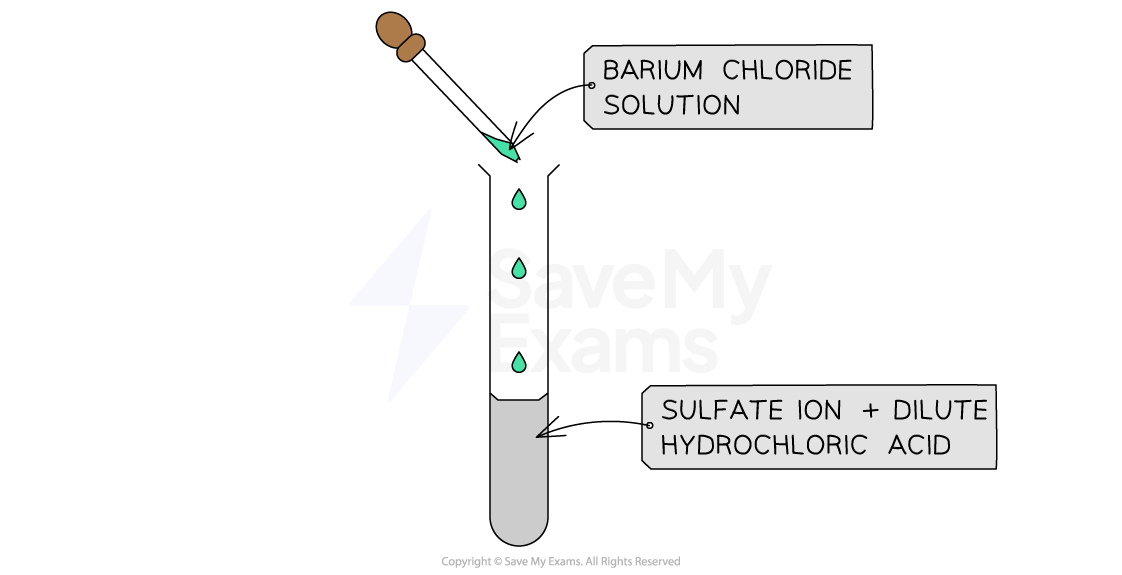
Hydrochloric acid and the sulphate ions are mixed to remove impurities that may effect the result
CO3 or SO3 may also create a white precipitate if present
Barium sulphate is then added
If a white precipitate is formed, sulphate was present as a reactant
Halides
Halides are ions with a negative 1 charge - for example, Cl-, Br- + I-
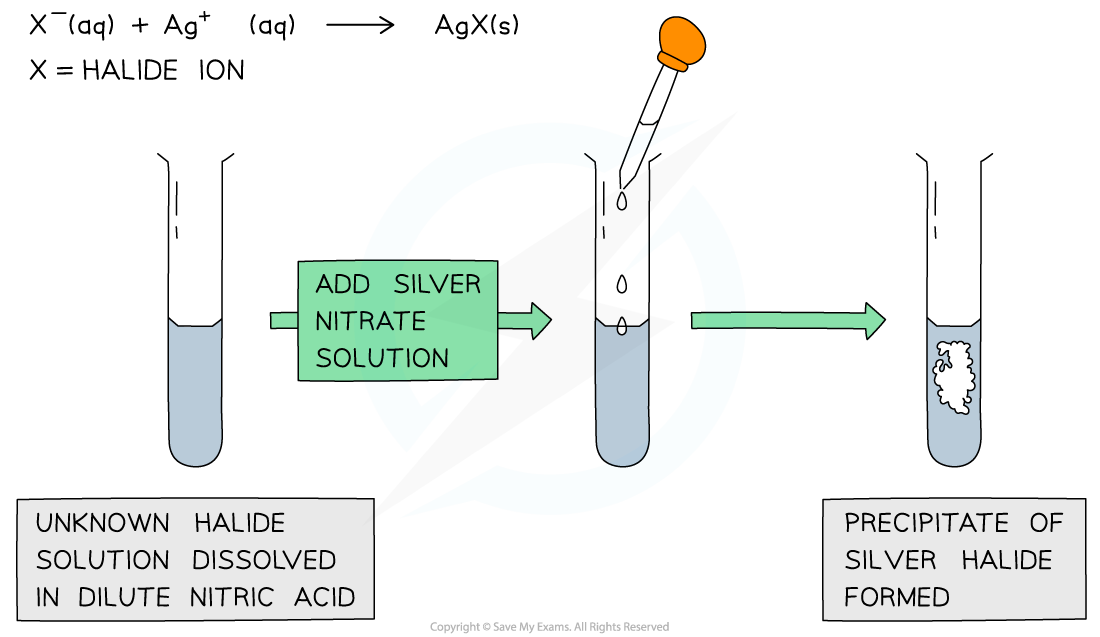
Dilute nitric acid is mixed with the halide to remove impurities - CO32- or SO32-
Silver nitrate is then added, to produce a precipitate if present
Chloride and silver nitrate - white precipitate
Bromide and silver nitrate - cream precipitate
Iodide and silver nitrate - yellow precipitate
Tests for cations
Cations are positively charged ions and they are mainly metals (not NH4+ - ammonium)
Flame tests
Take a platinum or nichrome wire loop and clean by dipping in dilute HCl, rinsing in distilled water and heating over a bunsen burner
Dip the loop into the substance that is being tested
Hold the loop in the clear blue section of the flame
