Reactivity 3.1
Brønsted-Lowry Theory - defines acids and bases in terms of proton transfer
Acids = donates protons
donate H+ to water → hydronium ions (H3O+)
Bases = accepts protons
accept H+ from water → hydroxide ions (OH-)
proton - hydrogen ion
aqueous H+ or H3O+ (hydronium)
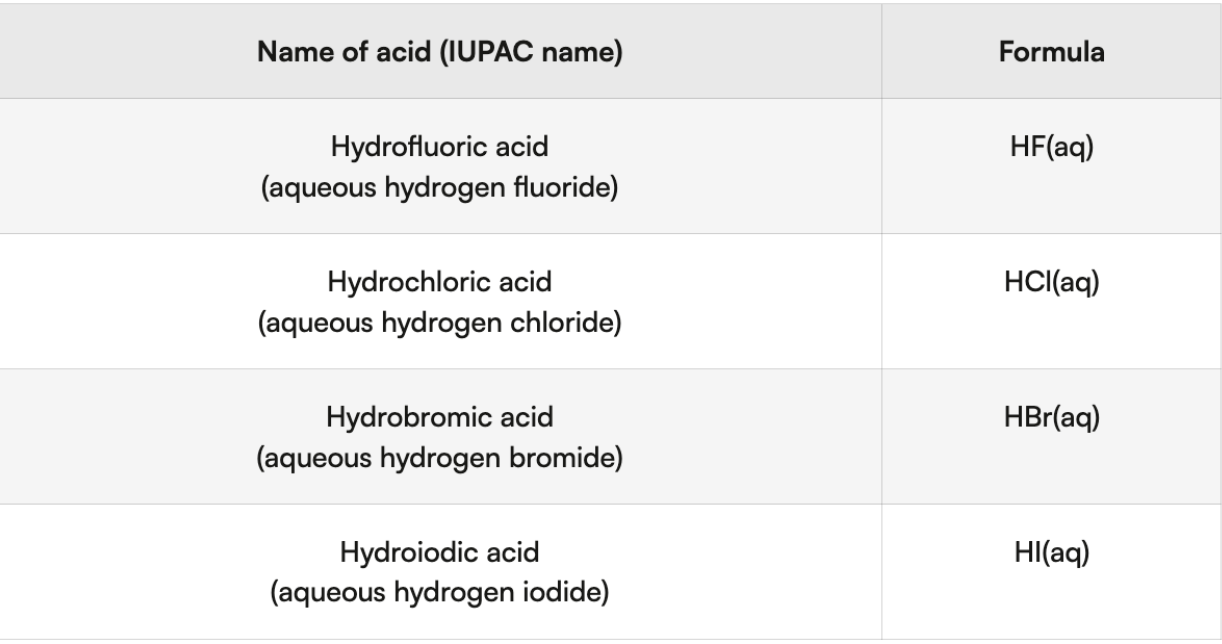
Binary acids - formed when halide reacts w hydrogen cation
Halide = F, Cl, Br, I
Name: “hydro-halogen first syllable of halide -ic acid”
No O → write hydro
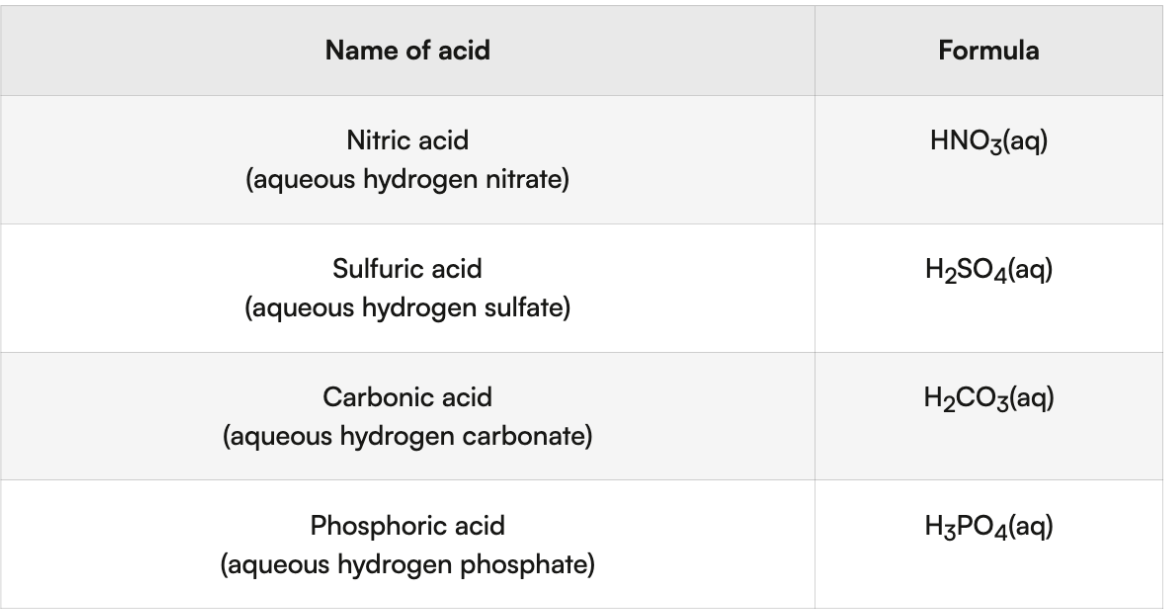
Oxyacids - formed when polyatomic ion reacts w hydrogen
Name: “polyatomic- replace ate with ic- acid”
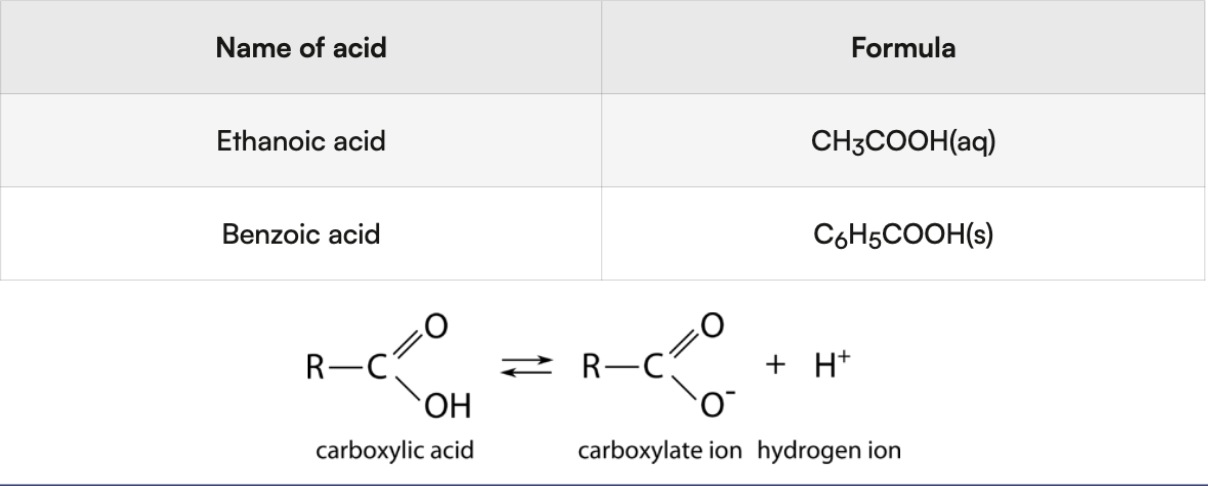
Organic acids - contain carboxyl group (COOH)
H bonded to O in carboxyl group → acidic hydrogen that gets donated
Name: Carboxylic acid IUPAC naming rules
suffix “oic acid”
Not all species with hydrogen behave like acids
ex. Methane
does not donate hydrogens → not an acid
acids classified based on number of protons they can donate
monoprotic - 1 proton
HCL, HNO3, carboxylic acids, etc
diprotic - 2 protons
H2SO4, H2CO3, etc
triprotic - 3 protons
H3PO4, etc
(below are bases)
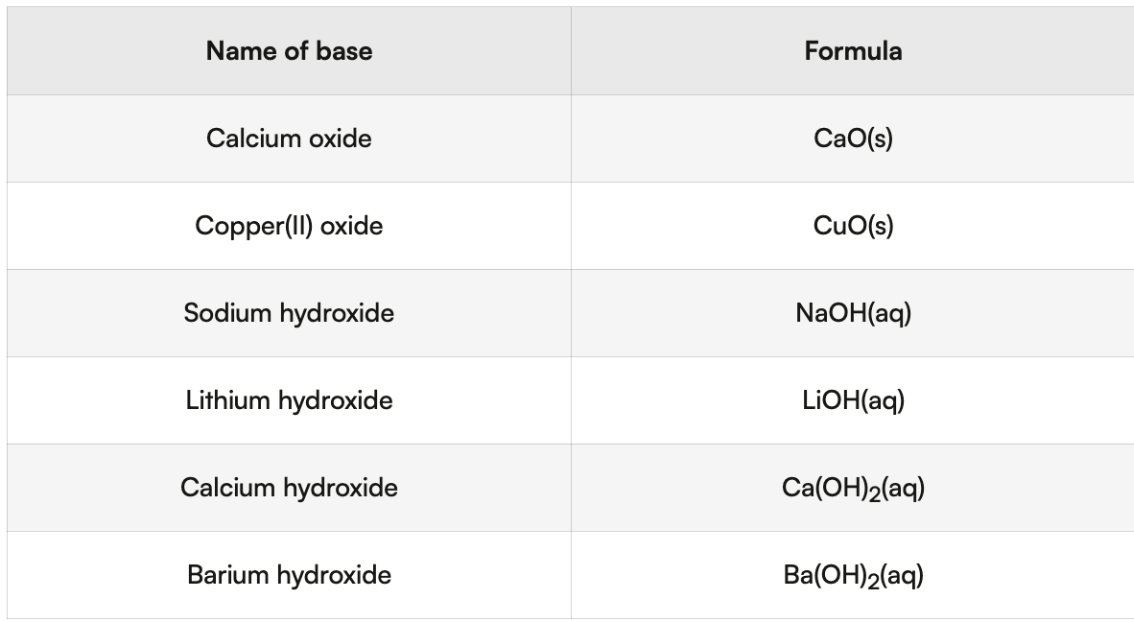
metal hydroxides/oxides
Name: ionic compound naming rules (metal numeral non-metal ide)
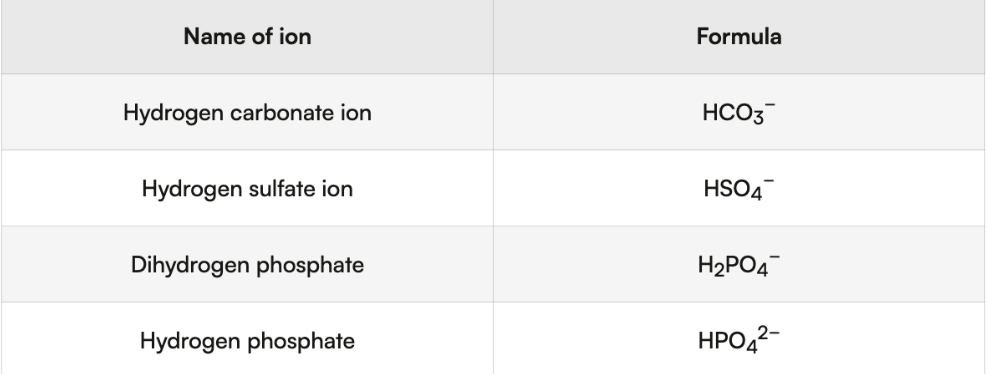
carbonates and hydrogen carbonates
Name: ionic compound naming rules (metal + polyatomic ion)

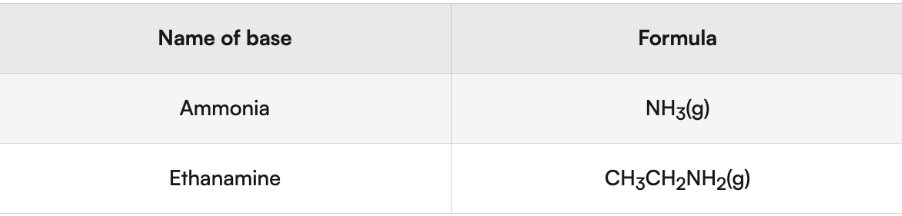
Ammonia (NH3) and amines/amides: nitrogen containing compounds
amine: R-NH2
end in amine
Amide: R-CONH2
end in amide
Nitrogen atom accepts proton → coordinate bond
