Chemical formulae
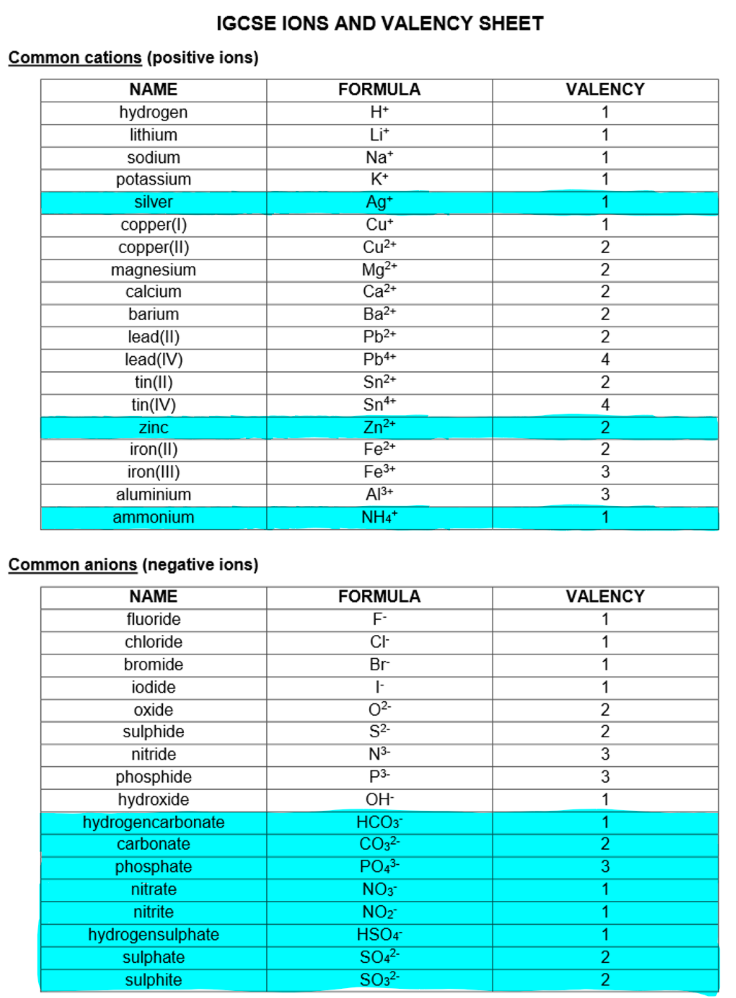
Chemical formula: A way to express the elements (atoms or ions) in a compound, showing the number of atoms/ions of each element using symbols and numbers.
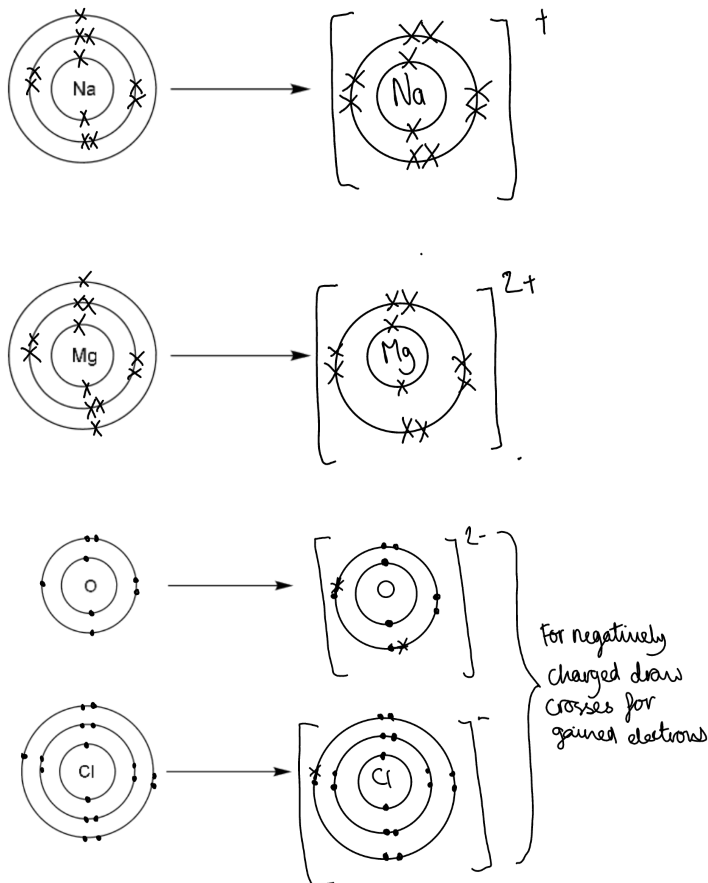
Ion formation:
Ions are charged atoms or groups of atoms formed by gaining or losing electrons.
Metal and hydrogen atoms lose electrons to form positively charged ions (cations).
Non-metal atoms gain electrons to form negatively charged ions (anions).
