BIOL111
Chapter 1
1.1
What is science? - Knowledge that covers general truths r operation of general laws, especially when aquired or tested by the scientific method.
What is Biology? - The study of life. Biologists study everything from submicroscopic views of cells to the entire planet as well as living organisms and vital processes.
The Scientific Method:
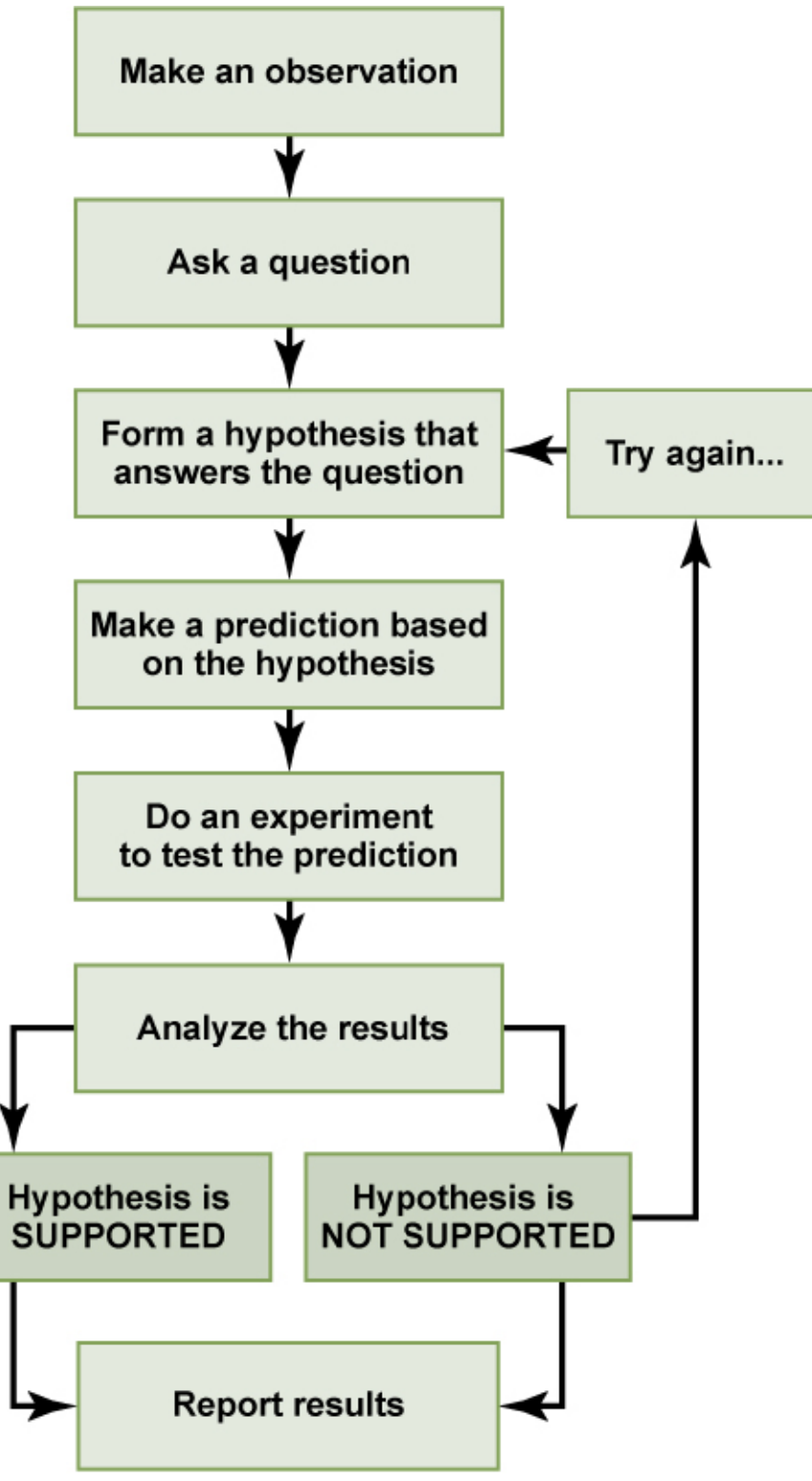
Basic Science vs. Applied Science:
Basic: Science for the sake of knowledge with no immediately apparent use of the information.
Applied: Goes into science with a problem and experiments to try and solve it.
Inductive vs. Deductive Reasoning
Inductive: When a conclusion is used to predict the results of a specific situation. Hypothesis based science. Takes a general fact and applies it to something else.
Deductive: Takes a generally known fact and applies i to a specific instance. When a conclusion is drawn from observations.
Basic Characteristics of Life:
1. Metabolism
2. Growth
3. Reproduction
4. Order
5. Response to Stimuli
6. Evolutionary Adaptation
7. Homeostasis.
Cell Theory:
- All cells arise from other cells.
- All living things are 1 or more cells.
- Chemical reactions necessary for life (AKA metabolism) occur within the cell.
- Cells contain hereditary information in the form of DNA
- Cells are the smallest functional unit of life.