all acids are aqueous
HCl(g) = hydrogen chloride
HCl(aq) = hydrochloric acid
not all bases dissolve in water
insoluble bases : CaO + Cu(OH)2
soluble base → alkali
all alkali = bases but not all bases are alkali
includes group 1 metal hydroxides
acid/base activity of compounds shown by reacting compounds w water
Acid + H2O → hydronium ion + acid anion
Base + H2O → hydroxide + base cation (dissociation)
organic base + H2O → BH+ + OH-
reaction not w water
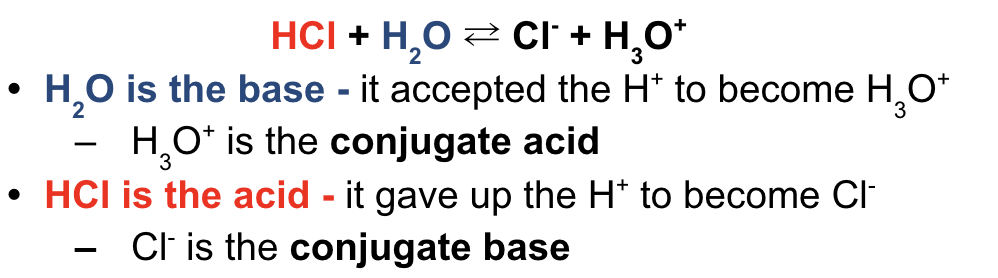
identify acid (donor) and base (acceptor)
show transfer of H+ from acid → base
conjugate acid - species made when base accepts H+ from acid
opposite of base
conjugate base - species made when acid donates H+ to base
opposite of acid
can determine from acid and base for the reverse reaction
assume reaction is going forward unless otherwise specified
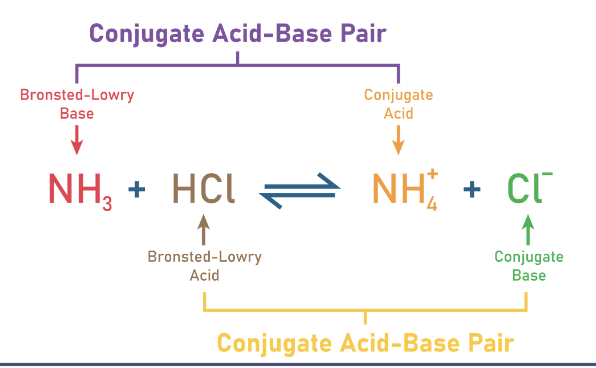
conjugate pair includes 2 species
acid reactant + conjugate base product
base reactant + conjugate acid product
amphiprotic - species acts as both acid and base (water)
amphirotic vs amphoteric
mean same thing
BUT amphiprotic refers to bronsted-lowry acid/bases
amphoteric refers to all acids/bases
B-L, Arrhenius, Lewis, etc
Examples of amphiprotic species
acids react w bases → neutral solutions formed
neutralization reactions ex
acid + metal oxide/hydroxide
produce salt (ionic compound) + water
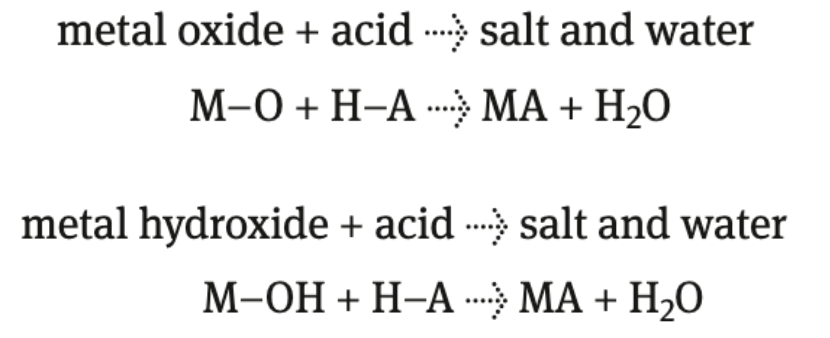
acid + carbonate/hydrogencarbonate
produce salt + CO2(g) + water
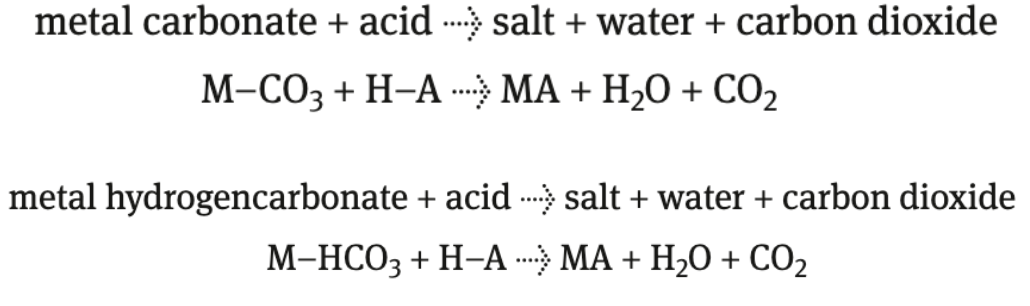
acid + ammonia/amine
produce salt
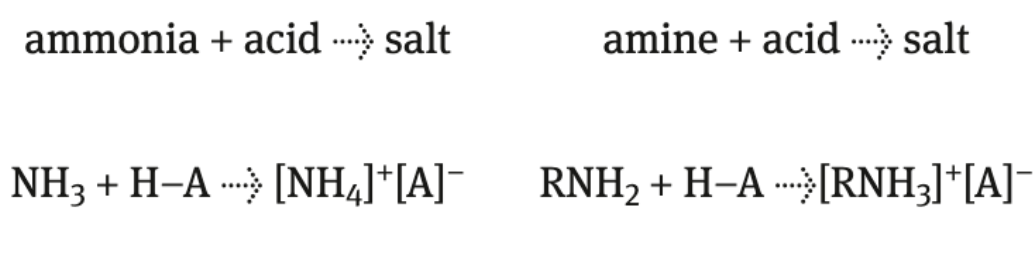
