Birth & Lifespan II: Lactation and the Oxytocin System
Introduction
This section focuses on the physiological processes enabling lactation, with a particular emphasis on the role of the oxytocin system. It will cover:
The structural features of the magnocellular neurosecretory system involved in oxytocin production.
The diverse actions of oxytocin, particularly in parturition and lactation.
The essential role of oxytocin in milk ejection, as demonstrated by knockout studies.
Adaptive changes in the oxytocin system during pregnancy and lactation, including:
Oxytocin accumulation (mRNA expression, steroid regulation, neuronal quiescence).
Action potential synchrony in oxytocin neurons (neuronal insulation, synchronized inputs).
Increased mammary gland sensitivity to oxytocin (receptor expression and regulation).
The structure and signaling pathways of the oxytocin receptor.
The Magnocellular Neurosecretory System
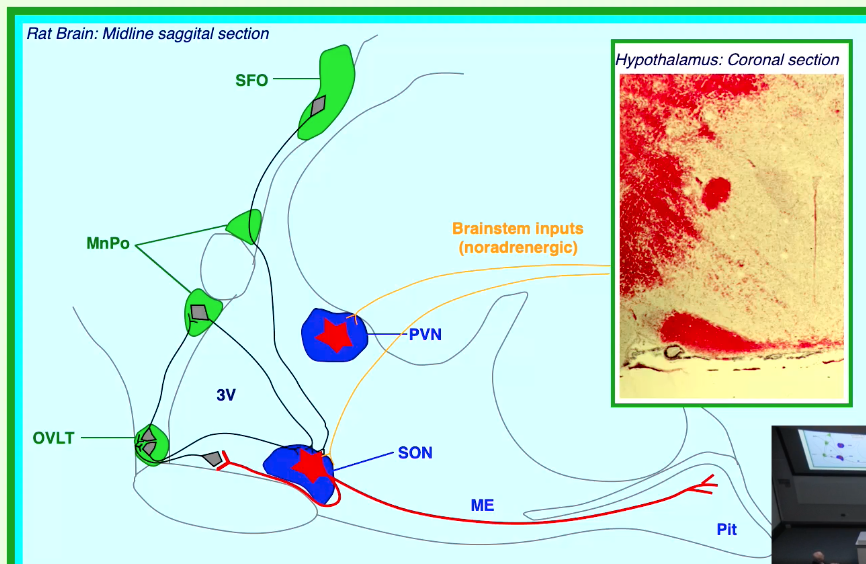
Oxytocin is a hormone produced by magnocellular neurons located in specific nuclei of the hypothalamus.
Key Hypothalamic Nuclei:
Supraoptic Nucleus (SON)
Paraventricular Nucleus (PVN)
Projections: Axons from these magnocellular neurons project to the posterior pituitary (neurohypophysis).
Storage and Release: Oxytocin (and vasopressin) are synthesized in the cell bodies in the SON and PVN, transported down the axons, and stored in nerve terminals in the posterior pituitary. Upon appropriate stimulation, they are released into the bloodstream.
Inputs to the System:
Circumventricular Organs: Regions like the Organum Vasculosum of the Lamina Terminalis (OVLT), Subfornical Organ (SFO), and Median Preoptic Nucleus (MnPo) can sense factors in the blood and project to the SON/PVN.
Brainstem Inputs: Noradrenergic inputs from the brainstem also modulate the activity of these hypothalamic nuclei.