See what colour the compound turns
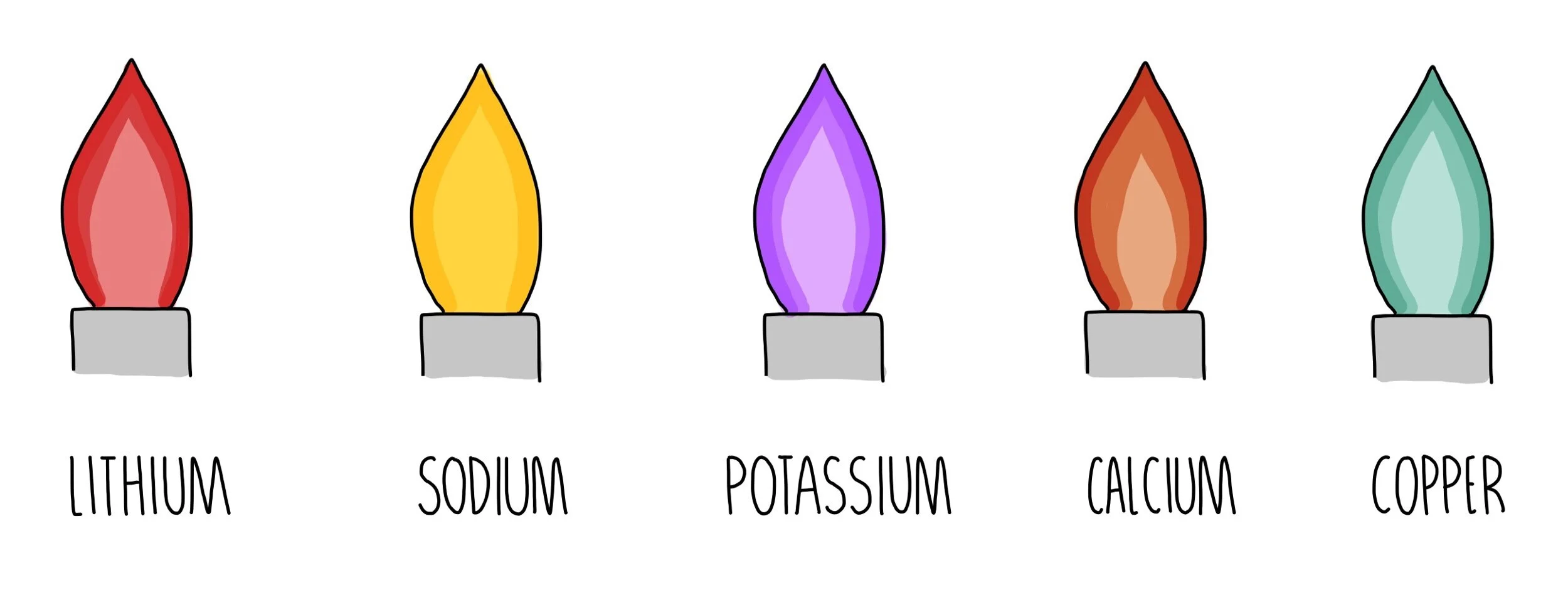
Lithium turns crimson, sodium turns yellow, potassium turns lilac, copper turns orange-red and copper turns green
If there are more than two cations present in the sample, the colours are likely to mix
Metal hydroxide test for cations
Used for metal ions, by reacting them with sodium hydroxide (NaOH)
Possible as some metals form coloured precipitates when reacting with OH ions
Copper forms a BLUE precipitate when reacting with NaOH
Cu2+ + 2OH- → Cu(OH)2 (half equation)
Copper + sodium hydroxide → sodium chloride + copper hydroxide
Calcium forms a WHITE precipitate
Ca2+ + 2OH- → Ca(OH)2
Calcium + sodium hydroxide → sodium chloride + calcium hydroxide
Iron(II) forms a GREEN precipitate
Fe3+ + 2OH- → Fe(OH)3
Iron + sodium hydroxide → sodium chloride + iron hydroxide
Iron(III) forms a brown precipitate
Fe2+ + 2OH- → Fe(OH)2
Iron + sodium hydroxide → sodium chloride + iron hydroxide
Magnesium forms a WHITE precipitate
Mg2+ + 2OH- → Mg(OH)2
Magnesium + sodium hydroxide → sodium chloride + magnesium hydroxide