strength of acid/base depends on dissociation (ionization) in water
all acids aqueous
not all bases dissolve/dissociate in water
strong acids/bases → COMPLETELY dissociate
dissociation goes to completion
use →
weak acids/bases → PARTIALLY dissociate
dissociation exists in equilibrium
use ⇄
Strong acids
completely dissociate → splits into individual ions
hydrogen ion written H+ or H3O+
HCl, HBr, HI, HNO3, H2SO4
H2SO4 is diprotic
ONLY the FIRST dissociation is considered strong
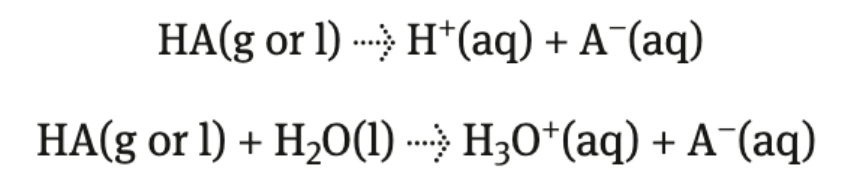
weak acids
organic acids usually weak
carboxylic acids
partially dissociate → dissociation exists in equilibrium → equilibrium lies to the left
Kc < 1
the H bonded to the O in carboxyl group is the acidic hydrogen
only H that gets donated
DO NOT remove H from a carbon/nitrogen
small amount of H+ ions in solution relative to HA
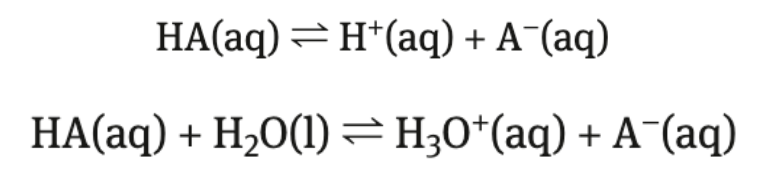
strong bases
completely dissociate → splits into its individual ions
only alkalis dissolve/dissociate in water
group 1 metal hydroxides + barium hydroxide (Ba(OH)2)
dissociation of barium hydroxide produces 2 moles of hydroxide ions
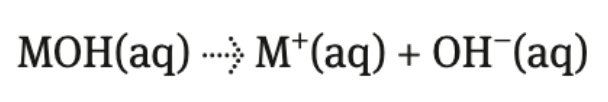
weak bases
bases other than group 1 + group 2 hydroxides are weak
oxides, group 13 hydroxides, carbonates, hydrogencarbonates, ammonia, amines/amides
partially dissociate → dissociation exists in equilibriumm → equilibrium lies left
Kc < 1
nitrogen atom accept the hydrogen → coordinate covalent bond
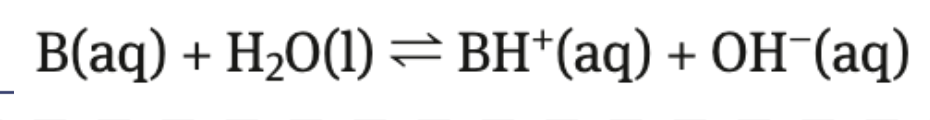
Strong acids/bases → WEAK conjugates
completely dissociate → forward reaction favored → equilibrium lies right