Actions of Oxytocin
Oxytocin has several well-defined physiological roles:
1. Parturition (Childbirth)
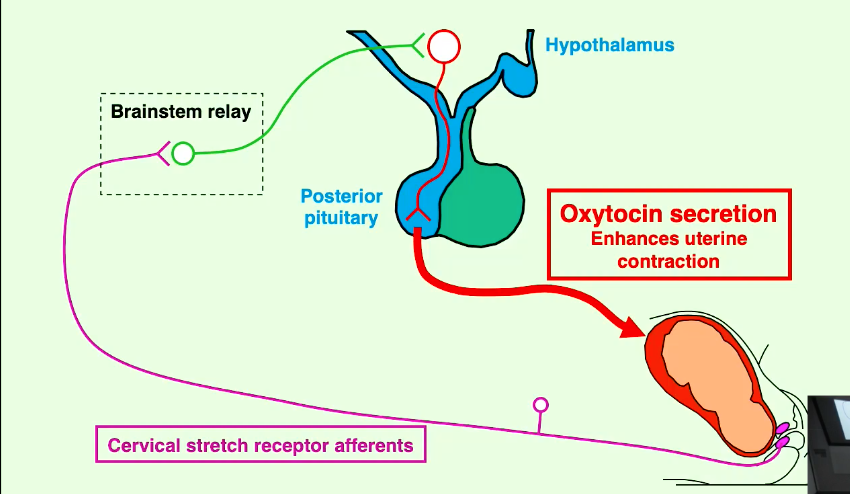
During labour, stretching of the cervix by the fetus initiates a neurohormonal reflex (Ferguson reflex).
Afferent signals from cervical stretch receptors are relayed via the brainstem to the hypothalamus.
This stimulates the release of oxytocin from the posterior pituitary.
Oxytocin then acts on the uterus (myometrium) to enhance uterine contractions.
This forms a positive feedback loop, where uterine contractions cause further cervical stretch, leading to more oxytocin release and stronger contractions, ultimately culminating in birth.
2. Lactation (Milk Ejection)
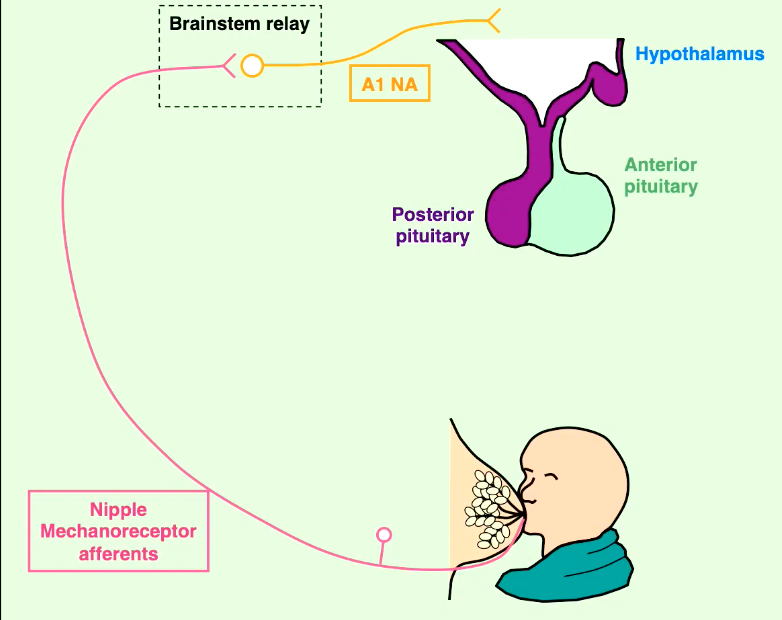
Suckling by the infant at the nipple stimulates mechanoreceptors.
Afferent nerve impulses are relayed via the brainstem (e.g., A1 noradrenergic neurons) to the hypothalamus.
This triggers the release of oxytocin from the posterior pituitary and prolactin from the anterior pituitary.
Oxytocin's Role: Causes contraction of myoepithelial cells surrounding the alveoli and ducts in the mammary gland, leading to milk ejection (the milk let-down reflex).
Prolactin's Role: Stimulates milk secretion (synthesis) by the epithelial cells of the mammary alveoli.
Other Effects during Lactation:
Suckling-induced hormone release can also inhibit ovulation by affecting Gonadotropin-Releasing Hormone (GnRH) and Prolactin Inhibiting Hormone (PIH, likely dopamine), thereby suppressing LH/FSH release (lactational amenorrhea).
