Cycloalkane Nomenclature, Functional Groups, and Degree of Substitution
Cycloalkane Nomenclature
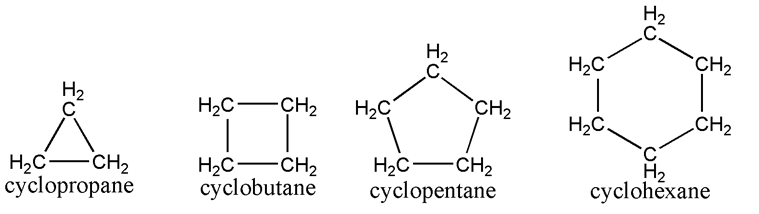
- - alkanes where all the carbons are bonded to each other in a ring-shaped formation
General Rules
- use the prefix cyclo- in front of the parent chain name indicating how many carbons there are
- example- use cyclopentane to refer to a ring-shaped alkane with 5 carbons instead of pentane
- use the same rules for naming substituents as you would with alkanes
- if there is only one substituent, the carbon that the substituent is attached to is automatically carbon 1.
- you don’t need to put a number in front of the substituent
- if there is more than one substituent, the carbon attached to the substituent whose name comes first in the alphabet is carbon one
Examples
try to solve them before looking at the answers
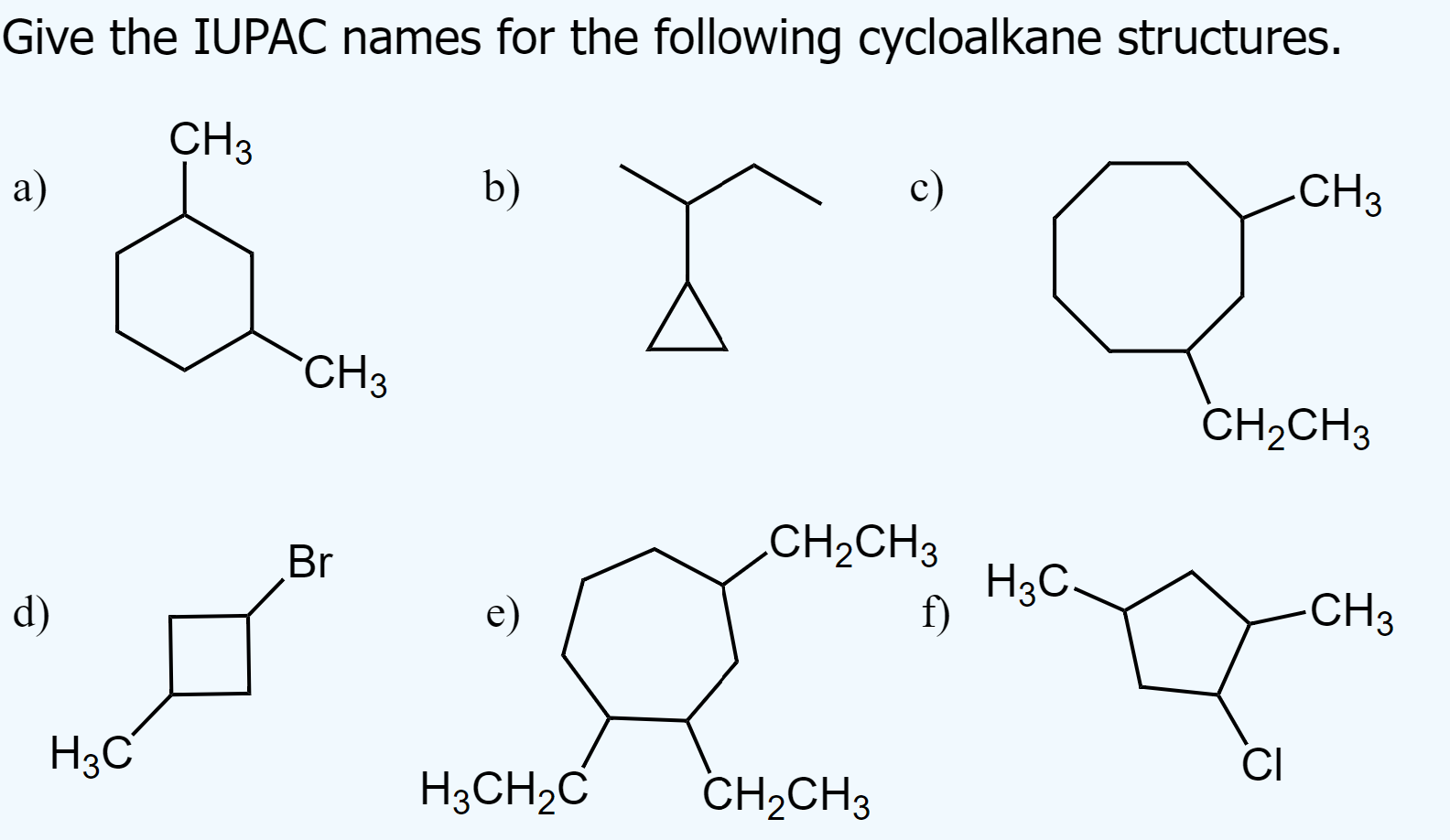
a) 1,3-dimethylcyclohexane
b) 2-cyclopropylbutane
c) 1-ethyl-3-methylcyclooctane
d) 1-bromo-3-methylcyclobutane
e) 1,2,4-triethylcycloheptane
f) 1-chloro-2,4-dimethylcyclopentane
Functional Groups
- - there is an OH group off a carbon
1. change the ending from -ane to -anol add the carbon number that the OH group is attached to 2. example- 1-propanol
- - looks like R-O-R where the R group is another alkane
1. end the molecule’s name with ether 2. example- dimethyl ether
- - alkanes with halogens
1. example- chloromethane or methyl chloride
- - a nitrogen atom bonded to between 1-3 carbons/carbon chains and the rest being hydrogens