strong species want to dissociate
weak acids/bases → STRONG conjugates
partially dissociate → reverse reaction favored → equilibrium lies left
weak species do not want to dissociate
Distinguishing btwn strong and weak
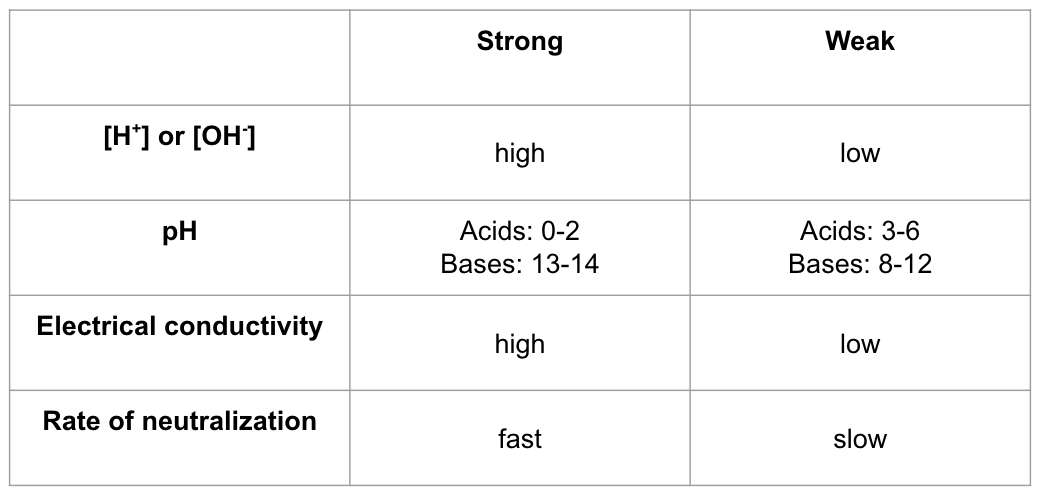
pH scale - determines how acidic/basic an aqueous solution based on concentration of H+ ions in a solution
inverse, logarithmic scale
a change in 1 pH unit → tenfold change in H+ ion concentration
more H+ → lower pH → acidic
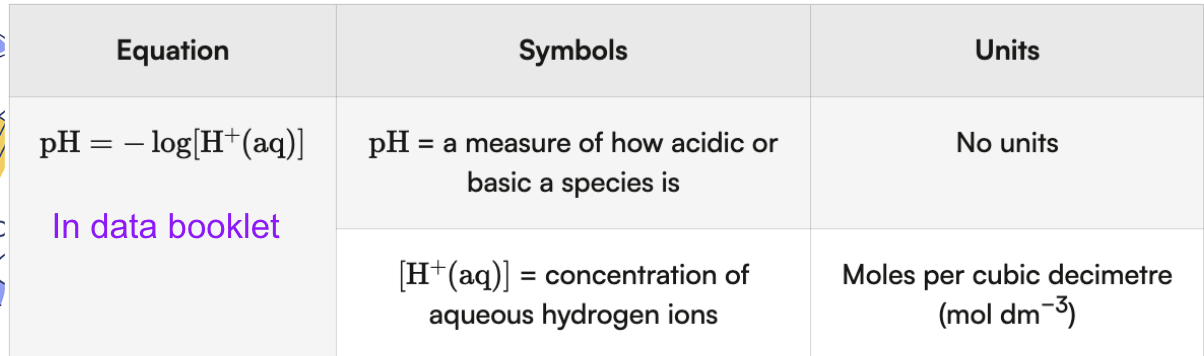
Equation also expressed as: [H+] = 10^(-pH)
Strong acids: [H+] = initial concentration of acid
[H+] of H2SO4 is not 2 times the initial concentration
weak acids: need ICE table to determine [H+]
If given pH of strong/weak acid
put answer in scientific notation
calculating pOH (for bases)
pH still determines acidity/basicity

pH + pOH = 14
only at 298K
Titration - determines concentration of an unknown acid/base solution
fill burette w solution of known concentration (titrant)
add titrant to volume of unknown solution (analyte)
titrate until equivalence point
point where acid and base are stoichiometrically equivalent
solution contains only salt and water
indicator changes color
Occurs btwn
strong acid + strong base
weak acid + strong base
strong acid + weak base
weak acid + weak base
4 different types of pH curves depend on strength of species
neutralization reactions show change in pH over time
data collected
volume of titrant from burette
pH of analyte solution
taken at regular intervals of added titrant volume
data plotted as pH vs volume of titrant → pH curve
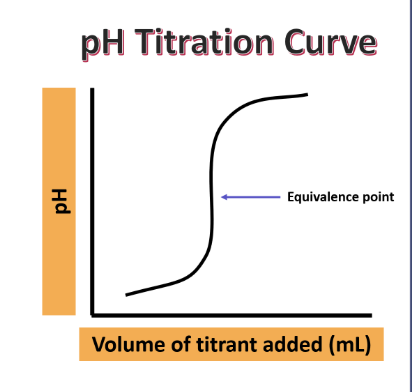