Oxytocin and other neurochemical changes (e.g., involving opioids) contribute to maternal behaviour.
Essential Role of Oxytocin in Lactation: Evidence from Knockout Mice
Studies on oxytocin-knockout mice (homozygous -/-) have clarified its essential functions.
Findings:
Normal gestation length and litter size.
Normal parturition (childbirth process).
Mammary glands produce normal milk.
Normal maternal behaviour is observed.
However, pups born to oxytocin-knockout mothers die within 2 days due to inability to obtain milk.
Lactation (milk ejection) can be restored in these mothers by administering oxytocin.
Conclusion: These findings demonstrate that oxytocin is only essential for lactation (specifically, for milk ejection) and not for parturition or reproductive behaviour in mice under laboratory conditions.
Adaptations of the Oxytocin System for Lactation
The oxytocin system undergoes significant adaptations during pregnancy and lactation to ensure efficient milk ejection.
Adaptation 1: Oxytocin Accumulation
Increased stores of oxytocin are built up in the posterior pituitary in preparation for the demands of parturition and lactation.
a. mRNA Expression:
The expression of oxytocin mRNA significantly increases in the Paraventricular Nucleus (PVN) of the hypothalamus during late pregnancy and, most markedly, during lactation. For example, on day 5 of lactation, oxytocin mRNA levels can be nearly double those seen in virgin rats.
b. Steroid Regulation:
Hormonal changes during pregnancy play a key role in regulating oxytocin mRNA expression.
Estrogen: Levels rise significantly during pregnancy. Estrogen treatment in ovariectomized (OVX) female rats mimics the changes in oxytocin mRNA expression seen during pregnancy, suggesting it stimulates oxytocin synthesis.
Progesterone: Levels are high during pregnancy and fall around parturition. Progesterone treatment in OVX rats inhibits the estrogen-induced changes in oxytocin mRNA expression and can prolong pregnancy. This suggests progesterone normally suppresses oxytocin synthesis/accumulation during gestation.
c. Neuronal Quiescence (Opioid Suppression):
During pregnancy, there is an increased opioid suppression of the activity of oxytocin neurons in the SON. This helps prevent premature release of oxytocin and allows for its accumulation.
Administration of naloxone (an opioid receptor antagonist) to 21-day pregnant rats causes a significant increase in plasma oxytocin levels and oxytocin release from the SON, indicating that an endogenous opioid tone is actively suppressing release.
Summary of Accumulation: During pregnancy, rising estrogen levels promote oxytocin synthesis, while high progesterone and increased opioid tone suppress its release, leading to a net accumulation of oxytocin.
Adaptation 2: Action Potential Synchrony in Oxytocin Neurons
For effective milk ejection, a large bolus of oxytocin needs to be released synchronously. This requires coordinated firing of oxytocin neurons.
Suckling Response and Pulsatile Release:
Suckling by pups triggers high-frequency bursts of action potentials in oxytocin neurons.
Each burst results in a pulse of oxytocin release, leading to a transient increase in intramammary pressure and milk ejection.
These bursts are highly synchronized across the population of oxytocin neurons in both the SON and PVN.
Mechanisms Enhancing Synchrony:
a. Neuronal Insulation (Glial Retraction):
During lactation, there is a remarkable structural plasticity in the hypothalamus. Astrocytic glial cells (which normally ensheath neurons and synapses) retract their processes from oxytocin neurons.
This reduction in glial coverage increases direct neuron-neuron apposition (juxtaposition of neuronal cell bodies and dendrites) and the number of shared synapses.
This "uncovering" of neuronal membranes is thought to facilitate intercellular communication (e.g., via ephaptic interactions or local peptide release) and promote synchronous firing. GFAP (Glial Fibrillary Acidic Protein) is a marker for astrocytes, and changes in its expression/morphology are observed.
b. Synchronized Inputs:
An increase in shared synaptic inputs, particularly GABAergic inputs, onto clusters of oxytocin neurons is observed during lactation. This can help coordinate their activity. Double synapses, where one presynaptic terminal contacts two postsynaptic oxytocin neurons, are also more common.
