Lesson 5: Polar Bonds & Polar Molecule
Molecular Polarity
Polar Molecule - a molecule in which the uneven distribution of electrons results in a positive charge at one end and a negative charge at the other end \n
Non-polar Molecule - a molecule in which the electrons are equally distributed among the atoms resulting in no localized charges
Polar Molecules
Non-polar Molecules
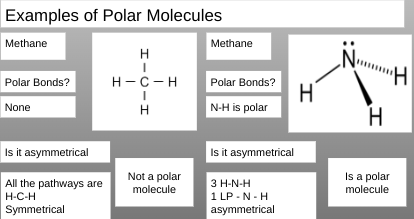
How to Determine if a Molecule is Polar or Non-polar
Draw the Lewis Structure of the molecule
Ask the question “are polar bonds present?”
Ask the question “is the molecule asymmetrical?”
What is meant by asymmetrical as it relates to molecules is there a different path from a peripheral atom through the central atom and on to another peripheral atom any where within the molecule \n
If the answer to both is yes then it is a polar molecule
Summary