Main pieces of info from pH curve
starting of pH analyte
equivalence point
halfway up steepest part
ending pH of neutralized solution
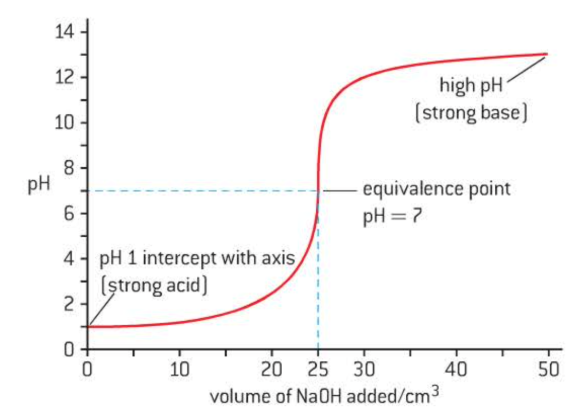
Strong acid + strong base
low starting point = strength of acid
sharp jump in pH at equivalence
equivalence point at pH = 7
high ending point indicates base strength
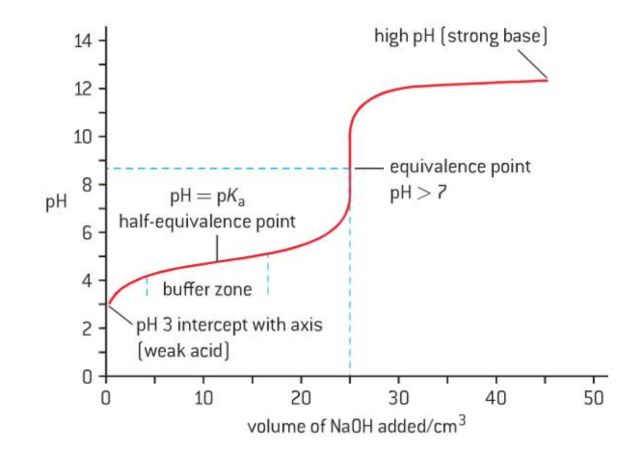
weak acid + strong base
pH > 2 at starting point → weak acid
buffer zone - solution acts as buffer
resits changes in pH
sharp jump in pH at equivalnce
equivalence at pH > 7
high ending point → strong base
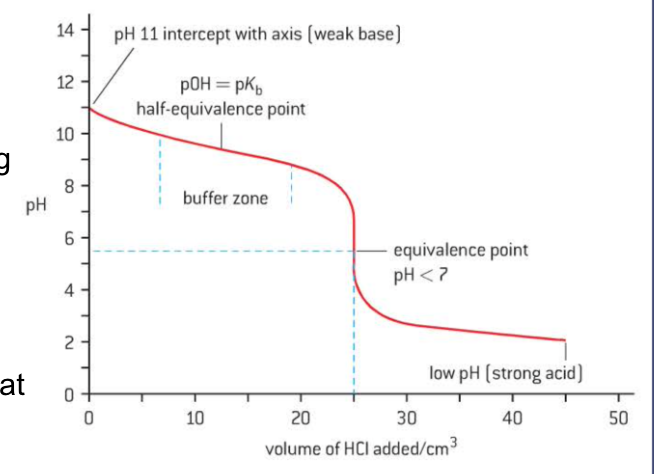
strong acid + weak base
pH < 13 starting point → weak base
buffer zone - solution acts as buffer
resists change in pH
sharp jump in PH at equivalnce
equivalence at pH < 7
low ending point → strong acid
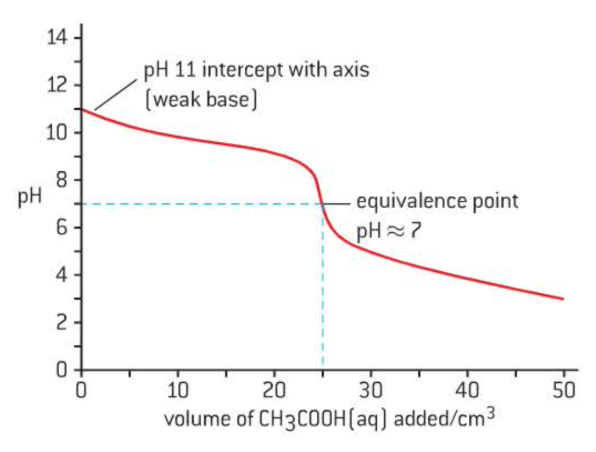
weak acid + weak base
pH < 13 starting point → weak base
gradual change throughout titration
small jump in pH at equivalence
difficult to determine equivalence point but usually ~7
pH >2 ending point → weak acid
titrant and analyte can switch in grpahs
you can add acid → base or base → acid
Water → amphoteric
both weak acid + weak base
partially dissociates → autio-ionization of water
exists in equilibrium → has constant Kw
Kw - ionic product of water
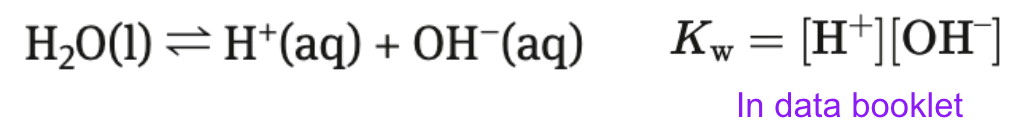
At 298 K (25 C) pure water has
[H+] = 1.00 × 10^(-7)
[OH-] = 1.00 × 10^(-7)
Kw = 1.00 × 10^(-14)
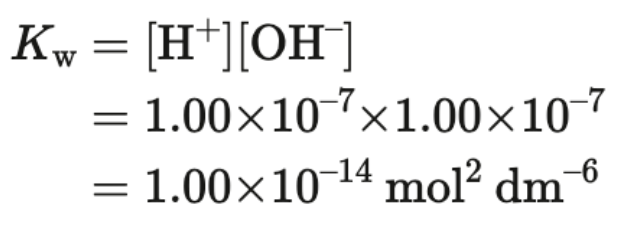
Kw = temperature dependent
only 1.00 × 10^(-14) at 298 K
T increases → pH of pure water decreases