Aluminium forms a white precipitate, which turns colourless if NaOH is in excess
Al3+ + 3OH- → Al(OH)3
Aluminium + sodium hydroxide → sodium chloride + aluminium hydroxide
Flame emission spectroscopy
When metal ions are heated, they emit light
Wave lengths of light that are emitted are specific to particular metal ions
Spectroscopes detect the individual wavelengths of light that are emitted
Every metal has a different pattern of wave lengths (when heated), which can be used for identification
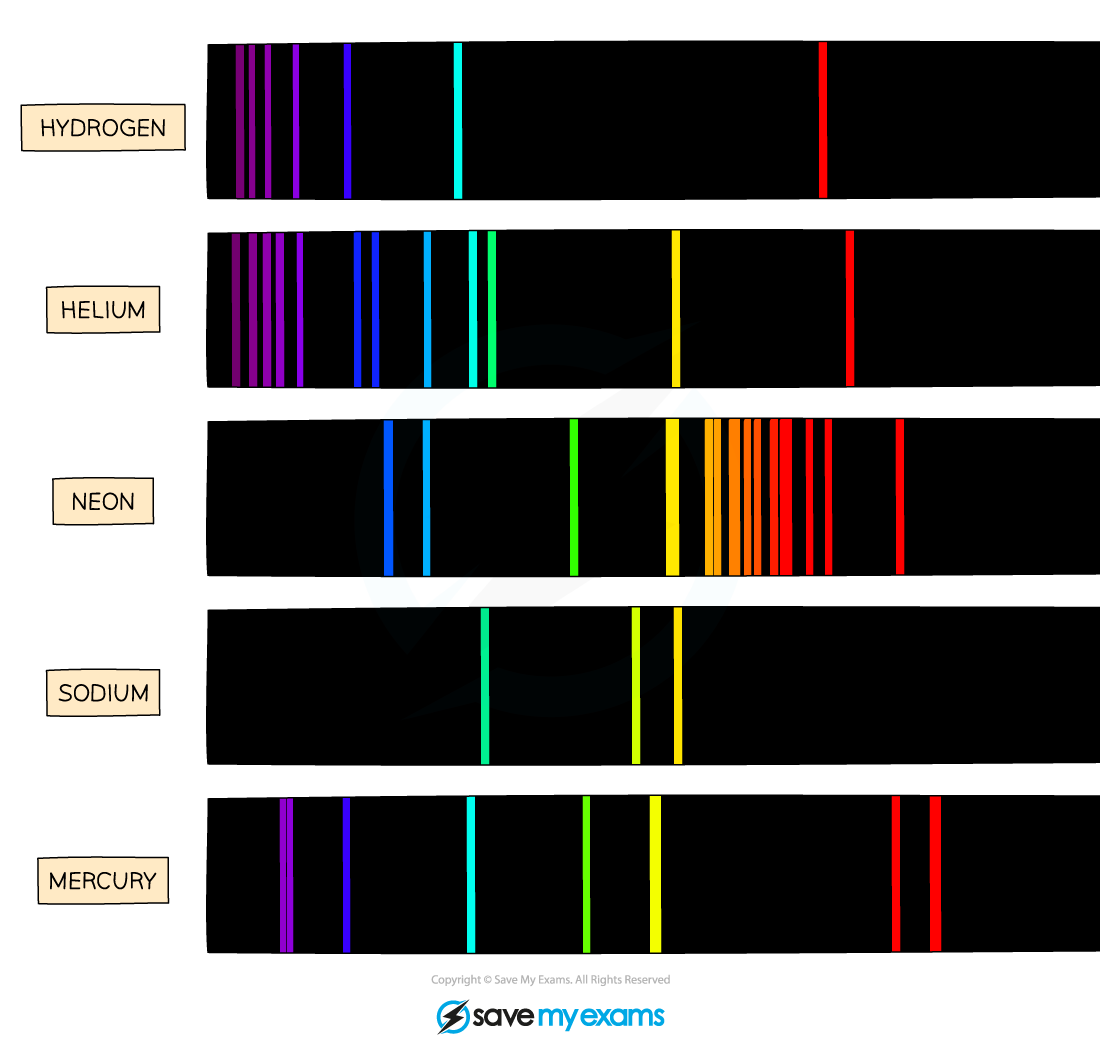
The intensity of the line shows the concentration of the ion, and therefore the concentration of the iron
If a sample contains multiple ions, the spectrum will show the lines for all of them
From this, we can identify both ions and their concentrations
Flame tests (manual test) VS flame emission spectroscopy (instrumental)
Flame tests are basic and cheap, although less accurate
FEM is expensive (the machinery), but also more sensitive to change, more accurate and very fast (can be automated entirely)
DONE!!!