1. use the suffix -amine 2. example- ethylamine
- - a carbon at the end of the parent chain double-bonded to an oxygen atom
1. use the suffix -al 2. example- ethanal
- - a carbon within the parent chain double-bonded to an oxygen atom
1. use the suffix -anone 2. example- propanone
- - looks like:
1. 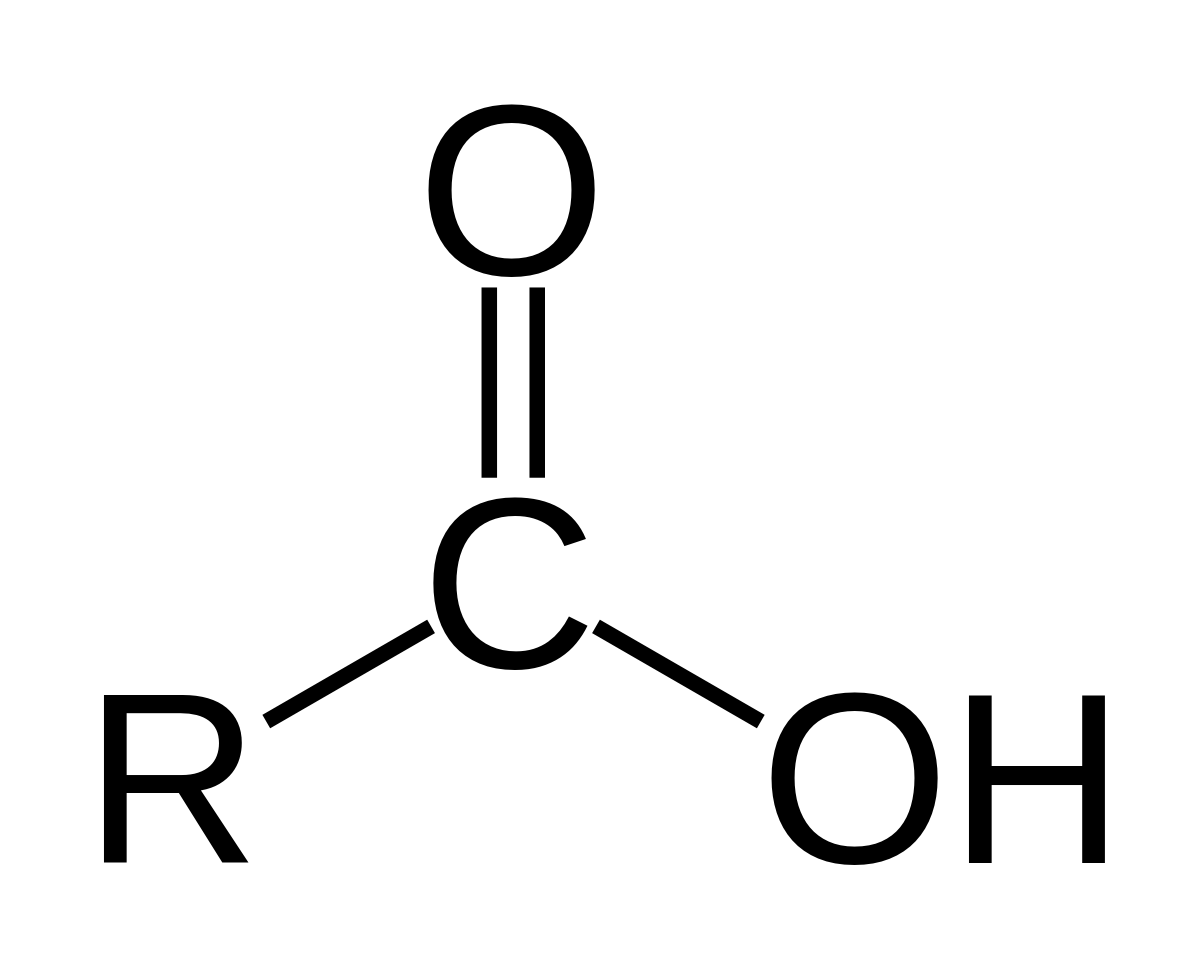 2. use the suffix -oic acid
3. example- ethanoic acid
2. use the suffix -oic acid
3. example- ethanoic acid
- - looks like:
1. 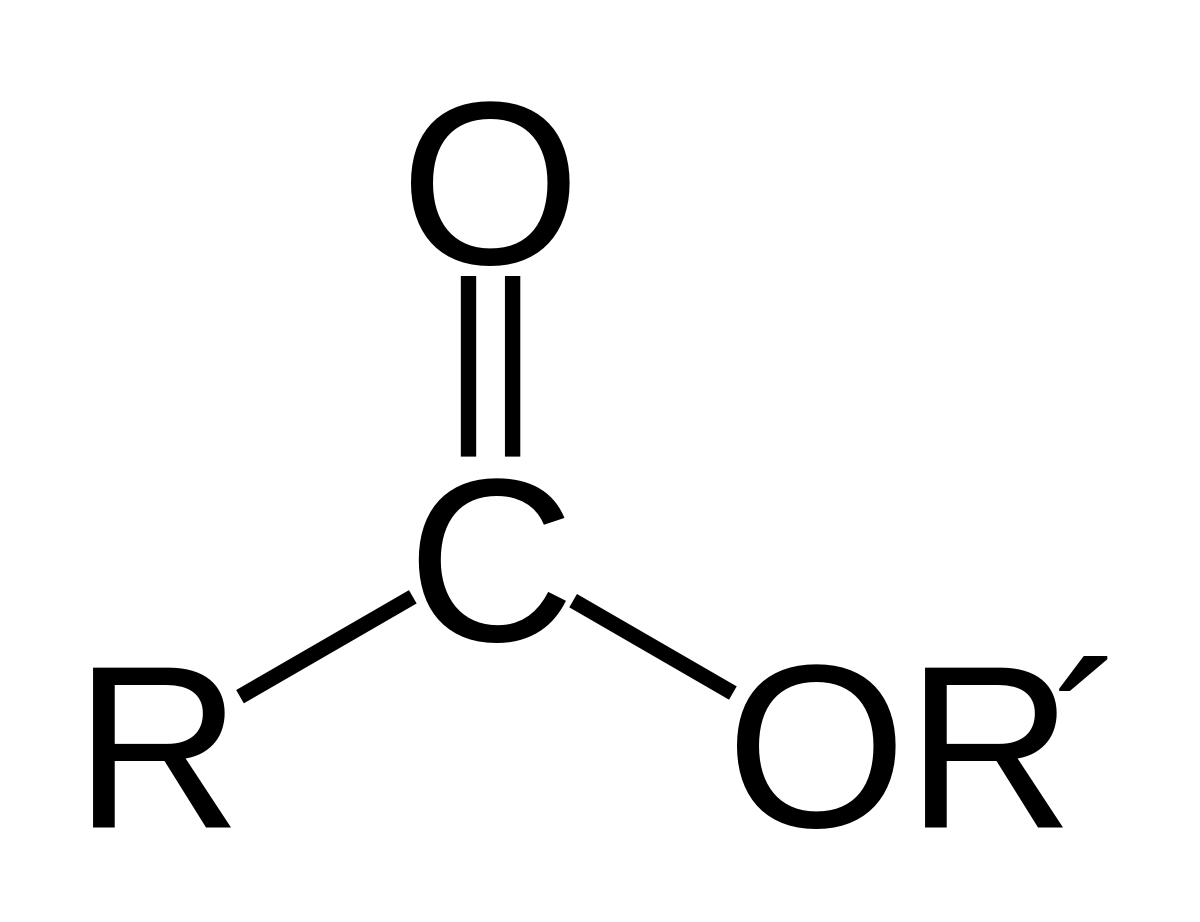 2. use the suffix -oate
3. example- ethanoate
2. use the suffix -oate
3. example- ethanoate
- - looks like:
1. 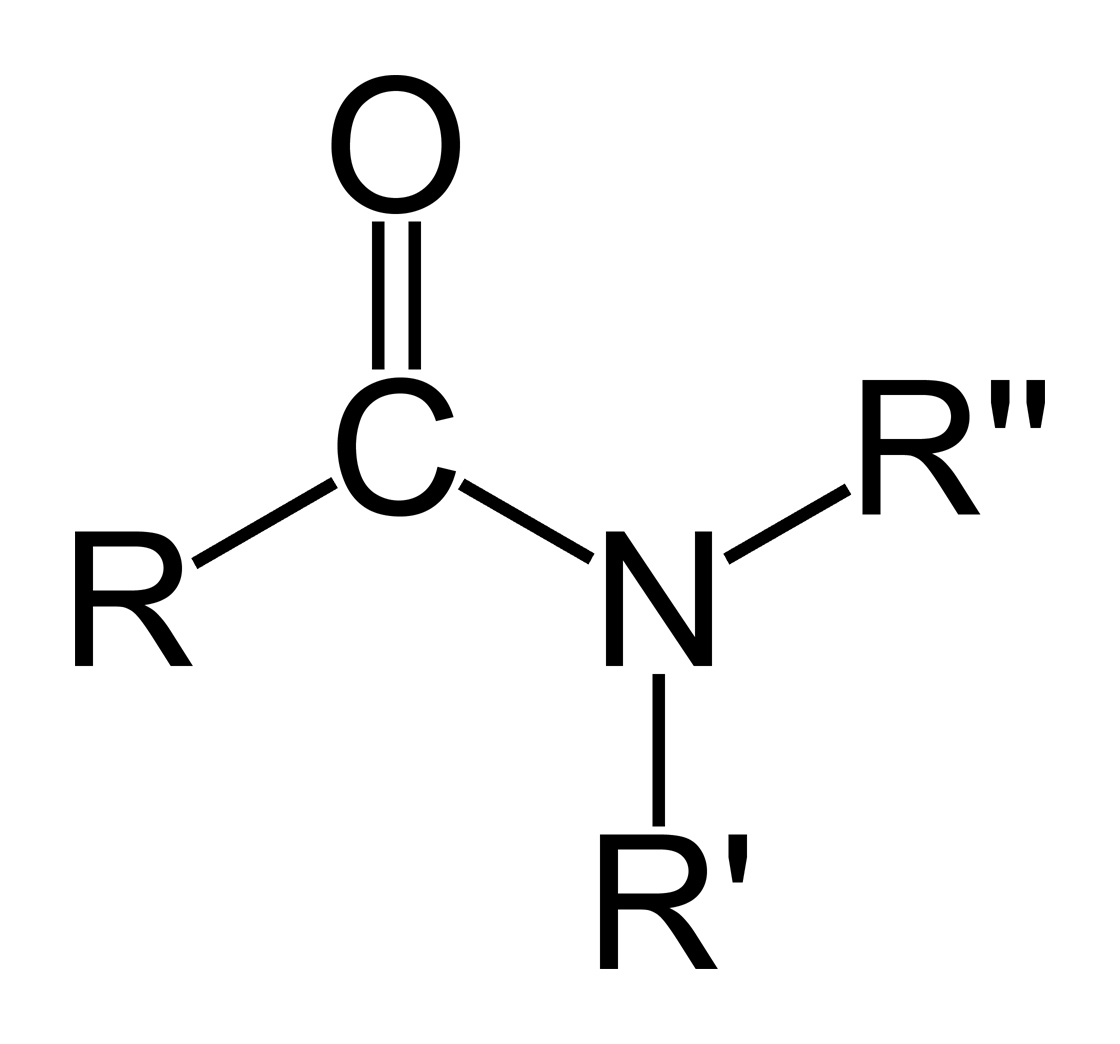 2. use the suffix -amide
3. example- ethanamide
2. use the suffix -amide
3. example- ethanamide
- - a six-carbon ring with alternating double and single bonds
1. also called benzene 2. use the suffix -benzene if it’s the parent chain and the prefix -phenyl if it’s considered a substituent
Degree of Substitution
Carbons and Amines
- - one carbon/carbon group is attached to it
- - two carbons/carbon groups are attached to it
- - three carbons/carbon groups are attached to it
- amines end here
- - four carbons/carbon groups are attached to it
Alcohols and Haloalkanes
- - one carbon/carbon group is attached to the carbon that is attached to it
- - two carbons/carbon groups are attached to the carbon that is attached to it
- - three carbons/carbon groups are attached to the carbon that is attached to it