endothermic
shifts right
more [H+]
still neutral
weak acid → Ka
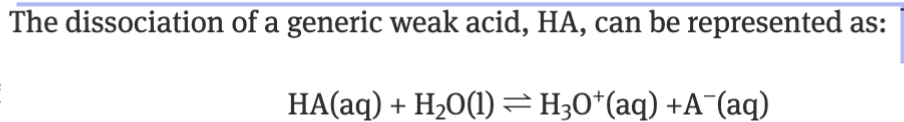
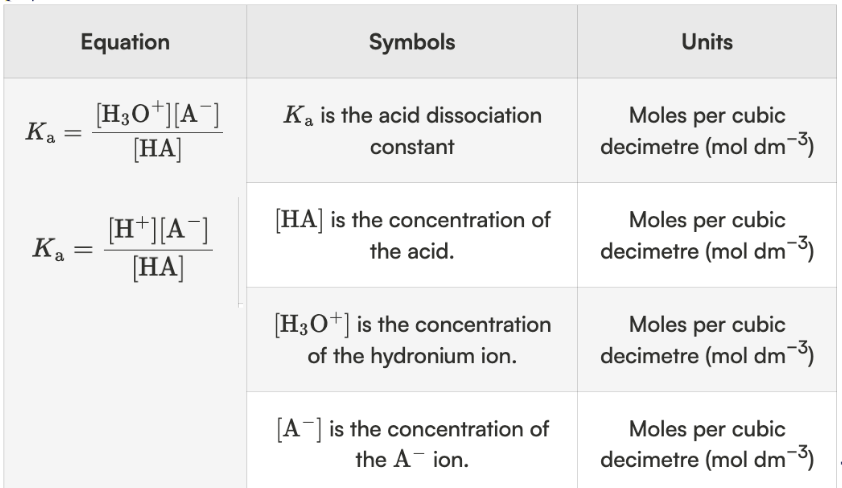
water not included bc it has no concentration
only aqueous species in Ka
weak acids only partially dissociate → Ka < 1
Ka increases closer to 1 → strength of acid increases
weak base → Kb
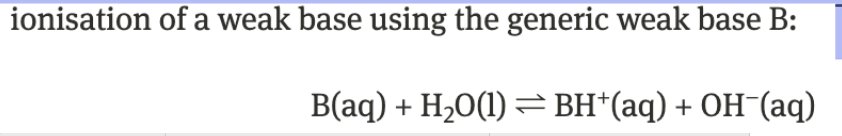

water not included bc it has no concentration
only aqueous species in Kb
weak bases only partially dissociate → Kb < 1
Kb increases closer to 1 → strength of base increases
Ka and Kb → temperature dependent
Le Chat applies
use Ka or Kb to calculate Gibbs
lowercase p in front of variable in chem → function “-log”
equilibirum constants → pKa and pKb
also use pK values to determine Ka or Kb
strength increases → Ka increases
pKa decreases (smaller = stronger)
strength increases → Kb increases
pKb decreases (smaller = stronger)

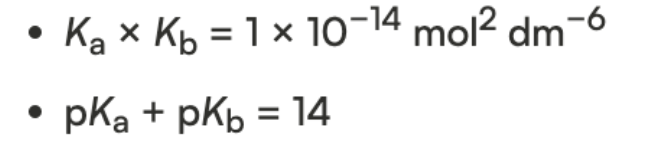
weak acids need ICE tables to determine [H+] at equilibrium
Kc ≤ 10^(-4) → change in reactant is negligible
Ka and Kb usually ≤ 10^(-4) → initial = equilibrium
ONLY IGNORE FOR REACTANT (HA or B)
assume very little dissociation
equilibrium value of products is not negligible or 0
if given weak base and asked to calculate Kb → extra steps to have OH-
use pH to find [H+] or pOH
determine [OH-] equilibrium using Kw or pOH
use an ICE table to determine [BH+] and [B]
plug in and solve for Kb
if given pK instead of Ka/Kb → find K before ICE
not all salt solutions are neutral in terms of pH
salts go through hydrolysis reactions in water (acid/base reaction with water) → slightly acidic/basic solution