Biological Heirarchy:
- Like nesting dolls. Smaller things play a role in a bigger system.
1. Atom (smallest)
2. Molecult
3. Organelle
4. Cell
5. Tissue
6. Organ
7. Organ System
8. Organism
9. Population
10. Community
11. Ecosystem
12. Biosphere (largest)
1.2
Section goals:
• Identify and describe the properties of life
• Describe the levels of organization among living things
• Recognize and interpret a phylogenetic tree
• List examples of different subdisciplines in biology
Properties of Life:
- Order: Organisms are highly organized structures made of one or more cells. Cells go on to make higher structures (tissues → organ → organ system, etc.)
- Sensitivity (AKA Response to Stimuli): Living things react to stimuli. Movement towards stimuli is “positive stimuli”, movement away from stimuli is “negative stimuli”
- Reproduction: When reproduction occurs, DNA containing genes are passed along to an organism’s offspring.
- Adaptation: Enhances reproductive potential including ability to survive and reproduce.
- Growth & Development: Organisms grow because of genes providing specific instructions that direct cellular growth and development. This ensures species’ young exhibit many characteristics of the parent.
- Regulation/Homeostasis: Organisms maintain a narrow range of internal conditions through homeostasis.
- Energy Processing: METABOLISM. Living things require a source of energy, metabolism is how it processes it.
- Evolution: Mutations allow possibility for organisms to adapt. Organisms that evolve to fit environment are more likely to live and reproduce.
Chapter 2
Class
Moderation of Temperature
Water moderates air temperature
Absorbs heat form air that is warmer and releases stored heat to air that is cooler.
Allows water to minimize temperature fluctuations.
Heat vs. Temperature:
Heat - Thermal energy. IE energy from object A (hot) transfers to object B (cold). Describes avg. energy level in a system.
Temperature:
Water has a high specific heat
Specific heat: The amount of heat that must be absorbed or lost for 1 gram of that substance to change temp. by 1*C.
Water has a high specific heat.
Heat absorbed when hydrogen bonds reak, released when they form.
Evaporative cooling
Water has a high heat of vaporization (related to cohesion by hyrdorgen bonds.)
Expansion upon freezing.
Water expands as it freezes.
Solute concentration in solutions
Chemical reactions are affected by concentrations of reactants and products.
Most biochem reactions occur in water.
Concentration - The amount of solute in a defined volume.
i.e. blood alcohol testing, blood sugar.
What is a mole?
Represents an exact number of molecules.
Similar to how a dozen = 12.
6.02 × 10²3 molecules (avogadro’s number).
Molar Mass
a mole = quantity of a substance that has the same number of atoms found in 12g of carbon-12.
molar mass (g/mol) is defined as the mass of one mole of a substance.
AKA molecular weight.
The mass of one mole of a substance is atomic mass
Molarity
AKA molecular weight.
The mass of one mole of a substance is atomic mass number of moles per lirre of solile.
Write aswers im MMOL \
Understanding PH
SI 9/11
Acids/Bases/pH Scale
Acid Def: Solution that has a high concentration of hydrogen ions.
Base Def: Solution that has a high concentration of hydroxide ions.
The more difference between hydroxide and hydrogen ions is the stronger base/acid.
pH Def: Potential of hydrogen.
Practice solving pH Questions
pH = -log[H+]
Problem Set Question.
Diffusion = tendency for molecules of a substance to spread evenly throughout the celll\
Substances
Osmosis: movement of WATER SPECIFICALLY across a semipermeable membrane. Affected by concentration gradient of dissolved substances.
Tonicity: the ability of a soliutation to cause a cell to gain or lose water. Has create impact on cells w/o water (ie animal cells)
10/4/24
Cell division results in 2 daughter cells.
Each gets a copy of DNA (which replicates before cell division)
Genome consists of prokaryotes and eukaryotes
Prokaryotes
1 double-stranded circular DNA in the nucleoid.
Has smaller loops of DNA (Plasmids) that are not necessary to grow but have important information
To transfer genes in prokaryotes they transfer plasmids.
Eukaryotes
Several linear double stranded DNA in form of chromosomes.
Learn chromosome structure (how double helix forms structures that eventually results in the metaphase chromosome.
Chromosome = 2 chromatids. Chromatids separate during cell division.
Chromosomes made of chromatin (DNA & Protein) that condenses during cell divison.
In animals:
Somatic cells have two sets of chromosoomes.
Gametes (SEX CELLS) have one set of chromosomes
Cell cycle occurs in phases
Mitotic phase
Interphase
G1, S, G2
G1 - First Gap: No change evident but cell is biochemically active
Cell growth, increase in cytoplasm, synthesis of protein.
S Phase: DNA Synthesis and replication occurs.
G2: Second Gap
- Cell grwth, nergy replenished, organelles reprduce, cytoskeleton breaks down.
Cell divison = Mitosis - Nucleus divides Cytokinesis - The division of cytoplasm.
Mitosis = PPMAT