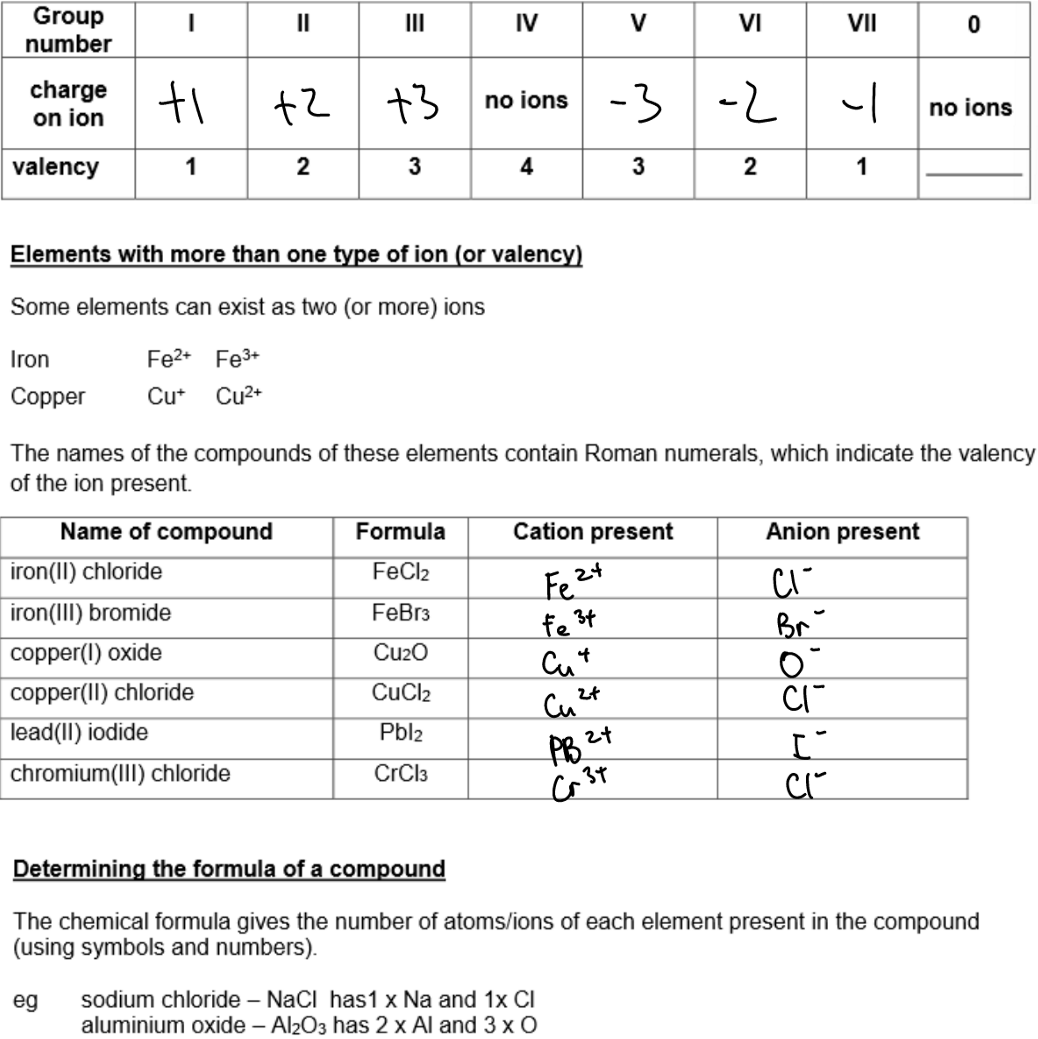
Group 4 elements DON'T FORM IONS!
This is because they will become so positively charged they just gain all the electrons back again.

Valency:
The number representing the combining power of an atom (or group of atoms).
It is equal to the number of electrons an atom needs to lose or gain to achieve a stable, full outer electron shell.

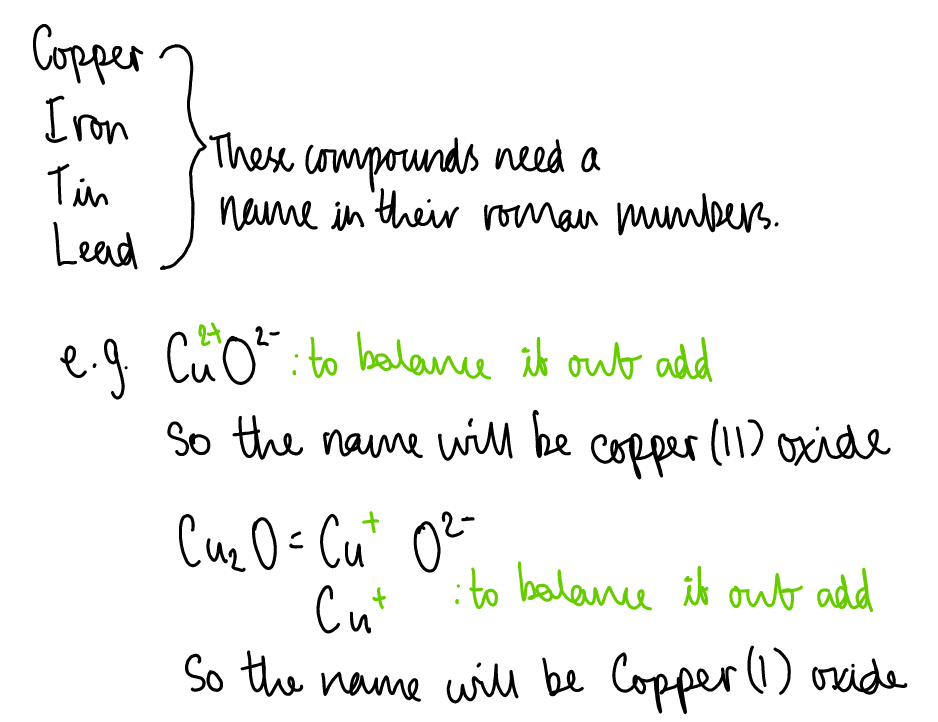
Diatomic molecules:
Diatomic molecules are molecules formed when 2 atoms chemically join together. Some elements exist as diatomic molecules. E.g.