ionic compounds dissociate in water → ions
ions can react w water in acid-base reactions
NaOH and HCl are strong
salts of strong acids + strong bases
strong acids/bases → fully dissociate → salts do not have acid-base activity
conjugates of strong acids/bases do not hydrolyze
salt solutions formed from neutralization reactions btwn strong acid/base are neutral (pH = 7)
salts of weak acids + strong bases
produce solutions w pH > 7
salts of strong acids + weak bses
produce solutions w pH < 7
salts of weak acids + weak bases
pH of salt solution depends on Ka and Kb of species in reaction
Ka = Kb → neutral (7)
Ka > Kb → acidic (<7)
Ka < Kb → basic (>7)
stronger species dictates pH
equal = neutral 7
acid > base → pH < 7
acid < base → pH > 7
steps to determine pH of a salt solution
write salt dissociation equation
salt = conjugate
write hydrolysis equation for conjugate species that hydrolyzes
conjugates of strong acids/bases do not hydrolyze
use Ka or Kb to find [H+] or [OH-]
use ICE table
calculate pH
indicator - species that changes color depending on pH
litmus paper
blue + acidic → red
red + basic → blue
universal indicator
pH paper
solution
phenolphthalein
titration lab
indicator composition
weak acid/base where dissociated/undissociated forms have different colors
end point - point where indicators change colors during titrations
usually equivalence point
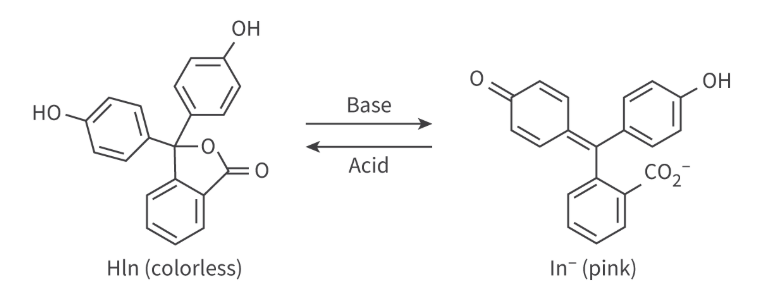
Example: Phenolphthalein
adding acid (more H+) → reaction shifts left to Hln → solution looks colorless
adding base (less H+) → reaction shifts right to ln- → solution looks pink