Summary of Synchrony Factors: During lactation, glial retraction and increased shared synaptic inputs, along with local effects of oxytocin (autoregulation) and reduced opioid inhibition, contribute to the synchronous, high-frequency firing of oxytocin neurons necessary for pulsatile milk ejection.
Adaptation 3: Mammary Gland Sensitivity to Oxytocin
The mammary gland itself becomes more responsive to oxytocin.
a. Mammary Gland Structure:
The mammary gland is composed of glandular tissue (alveoli and ducts) embedded in pectoral fat, supported by suspensory ligaments.
Alveoli: Milk is synthesized by epithelial milk-secreting cells lining the alveoli.
Myoepithelial Cells: A network of contractile myoepithelial cells surrounds the alveoli and ducts. These are the target cells for oxytocin.
Muscle cells are also present in the walls of larger ducts.
b. Oxytocin Receptor Expression in Mammary Gland:
Oxytocin receptors (OTR) are expressed in mammary cells, specifically on the myoepithelial cells.
Increased Expression During Pregnancy: The concentration of oxytocin receptors in the mammary gland significantly increases during pregnancy, peaks around parturition, and remains high during lactation. This upregulation sensitizes the gland to oxytocin.
Differential Steroid Regulation:
Estrogen: Stimulates OTR mRNA expression in both the mammary gland and the uterus.
Progesterone: Has a complex role. While it generally promotes uterine quiescence, its effects on mammary OTR expression can differ. In some contexts, progesterone can inhibit estrogen-induced OTR expression.
Tamoxifen (TAM), an estrogen receptor modulator, can block estrogen's effects on OTR expression.
c. Steroidal Interactions at the Receptor Level:
Estrogen: Increases OTR expression.
Progesterone: While its primary role during pregnancy is to maintain uterine quiescence, at the level of the mammary gland, progesterone may also modulate oxytocin action. Some evidence suggests progesterone might cause allosteric hindrance of the oxytocin receptor, reducing its binding affinity or signaling efficiency until progesterone levels fall at parturition.
Summary of Adaptations: During pregnancy, oxytocin accumulates due to hormonal influences (increased estrogen, progesterone suppression of release, opioid inhibition). During lactation, neuronal synchrony (facilitated by glial changes and local oxytocin) and increased mammary gland sensitivity (due to upregulated OTR expression, influenced by estrogen and falling progesterone) ensure efficient, pulsatile oxytocin release and effective milk ejection.
The Oxytocin Receptor (OTR)
Structure:
The OTR is a 389-amino acid G protein-coupled receptor (GPCR) belonging to Class 1 (specifically the Oxytocin/Vasopressin subfamily).
It has the typical seven transmembrane domain structure.
The gene for OTR was cloned by Kimura et al. in 1994 and is located on chromosome 3 in humans.
OTRs are expressed in various central (brain) and peripheral tissues (e.g., uterus, mammary gland).
Regulation:
OTR function can be regulated by:
Dimerization: OTRs can form homodimers (OTR-OTR).
Heterodimerization: OTRs can form heterodimers with other GPCRs, which can modify their signaling properties.
Internalization: Upon ligand binding, OTRs can be internalized (removed from the cell surface), which is a mechanism for desensitization and receptor downregulation.
Signaling Pathways:
The OTR primarily couples to Gq/11 G proteins.
Upon oxytocin binding, the activated Gq/11 protein (specifically the Gαq subunit) stimulates Phospholipase C (PLC).
PLC hydrolyzes phosphatidylinositol 4,5-bisphosphate (PIP2) into inositol trisphosphate (IP3) and diacylglycerol (DAG).
IP3 triggers the release of Ca2+ from intracellular stores (sarcoplasmic/endoplasmic reticulum).
DAG, along with Ca2+, activates Protein Kinase C (PKC).
The rise in intracellular Ca2+ is the primary signal for contraction in myoepithelial cells (and uterine smooth muscle).