CHEM2070 Midterm #2 Study Guide
Exam Guidelines for CHEM2070 Midterm #2
Date and Time: Thursday, 4/9, 1pm – 2:10pm
Study Materials in Order of Importance
Lecture Notes & Slides
Include crucial components such as:
Definitions of key terms.
Practice problems along with slides that include answers.
Exam Format:
The exam will consist of 40 multiple choice questions that cover:
Calculations
Definitions
Questions similar to practice problems and homework.
Topics Covered in the Notes and Slides
Chapter 11: Hydrocarbons
Key Definitions:
Organic vs. Inorganic:
Organic compounds contain carbon, while inorganic compounds typically do not.

Saturated vs. Unsaturated:
Saturated hydrocarbons contain single bonds only, while unsaturated hydrocarbons contain double or triple bonds.

Alkanes:
Naming conventions for alkanes with 1 to 4 carbons.
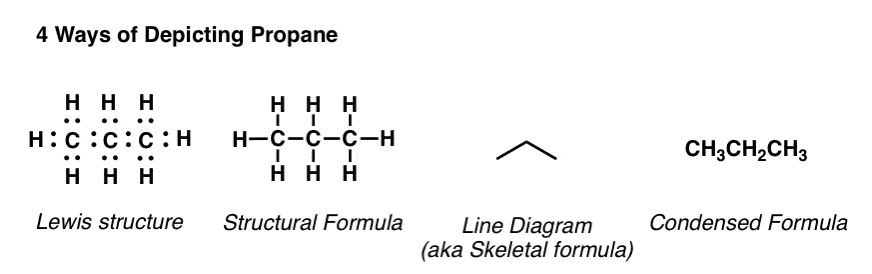
Structural formulas:
Condensed Formula: A simplified version that shows only the connections between atoms.
Expanded Formula: A full structural representation showing every bond.
Line-Angle Formula: A shorthand notation for organic molecules that represents bonds using lines.
Molecular Formula: A chemical formula that shows the number and type of atoms present.
Cyclic Alkanes: Alkanes that form cyclic structures.
Alkanes with Substituents: Understanding IUPAC naming rules for alkanes with branches.
Properties of Alkanes:
Discuss properties based on carbon chain length.
Examine combustion and density of alkanes.
Alkenes and Alkynes:
Definitions of alkenes (double bonds) and alkynes (triple bonds).
Cis vs. Trans Isomers:
Isomers that differ in spatial arrangement.
Naming conventions for these isomers.
Aromatic Compounds:
Definition and characteristics of aromatic compounds.
Chapter 12: Alcohols, Thiols, Ethers, Aldehydes, and Ketones
Alcohols:
Structure and IUPAC naming rules for alcohols.
Phenol:
Structure and naming examples for phenolic compounds.
Thiols:
Basic structure and naming convention for thiol groups.
Ethers:
Structure and naming rules for ether compounds.
Properties of Alcohols:
Distinction among primary, secondary, and tertiary alcohols.
Solubility in water for alcohols depending on structure.
Ketones:
Definition and IUPAC naming rules for keto compounds.
Aldehydes:
Definition and IUPAC naming rules for aldehyde groups.
Water solubility characteristics of aldehydes and ketones.
Chapter 13: Carbohydrates
Functions and Chemical Formula:
Overview function of carbohydrates and general formula.
Monosaccharides:
Names based on chain length and types:
Aldo vs. Keto classifications.
Numbering of carbon atoms.
Chiral Molecules:
Definition of chiral molecules.
Enantiomers:
Definition and characteristics of enantiomers.
Fischer Projections:
Definition, numbering rules, and assigning D vs. L configurations.
Examples: D-galactose, D/L-glucose.
Importance in Health:
Role of monosaccharides in health and blood sugar management.
Specific examples include D-fructose and their structural relevance.
Haworth Structure:
Definition including alpha vs. beta isomers of monosaccharides.
Discussion of mutarotation and 5-membered Haworth structures.
Chemical Properties of Monosaccharides:
Reactions including aldehyde oxidation and reduction.
Disaccharides:
Definition and examples:
Maltose: Component parts and bond type.
Lactose: Example in disaccharides.
Sucrose: Example in disaccharides.
Polysaccharides:
Definition and functions in biological systems.
Major examples: Amylose, Amylopectin, Glycogen, Cellulose.
Chapter 15: Lipids
Definitions, Examples, and Functions:
Overview definition of lipids and their various functions in biological contexts.
General Classifications:
Fatty acids and steroids as primary categories of lipids.
Fatty Acids Structures:
Saturated Fatty Acids: Characterized by single bonds.
Lipid Number Nomenclature: Understanding how lipids are named based on carbon chain length and saturation.
Monounsaturated Fatty Acids: Structures and distinctions (cis vs. trans).
Polyunsaturated Fatty Acids: Description and significance.
Properties of Fatty Acids:
Comparative properties of saturated vs. unsaturated fatty acids.
Omega Fatty Acids: Importance and definitions for these fatty acids.
Waxes:
Definition and chemical structures of waxes.
Triacylglycerols:
Definition, structure, types of bonds, and understanding mixed triacylglycerols.
Chemical Properties:
Reactions including hydrogenation reactions, saponification, and hydrolysis.
Phospholipids:
Definition, function, structure, and components including head groups.
Sphingomyelins:
Definition and primary components.
Steroids:
Structure and classification of steroids.
Types of steroids discussed:
Cholesterol
Bile salts
Lipoproteins
Steroid hormones.
Cell Membranes:
Definition and introduction to the fluid mosaic model of cell membranes.
Types of transport mechanisms used by the membranes.
Additional Study Resources
Homework Problems:
Relevant chapters for practice problems include:
Chapter 11, 12, 13, 14, and 15.
Recommendation to rework these problems and identify any areas of confusion.
Textbook:
Suggestions for chapters to review to fill in any understanding gaps:
Chapter 11: Parts 1, 2, 3, 4, 5, 6, 8
Chapter 12: Parts 1, 2, 3
Chapter 14: Parts 1, 2, 3, 4, 5, 6
Chapter 13: Parts 1, 2, 3, 4, 5, 6, 7
Chapter 15: Parts 1, 2, 3, 4, 5, 6, 7
Additional Materials:
PowerPoint Presentations: "Examples of Worked Problems".
Audio Recordings: For supplementary learning.
What to Bring to the Exam
Required Materials:
Green Scantron form
Pencil
Student ID (to be shown when handing in the exam, or during distribution).
Calculator (non-graphing, no cell phones allowed).