for all indicators
adding acid → equilibrium shifts to undissociated form Hln
solution = color of HIn
adding base → equilibrium shifts to dissociated form ln-
solution = color of ln-
indicators change color when pH = pKa
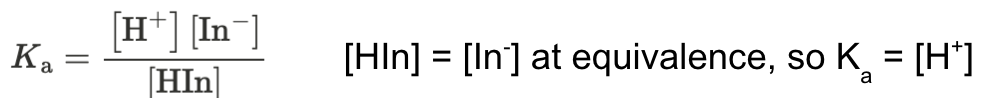
choosing an indicator
determine type of titration
determine pH of salt solution at equivalence
choose indicator whose pKa is closest to pH at equivalence point
table of indicators/ranges in data booklet*
buffer - solution that resists changes in pH when small amounts of acid/base added
work via Le Chat
quantity of H+ shifts equilibrium
Making buffers
1: make solution of weak acid/base + conjugate salt
acid buffer - weak acid + salt of conjugate base
base buffer - weak base + salt of conjugate acid
counter-ion of salt must come from strong species → does not have acid/base activity group
group 1 cation or halogen anion
equal amounts acid/base + salt added → buffer activity works equally in both directions
2: half neutralization - weak + strong
half neutralization of weak acid/base + strong acid/base
titrate → mols of strong = ½ mols of weak
stoich
buffer region of pH curve → solution acts as buffer
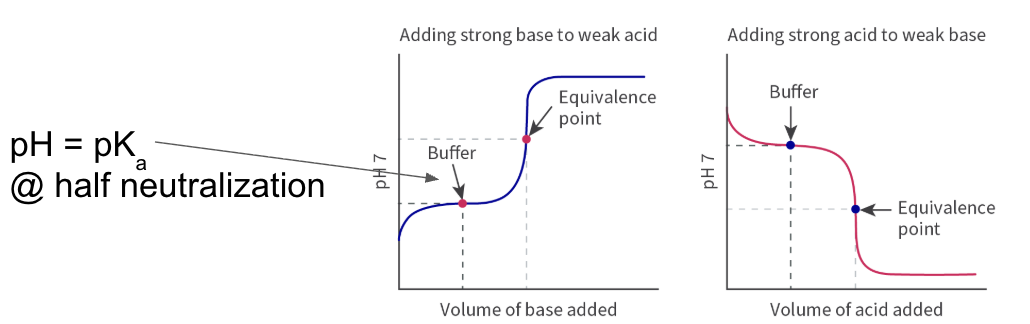
pH of buffer determined by
pKa or pKb
relative ratio of concentrations of acid/base and conjugate
adding 2 solutions (weak + conjugate) → dilution
need calculate mols and new molarity for ICE table
Ka/Kb used to determine [H+]/[OH-] then pH caluclated
dilution does not change number of mols
changes concentrtion
higher concentration → greater buffering capacity
effectiveness to resist change in pH